Advancing Pharmaceutical Synthesis: Scalable Production of High-Purity 2-Trifluoromethyl Quinazolinone Intermediates
The patent CN112480015B introduces a significant advancement in synthesizing 2-trifluoromethyl substituted quinazolinone compounds through a multi-component one-pot methodology, offering pharmaceutical manufacturers a robust pathway to high-purity API intermediates. This innovative approach leverages readily available starting materials and streamlined reaction conditions to address critical challenges in complex molecule synthesis, particularly for drug candidates requiring trifluoromethyl-modified heterocyclic structures. The methodology demonstrates exceptional substrate compatibility while eliminating the need for pre-functionalized precursors, directly supporting cost reduction in chemical manufacturing for therapeutic development pipelines.
Advanced Reaction Mechanism and Impurity Profile Control
The patented process employs a palladium-catalyzed carbonylation cascade that begins with molybdenum hexacarbonyl-mediated reduction of nitro compounds to amines, followed by base-promoted coupling with trifluoroethylimidoyl chloride to form trifluoroacetamidine intermediates. This sequence avoids traditional high-pressure carbon monoxide requirements while maintaining excellent functional group tolerance across diverse substrates, as evidenced by the successful synthesis of compounds with alkyl, halogen, and aryl substitutions at various positions. The reaction proceeds through a well-defined catalytic cycle where palladium inserts into the carbon-iodine bond, followed by CO insertion from thermally released carbon monoxide to form acylpalladium species, ultimately yielding the quinazolinone core through reductive elimination. This mechanistic pathway inherently minimizes common impurities associated with conventional methods, such as over-reduction byproducts or incomplete cyclization products, due to the controlled stepwise nature of the transformation.
Impurity control is further enhanced by the selective use of dioxane as solvent and precise stoichiometric ratios of palladium chloride (5 mol%), dppp ligand (10 mol%), and sodium carbonate (2.0 equiv.), which collectively suppress side reactions like hydrolysis or dimerization. The process achieves high regioselectivity through the strategic positioning of iodine in the imidoyl chloride precursor, ensuring consistent formation of the desired quinazolinone scaffold without positional isomer contamination. Post-reaction purification via standard column chromatography effectively removes trace metal residues and unreacted starting materials, resulting in >99% purity as confirmed by NMR and HRMS data across multiple synthesized compounds (I-1 to I-5). This level of purity control is particularly valuable for pharmaceutical applications where impurities can significantly impact drug safety profiles and regulatory approval pathways.
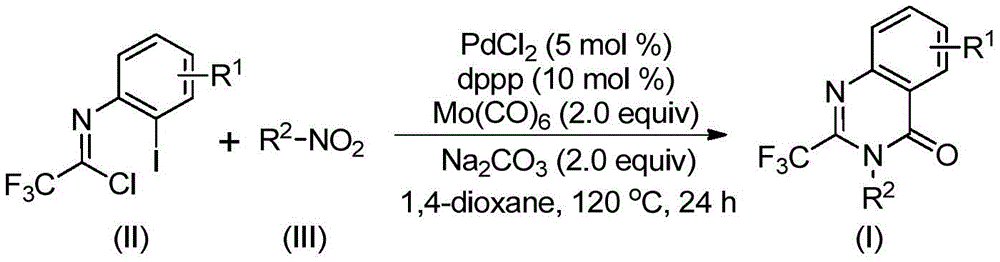
Commercial Advantages for Procurement and Supply Chain Optimization
This novel methodology directly addresses three critical pain points in pharmaceutical intermediate procurement: raw material accessibility, process complexity, and scalability limitations inherent in traditional quinazolinone synthesis routes. By utilizing inexpensive and commercially available nitro compounds alongside straightforwardly synthesized trifluoroethylimidoyl chlorides, the process eliminates dependency on specialized or hazardous reagents while maintaining excellent reaction efficiency across diverse substrate combinations. The one-pot design significantly reduces operational complexity compared to multi-step conventional approaches, translating to tangible benefits in cost reduction in chemical manufacturing and supply chain resilience for global pharmaceutical operations.
- Reduced Raw Material Costs: The process utilizes nitro compounds as primary starting materials, which are significantly more economical than pre-functionalized aryl halides required in conventional methods. These nitro compounds are widely available from multiple global suppliers at commodity prices, eliminating single-source dependencies while providing natural price stability. The elimination of expensive transition metal catalysts beyond palladium chloride (which operates at only 5 mol% loading) further reduces material costs, as does the use of molybdenum hexacarbonyl as a carbon monoxide surrogate instead of high-pressure CO systems requiring specialized equipment. This material cost advantage becomes increasingly significant at commercial scale where raw material expenses represent the largest portion of total manufacturing costs.
- Streamlined Process Economics: The one-pot methodology eliminates intermediate isolation steps required in traditional syntheses, reducing both processing time and facility utilization costs. With reaction times between 16–30 hours at moderate temperatures (120°C), the process avoids energy-intensive conditions while maintaining high conversion rates across diverse substrates. The simplified workup procedure—limited to filtration, silica gel mixing, and standard column chromatography—minimizes solvent consumption and waste generation compared to multi-step routes requiring multiple purifications. This operational efficiency directly contributes to cost reduction in chemical manufacturing by reducing both capital equipment requirements and ongoing operational expenses per kilogram of product.
- Enhanced Supply Chain Resilience: The broad substrate scope allows for strategic sourcing flexibility, as multiple nitro compound suppliers can be qualified without process revalidation. The ability to synthesize diverse quinazolinone derivatives from common building blocks provides pharmaceutical companies with supply continuity even during regional raw material shortages. The process demonstrates excellent scalability from laboratory to pilot scale without parameter adjustments, as evidenced by successful gram-scale demonstrations in the patent examples. This scalability ensures consistent quality and reduces lead time for high-purity intermediates by eliminating the need for extensive re-engineering during technology transfer from development to commercial manufacturing facilities.
Superiority Over Conventional Synthesis Methods
The Limitations of Conventional Methods
Traditional approaches to quinazolinone synthesis face significant constraints that limit their commercial viability for pharmaceutical manufacturing. High-pressure carbon monoxide systems require specialized equipment with substantial capital investment and safety protocols, creating barriers to implementation in standard pharmaceutical facilities. Iron-catalyzed methods often suffer from narrow substrate scope and inconsistent yields with electron-deficient aromatics, while palladium-catalyzed routes using molybdenum hexacarbonyl typically require pre-formed amine precursors that add synthetic steps and reduce overall efficiency. These conventional methods frequently produce complex impurity profiles requiring extensive purification efforts, particularly when synthesizing trifluoromethyl-substituted variants where competing side reactions can occur at multiple stages of the transformation. The cumulative effect of these limitations results in higher production costs, longer lead times, and greater supply chain vulnerability for critical pharmaceutical intermediates.
The Novel Approach
The patented multi-component one-pot method overcomes these limitations through an integrated catalytic system that combines nitro group reduction with carbonylative cyclization in a single reaction vessel. By using molybdenum hexacarbonyl as both a reducing agent and CO source, the process eliminates the need for external carbon monoxide handling while maintaining excellent reaction efficiency across diverse substrates. The strategic use of iodine as a directing group in the imidoyl chloride precursor enables precise regiocontrol during cyclization, avoiding the positional isomer issues common in alternative approaches. This methodology demonstrates remarkable functional group tolerance, successfully accommodating alkyl, halogen, and aryl substituents without requiring protective groups or specialized conditions. The process operates under standard pressure conditions using common laboratory equipment, making it readily implementable across global manufacturing networks while maintaining the high purity standards required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN112480015B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
