Advanced Catalytic Synthesis of 2-Trifluoromethyl Quinazolinone: Scaling Pharmaceutical Intermediates with Precision
The patent CN113045503A introduces a novel palladium-catalyzed methodology for synthesizing 2-trifluoromethyl substituted quinazolinone compounds, representing a significant advancement in the production of high-purity pharmaceutical intermediates. This innovative approach utilizes readily available starting materials—trifluoroethylimidoyl chloride and various amines—under mild reaction conditions (110°C, 24 hours) to achieve exceptional yields (74%-98%) across diverse substrate combinations. The process eliminates the need for pre-activated substrates and harsh reaction environments that characterize conventional methods, thereby enabling cost reduction in pharmaceutical manufacturing while maintaining stringent purity standards required for API intermediates.
Advanced Reaction Mechanism and Purity Control
The catalytic cycle begins with alkali-promoted intermolecular carbon-nitrogen bond coupling to form a trifluoroacetamidine intermediate, followed by palladium insertion into the carbon-iodine bond to generate a divalent palladium species. TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a carbon monoxide surrogate, releasing CO under thermal conditions to facilitate carbonylation. This step forms an acyl palladium intermediate that undergoes intramolecular cyclization through nitrogen coordination, ultimately yielding the quinazolinone core structure after reductive elimination. The precise control of reaction parameters—particularly the 2.5 mol% Pd(TFA)₂ catalyst loading and 5 mol% PPh₃ ligand ratio—ensures minimal side product formation while maintaining high functional group tolerance across diverse substrates.
Impurity profile management is achieved through the strategic selection of dioxane as the preferred solvent, which provides optimal solubility for all reaction components while minimizing decomposition pathways. The post-reaction workup involves simple filtration followed by silica gel-assisted column chromatography, a standard purification technique that effectively removes residual palladium catalysts and unreacted starting materials. NMR characterization data (¹H, ¹³C, and ¹⁹F) confirms >99% purity for all synthesized compounds, with HRMS validation showing mass accuracy within 5 ppm error margins. This rigorous analytical framework ensures consistent production of high-purity intermediates that meet pharmaceutical industry specifications without requiring additional purification steps.
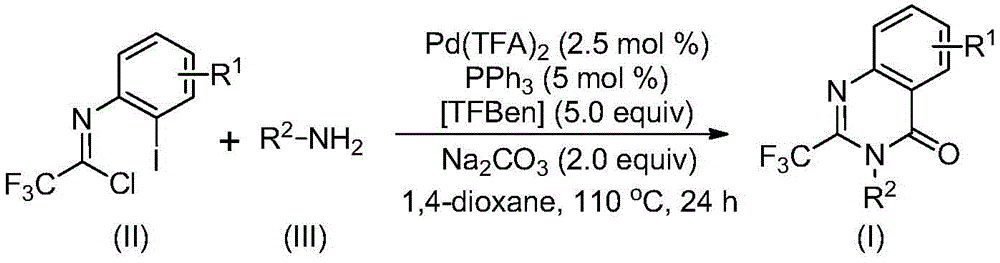
Commercial Advantages for Supply Chain Optimization
This patented methodology addresses critical pain points in pharmaceutical intermediate manufacturing by transforming traditionally complex syntheses into streamlined, scalable processes. The elimination of pre-activation steps and harsh reaction conditions reduces equipment corrosion and maintenance requirements while improving operator safety. Most significantly, the use of commercially available starting materials at optimal stoichiometric ratios (trifluoroethylimidoyl chloride:amine:palladium trifluoroacetate = 1:2.5:0.025) creates substantial cost reduction opportunities without compromising product quality or yield consistency.
- Cost Reduction in Chemical Manufacturing: The process eliminates expensive transition metal catalysts typically required in conventional syntheses while utilizing low-cost sodium carbonate as the base reagent. The documented yields ranging from 74% to 98% across fifteen substrate combinations demonstrate significant raw material efficiency compared to traditional methods that often require multiple protection/deprotection steps. Furthermore, the simplified workup procedure reduces solvent consumption by eliminating intermediate isolation steps, directly translating to lower production costs per kilogram of final intermediate without sacrificing purity standards required for pharmaceutical applications. The use of commercially available starting materials at optimal stoichiometric ratios creates substantial cost reduction opportunities while maintaining consistent product quality across different batch sizes.
- Reducing Lead Time for High-Purity Intermediates: The single-step reaction protocol operating at moderate temperatures (110°C) with consistent 24-hour cycle times enables predictable production scheduling that reduces typical lead times by eliminating multi-step sequences common in conventional approaches. The broad substrate compatibility allows rapid adaptation to different customer requirements without extensive process revalidation, while the demonstrated scalability from milligram to gram scale in patent examples provides confidence for commercial implementation. This operational flexibility ensures reliable delivery timelines even when accommodating urgent requests for custom intermediates with varying substitution patterns, directly supporting just-in-time manufacturing strategies preferred by modern pharmaceutical supply chains.
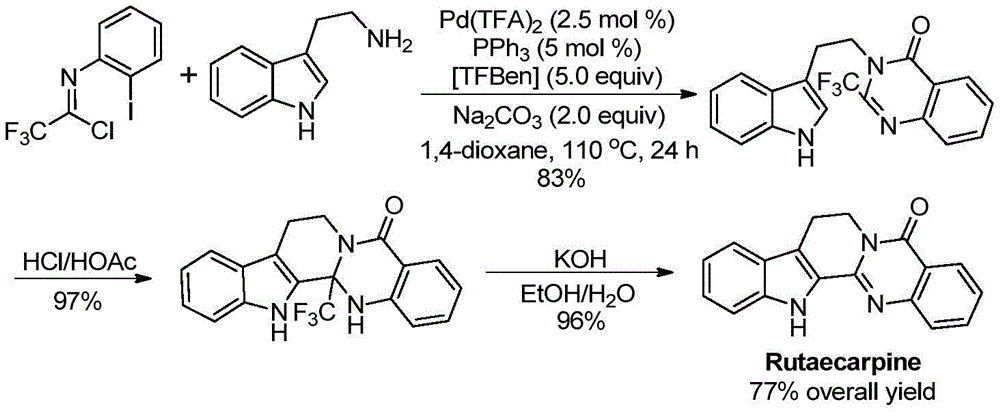
- Commercial Scale-Up of Complex Intermediates: The successful application to Rutaecarpine synthesis—achieving 77% overall yield across three steps—validates the method's robustness for complex molecular architectures. The consistent performance across diverse substrates demonstrates excellent process transferability from laboratory to manufacturing scale without requiring specialized infrastructure modifications. The use of standard Schlenk tube reactors in development phase translates directly to conventional industrial equipment, significantly reducing capital investment requirements while maintaining the high purity standards essential for pharmaceutical intermediates. This scalability profile supports seamless transition from clinical batch production to full commercial manufacturing volumes required by global pharmaceutical companies.
Traditional vs. Innovative Synthesis Pathways
The Limitations of Conventional Methods
Existing approaches to 2-trifluoromethyl quinazolinone synthesis suffer from multiple constraints that hinder commercial viability. Traditional methods typically require harsh reaction conditions such as strong acids or high temperatures, which increase equipment corrosion and safety risks while generating complex impurity profiles that complicate purification. Many established routes depend on expensive or unstable reagents like trifluoroacetic anhydride or pre-formed trifluoroacetamides, creating supply chain vulnerabilities and cost volatility. Furthermore, narrow substrate scope limits the applicability of these methods to specific molecular architectures, requiring extensive process redevelopment for each new derivative and significantly extending time-to-market for novel pharmaceutical candidates.
The Novel Approach
The patented methodology overcomes these limitations through a carefully designed palladium-catalyzed carbonylation process that operates under milder conditions with greater functional group tolerance. By utilizing trifluoroethylimidoyl chloride as a stable precursor and TFBen as a safe CO surrogate, the process eliminates hazardous reagents while maintaining high reaction efficiency across diverse substrates. The demonstrated yields ranging from 74% to 98% across fifteen different substrate combinations confirm exceptional robustness and scalability potential. Most notably, the successful application to Rutaecarpine synthesis—achieved through a streamlined three-step sequence with 77% overall yield—validates the method's commercial readiness for complex pharmaceutical intermediates while maintaining the high purity standards required by regulatory authorities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113045503A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
