Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN112480015B discloses a groundbreaking multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, a class of compounds renowned for their potent antifungal, antiviral, and anticancer properties. This innovation addresses the longstanding challenges associated with introducing the trifluoromethyl group, which is known to enhance metabolic stability and lipophilicity in drug candidates. By leveraging a palladium-catalyzed carbonylation cascade, this technology enables the direct transformation of inexpensive nitro compounds and trifluoroethylimidoyl chlorides into high-value intermediates. For R&D directors and procurement specialists, this represents a significant opportunity to streamline the supply chain for complex nitrogen-containing heterocycles while maintaining stringent purity standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the quinazolinone core often suffer from severe operational and economic drawbacks that hinder large-scale manufacturing. Conventional methods frequently rely on harsh reaction conditions, such as high-pressure carbon monoxide atmospheres, which necessitate specialized autoclaves and pose significant safety risks in a production environment. Furthermore, many existing protocols require expensive transition metal catalysts like ruthenium or platinum, or demand pre-activated substrates such as acid anhydrides and benzamides that increase the step count and overall cost of goods. These legacy processes are often plagued by low yields and narrow substrate compatibility, limiting the ability of medicinal chemists to explore diverse chemical space efficiently. The reliance on gaseous CO also introduces logistical complexities regarding storage and handling, making these routes less attractive for reliable pharmaceutical intermediate suppliers aiming for green and sustainable chemistry practices.
The Novel Approach
In stark contrast, the methodology described in CN112480015B offers a streamlined, one-pot solution that drastically simplifies the synthetic workflow. By utilizing solid molybdenum hexacarbonyl as a safe carbon monoxide surrogate, the process eliminates the need for high-pressure gas equipment, thereby enhancing operational safety and reducing capital expenditure. The reaction proceeds under relatively mild thermal conditions at 120°C in common organic solvents like 1,4-dioxane, demonstrating exceptional efficiency with reaction times ranging from 16 to 30 hours. This approach not only improves atom economy but also exhibits remarkable functional group tolerance, allowing for the incorporation of various halogens, alkyl groups, and electron-withdrawing substituents without compromising yield. The ability to synthesize these complex structures from cheap and readily available nitro compounds positions this technology as a superior choice for cost reduction in API manufacturing.
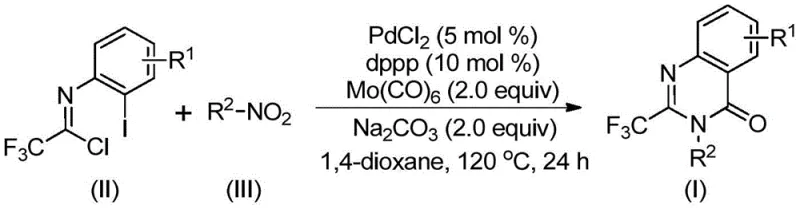
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The success of this synthesis lies in its elegant mechanistic pathway, which orchestrates multiple bond-forming events in a single vessel. The reaction initiates with the reduction of the nitro compound to an amine species by molybdenum hexacarbonyl, followed immediately by a base-promoted intermolecular coupling with trifluoroethylimidoyl chloride to form a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the imidoyl chloride moiety, generating a reactive divalent palladium species. As the temperature rises, the molybdenum complex releases carbon monoxide in situ, which then inserts into the carbon-palladium bond to form an acyl-palladium intermediate. This sequence culminates in the formation of a seven-membered cyclic palladium intermediate via nitrogen coordination, followed by reductive elimination to release the final 2-trifluoromethyl substituted quinazolinone product. Understanding this cascade is crucial for optimizing reaction parameters and ensuring consistent quality in commercial batches.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise approaches. The one-pot nature minimizes the isolation of unstable intermediates, thereby reducing the formation of degradation byproducts that often complicate purification. The chemoselectivity of the palladium system ensures that sensitive functional groups on the aromatic rings, such as fluorine or chlorine atoms, remain intact throughout the process. This high level of selectivity is vital for producing high-purity OLED material precursors or pharmaceutical intermediates where trace impurities can affect biological activity. The robustness of the catalytic cycle allows for the synthesis of diverse derivatives, as evidenced by the successful preparation of compounds with varying steric and electronic properties, confirming the versatility of this platform for generating complex molecular libraries.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
Implementing this synthesis requires careful attention to reagent stoichiometry and reaction conditions to maximize yield and purity. The protocol involves combining palladium chloride, the dppp ligand, sodium carbonate, and molybdenum hexacarbonyl with the specific trifluoroethylimidoyl chloride and nitro compound substrates in an anhydrous organic solvent. The mixture is then heated to facilitate the cascade reaction, after which standard workup procedures involving filtration and silica gel treatment are employed. For detailed operational parameters and specific molar ratios optimized for different substrates, please refer to the standardized synthesis guide below which outlines the precise execution of this patented technology.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow the carbonylation cascade and cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond simple chemistry. The shift from hazardous high-pressure gases to solid reagents significantly de-risks the manufacturing process, ensuring greater supply continuity and reducing the need for specialized safety infrastructure. Moreover, the use of commodity chemicals like nitro compounds as starting materials insulates the production cost from the volatility associated with exotic or custom-synthesized precursors. This stability in raw material sourcing is critical for maintaining long-term contracts and meeting the rigorous delivery schedules demanded by global pharmaceutical clients. The simplified downstream processing further enhances the economic viability of the project by reducing solvent consumption and waste generation.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the use of inexpensive nitro compounds lead to a substantial decrease in capital and operational expenditures. By avoiding the need for pre-activation steps and expensive noble metal catalysts like ruthenium, the overall cost of goods is significantly optimized. The high reaction efficiency and yield reported in the patent data suggest that less raw material is wasted, contributing to a leaner and more profitable manufacturing model. Additionally, the simplified purification process reduces the consumption of chromatography media and solvents, further driving down the variable costs associated with large-scale production runs.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved because the key starting materials, such as nitrobenzenes and trifluoroethylimidoyl chlorides, are widely available from multiple global vendors. This diversity in the supply base mitigates the risk of single-source bottlenecks that can disrupt production timelines. The robustness of the reaction conditions means that the process is less susceptible to minor fluctuations in environmental parameters, ensuring consistent batch-to-batch quality. For supply chain planners, this predictability allows for more accurate forecasting and inventory management, ultimately leading to shorter lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The transition from laboratory scale to industrial production is facilitated by the absence of gaseous carbon monoxide, which simplifies reactor design and safety protocols. The process generates fewer hazardous byproducts compared to traditional methods, aligning with increasingly strict environmental regulations and corporate sustainability goals. The ability to scale this reaction to gram levels and beyond without loss of efficiency demonstrates its readiness for commercial deployment. This scalability ensures that manufacturers can rapidly respond to market demand surges without compromising on environmental compliance or worker safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical aspects of adopting this technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using nitro compounds over traditional amines in this synthesis?
A: Nitro compounds are significantly cheaper and more readily available than pre-activated amines or acid anhydrides. This method utilizes them directly in a one-pot cascade, eliminating the need for separate reduction steps and reducing overall raw material costs.
Q: Does this process require high-pressure carbon monoxide gas?
A: No, the process avoids the safety hazards and specialized equipment associated with high-pressure CO gas. Instead, it uses solid molybdenum hexacarbonyl (Mo(CO)6) as a safe and convenient carbon monoxide substitute that releases CO in situ under heating.
Q: What is the substrate scope for the R2 group in the quinazolinone structure?
A: The method demonstrates excellent functional group tolerance. The R2 group can be varied widely to include C1-C10 alkyl chains, cycloalkyl groups like cyclohexyl, and various substituted or unsubstituted aryl groups, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed carbonylation technology for the next generation of therapeutic agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including the safe handling of molybdenum reagents and the rigorous QC labs necessary to verify stringent purity specifications. We are committed to delivering high-quality intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthetic route for your drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this method can optimize your budget without sacrificing quality. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you accelerate your timeline to market with a reliable and efficient supply of 2-trifluoromethyl substituted quinazolinones.
