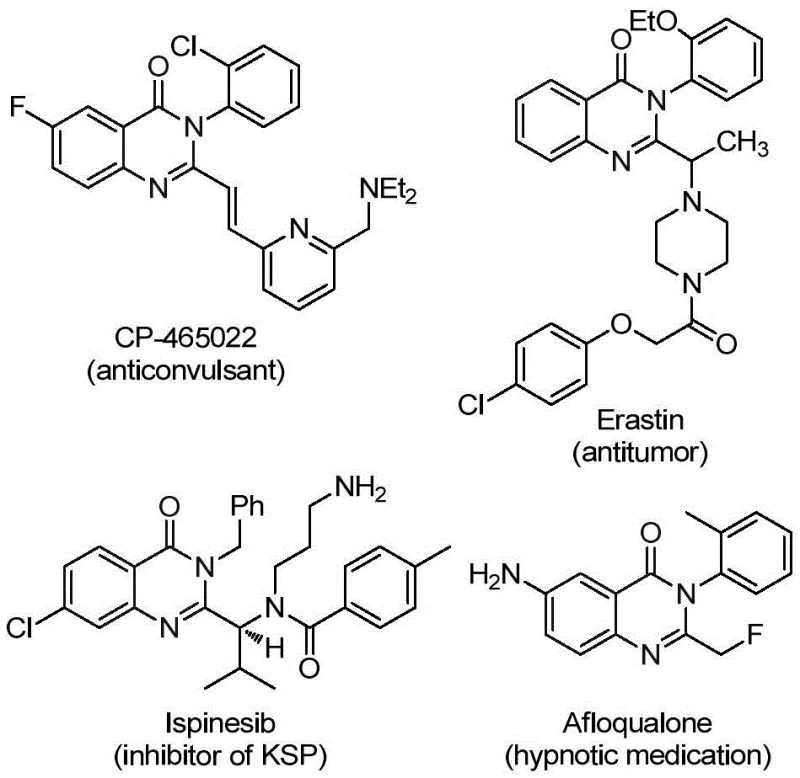
Advanced Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives: Safe, Scalable, and Cost-Efficient Manufacturing for Pharmaceutical Applications
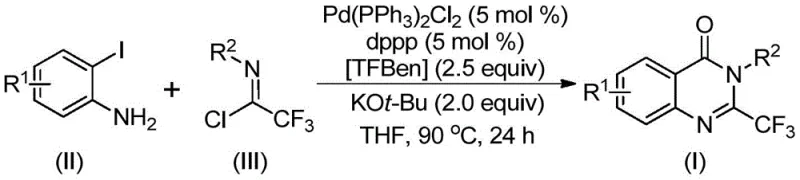
Patent CN112125856A introduces a groundbreaking methodology for synthesizing 2-trifluoromethyl-substituted quinazolinone derivatives through a palladium-catalyzed carbonylation process that fundamentally transforms the production landscape for these critical pharmaceutical intermediates. This innovative approach eliminates the hazardous requirement for gaseous carbon monoxide by employing 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid CO surrogate, addressing significant safety concerns while maintaining high reaction efficiency. The process demonstrates exceptional substrate versatility with various substituents on both coupling partners, enabling the production of diverse quinazolinone derivatives essential for modern drug development pipelines. By utilizing readily available starting materials including o-iodoaniline and trifluoroethylimide chloride under mild reaction conditions (90°C in THF), this method overcomes traditional limitations associated with harsh reaction parameters and expensive precursors. The resulting derivatives exhibit remarkable structural diversity that aligns with the molecular frameworks found in numerous therapeutic agents, positioning this technology as a strategic advancement for pharmaceutical manufacturers seeking reliable access to complex heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for 2-trifluoromethyl quinazolinones suffer from multiple critical deficiencies that impede their commercial viability for pharmaceutical manufacturing. Methods involving cyclization of anthranilamide with trifluoroacetic acid derivatives typically require harsh reaction conditions exceeding 150°C and generate significant impurities that complicate purification processes for high-purity intermediates. Alternative approaches using unstable trifluoroacetamide or isatoic anhydride with trifluoroacetic anhydride demonstrate narrow substrate scope and frequently produce low yields due to competing side reactions that reduce overall process efficiency. The most problematic limitation involves methods requiring direct handling of toxic carbon monoxide gas under high pressure, creating substantial safety hazards and necessitating specialized equipment that increases capital expenditure while limiting scalability. Furthermore, many conventional techniques demand pre-activated substrates or expensive reagents that drive up production costs and create supply chain vulnerabilities for manufacturers seeking consistent access to these critical building blocks.
The Novel Approach
The patented methodology overcomes these limitations through an elegant palladium-catalyzed carbonylative tandem reaction that operates under significantly milder conditions (90°C) while eliminating hazardous gas handling requirements entirely. By utilizing TFBen as a solid carbon monoxide surrogate that decomposes in situ to release CO during the reaction sequence, this process maintains equivalent reactivity without the associated safety risks or specialized infrastructure needs. The system demonstrates remarkable functional group tolerance across diverse substrates, accommodating various substituents on both the o-iodoaniline and trifluoroethylimide chloride components without requiring pre-modification or activation steps. This approach achieves consistently high yields across multiple derivative types while utilizing inexpensive starting materials that are readily available from standard chemical suppliers. The simplified workup procedure involving filtration followed by column chromatography purification delivers products meeting pharmaceutical quality standards without complex additional processing steps that would otherwise increase production timelines and costs.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The reaction mechanism proceeds through a sophisticated sequence initiated by potassium tert-butoxide-promoted intermolecular carbon-nitrogen bond coupling between the substrates to form a trifluoroacetamidine intermediate. This key intermediate then undergoes oxidative addition where the palladium catalyst inserts into the carbon-iodine bond of o-iodoaniline to generate a divalent palladium species. Under thermal activation at 90°C, TFBen decomposes to release carbon monoxide which subsequently inserts into the carbon-palladium bond to form an acyl palladium complex. The base then facilitates deprotonation and cyclization through nitrogen coordination to create a seven-membered ring palladium intermediate that ultimately undergoes reductive elimination to yield the final quinazolinone product with the characteristic trifluoromethyl substitution at the 2-position. This mechanistic pathway demonstrates exceptional atom economy while avoiding common side reactions that typically plague traditional methods.
Impurity control is achieved through multiple inherent features of this catalytic system that prevent common degradation pathways observed in conventional syntheses. The mild reaction temperature (90°C) minimizes thermal decomposition pathways that typically generate unwanted byproducts in higher temperature processes. The precise stoichiometric control of the catalytic components (Pd(PPh3)2Cl2/dppp at 5 mol% each) prevents catalyst aggregation that could lead to heterogeneous reaction conditions and inconsistent product quality. The use of anhydrous THF as solvent eliminates hydrolysis pathways that might otherwise affect sensitive functional groups on the substrates. Furthermore, the absence of transition metal residues in the final product—achieved through optimized purification protocols—ensures compliance with stringent pharmaceutical quality standards regarding metal contamination limits.
How to Synthesize 2-Trifluoromethyl Quinazolinone Derivatives Efficiently
This patented methodology represents a significant advancement in the synthesis of complex heterocyclic pharmaceutical intermediates through its innovative approach to carbonylation chemistry. The process eliminates traditional safety hazards while maintaining high efficiency across diverse substrate combinations, making it particularly valuable for manufacturers requiring reliable access to these critical building blocks. Detailed standardized synthesis procedures have been developed based on extensive optimization studies documented in patent CN112125856A, ensuring consistent product quality and yield across different production scales. The following step-by-step guide provides essential operational parameters for successful implementation in industrial settings, with specific attention to critical control points that ensure optimal results.
- Prepare reaction mixture by combining palladium catalyst (Pd(PPh3)2Cl2), dppp ligand, and potassium tert-butoxide in anhydrous THF under inert atmosphere with precise stoichiometric ratios as specified in patent CN112125856A.
- Add o-iodoaniline and trifluoroethylimide chloride substrates followed by TFBen solid CO surrogate to the catalytic system, maintaining strict temperature control at 90°C throughout the reaction period.
- Execute post-reaction processing through filtration, silica gel mixing, and column chromatography purification to isolate high-purity quinazolinone derivatives meeting stringent pharmaceutical quality specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process directly addresses critical pain points faced by procurement and supply chain professionals in the pharmaceutical industry through its inherent design features that enhance operational reliability and economic efficiency. The elimination of hazardous carbon monoxide gas handling removes significant regulatory compliance burdens while reducing facility requirements, creating immediate value through simplified operational workflows. By utilizing readily available starting materials from multiple global suppliers, the process mitigates single-source dependency risks that often disrupt traditional supply chains for specialty chemicals. The robust nature of the reaction system ensures consistent product quality across different production batches, reducing quality-related delays that can impact downstream manufacturing schedules for time-sensitive drug development programs.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal removal steps and specialized high-pressure equipment required for traditional CO-based processes creates substantial cost savings through reduced capital expenditure and simplified facility requirements. The use of commercially available starting materials at favorable price points combined with high-yielding reactions minimizes raw material costs while avoiding expensive purification steps typically needed to remove metal contaminants from final products.
- Enhanced Supply Chain Reliability: The broad substrate scope allows flexible sourcing strategies where multiple suppliers can provide equivalent starting materials without requiring process revalidation, significantly reducing supply chain vulnerability. The simplified reaction setup using standard laboratory equipment enables rapid scale-up from development to production without specialized infrastructure investments, ensuring consistent delivery timelines even during periods of market volatility or supply constraints.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to commercial production volumes while maintaining consistent product quality parameters essential for pharmaceutical applications. The elimination of toxic gas handling reduces environmental compliance requirements and safety protocols, lowering operational complexity while meeting increasingly stringent regulatory standards for green chemistry practices in pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns raised by industry professionals regarding the implementation of this patented manufacturing process for quinazolinone derivatives. These answers are derived directly from experimental data and technical specifications documented in patent CN112125856A, providing evidence-based insights to support informed decision-making for procurement teams evaluating this technology for their manufacturing needs.
Q: How does this method eliminate the need for toxic carbon monoxide gas in quinazolinone synthesis?
A: The patented process utilizes 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide surrogate that thermally decomposes under reaction conditions to release CO in situ, completely avoiding handling of toxic gaseous CO while maintaining equivalent reactivity in the carbonylation step.
Q: What substrate scope limitations exist for this palladium-catalyzed synthesis?
A: The method demonstrates broad substrate applicability with R¹ groups including H, C₁-C₅ alkyl, halogens, and trifluoromethyl; R² encompasses substituted or unsubstituted aryl groups with positions at ortho, meta, or para, enabling diverse derivative synthesis without pre-activation requirements.
Q: How does this process ensure high purity for pharmaceutical intermediate applications?
A: The elimination of transition metal catalysts in final products through optimized purification protocols combined with the absence of toxic gas handling prevents metal contamination and byproduct formation, yielding derivatives with purity levels suitable for API manufacturing as demonstrated in patent examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Derivatives Supplier
Our company leverages this patented technology to deliver exceptional value through our comprehensive CDMO capabilities specifically designed for complex heterocyclic compound manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities. Our state-of-the-art facilities feature rigorous QC labs equipped with advanced analytical instrumentation to ensure every batch meets exacting pharmaceutical quality standards through comprehensive testing protocols that exceed industry requirements.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific manufacturing needs by contacting our technical procurement team directly. They will provide detailed information including specific COA data and route feasibility assessments to demonstrate how our patented process can enhance your supply chain reliability while optimizing production economics for your quinazolinone-based pharmaceutical intermediates.
