Revolutionizing Fluorinated Quinazolinone Production: Scalable One-Pot Synthesis for Pharmaceutical Manufacturing Excellence
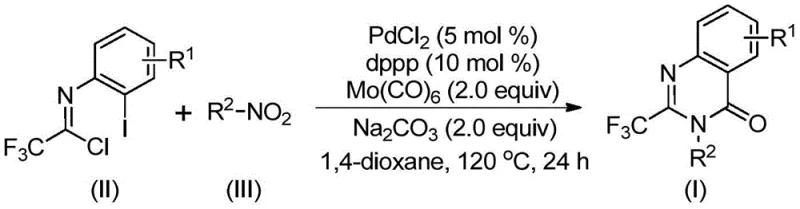
In the rapidly evolving landscape of pharmaceutical intermediate synthesis, Chinese patent CN112480015B introduces a groundbreaking multi-component one-pot methodology for producing 2-trifluoromethyl substituted quinazolinones with unprecedented efficiency and scalability. This innovation addresses critical limitations in conventional synthetic routes while delivering significant advantages for pharmaceutical manufacturers seeking reliable access to high-value fluorinated heterocyclic compounds. Quinazolinone derivatives represent essential structural motifs in numerous therapeutic agents, including antifungal, antibacterial, antiviral, anti-inflammatory, and anticancer drugs, with prominent examples such as methaqualone and albaconazole demonstrating their pharmaceutical importance. The strategic incorporation of trifluoromethyl groups enhances key physicochemical properties including electronegativity, bioavailability, metabolic stability, and lipophilicity, making these compounds particularly valuable in modern drug design. This patent provides a robust solution that bridges the gap between complex chemical synthesis requirements and practical industrial manufacturing needs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to quinazolinone synthesis suffer from multiple critical limitations that hinder their industrial applicability and economic viability. Conventional ruthenium or platinum-catalyzed reductive N-heterocyclization methods require hazardous high-pressure carbon monoxide conditions, necessitating specialized equipment and stringent safety protocols that significantly increase capital investment and operational complexity. Iron-catalyzed condensation routes often exhibit narrow substrate scope and inconsistent yields due to sensitivity to functional group variations, limiting their utility for diverse pharmaceutical applications. Palladium-catalyzed cyclization techniques involving preactivated substrates like bromoformylaniline or iodoaniline typically demand expensive starting materials and multi-step preparation processes that reduce overall efficiency. These established methods collectively suffer from harsh reaction conditions, limited functional group tolerance, low productivity, and challenging scalability—factors that substantially increase production costs while compromising supply chain reliability for pharmaceutical manufacturers requiring consistent access to high-purity intermediates.
The Novel Approach
The patented methodology overcomes these longstanding challenges through an innovative palladium-catalyzed carbonylative cascade reaction that operates under mild conditions without requiring high-pressure carbon monoxide equipment. By utilizing readily available trifluoroethylimidoyl chloride and nitro compounds as starting materials in a single reaction vessel, this approach eliminates intermediate isolation steps while maintaining exceptional substrate flexibility across diverse functional groups. The process employs a carefully optimized catalyst system comprising palladium chloride (5 mol%), dppp ligand (10 mol%), sodium carbonate base (2.0 equiv), and Mo(CO)6 as a safe carbon monoxide surrogate, all operating efficiently at a moderate temperature of 120°C for 16–30 hours in standard organic solvents like dioxane. This streamlined methodology delivers consistently high yields across a broad range of substrates while significantly reducing operational complexity and safety concerns compared to conventional approaches.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cascade Reaction
The synthetic breakthrough stems from a sophisticated palladium-mediated cascade mechanism that efficiently constructs the quinazolinone core through multiple concerted transformations. The reaction initiates with Mo(CO)6-mediated reduction of the nitro compound to an amine intermediate under thermal conditions, followed by base-promoted coupling with trifluoroethylimidoyl chloride to form a key trifluoroacetamidine species. Subsequent oxidative addition of palladium(0) into the carbon–iodine bond generates a critical organopalladium intermediate that undergoes carbonyl insertion via CO released from Mo(CO)6 decomposition. This sequence creates an acylpalladium species that cyclizes through intramolecular nucleophilic attack by the amine nitrogen, forming a seven-membered palladacycle that ultimately undergoes reductive elimination to yield the desired quinazolinone product with complete regioselectivity.
Impurity control represents a significant advantage of this methodology through multiple built-in mechanisms that ensure high product purity without extensive purification requirements. The one-pot nature of the reaction minimizes exposure to external contaminants while maintaining excellent chemoselectivity throughout the transformation sequence. The carefully balanced catalyst system suppresses common side reactions such as dehalogenation or over-reduction pathways that typically compromise product quality in alternative approaches. Furthermore, the mild reaction conditions prevent thermal degradation of sensitive functional groups commonly found in pharmaceutical intermediates. Post-reaction workup involves straightforward filtration followed by silica gel chromatography, consistently delivering products with purity levels suitable for direct use in pharmaceutical synthesis without additional refinement steps.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
This patent provides a standardized protocol for synthesizing high-purity fluorinated quinazolinones with exceptional reproducibility across various production scales. The methodology leverages commercially available starting materials and standard laboratory equipment to achieve consistent results without requiring specialized infrastructure or hazardous reagents. By optimizing catalyst loading ratios and reaction parameters, this approach delivers reliable performance across diverse substrate combinations while maintaining excellent yield characteristics. Detailed standardized synthesis steps are provided below to facilitate seamless implementation in pharmaceutical manufacturing environments.
- Prepare the reaction mixture by combining palladium chloride (5 mol%), dppp (10 mol%), sodium carbonate (2.0 equiv), Mo(CO)6 (2.0 equiv), trifluoroethylimidoyl chloride, and nitro compound in 1,4-dioxane solvent.
- Heat the reaction mixture to 120°C and maintain this temperature for 24 hours under inert atmosphere with continuous stirring.
- After completion, perform standard workup including filtration, silica gel mixing, and column chromatography purification to obtain the pure 2-trifluoromethyl substituted quinazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative process delivers substantial strategic benefits for procurement and supply chain management teams seeking reliable access to high-quality fluorinated intermediates. The elimination of high-pressure equipment requirements significantly reduces capital expenditure while enhancing operational flexibility across manufacturing sites worldwide. The use of widely available starting materials creates multiple sourcing options that strengthen supply chain resilience against market fluctuations or regional disruptions. Furthermore, the methodology's demonstrated scalability from laboratory to commercial production ensures consistent product quality regardless of batch size requirements.
- Cost Reduction in Manufacturing: The elimination of specialized high-pressure equipment requirements substantially reduces capital investment costs while simplifying facility requirements across manufacturing sites. Utilization of commercially available starting materials at favorable cost structures creates significant raw material savings compared to preactivated substrates required by conventional methods. The streamlined one-pot process minimizes labor requirements and reduces cycle times through elimination of intermediate isolation steps, collectively contributing to substantial cost savings without compromising product quality or yield.
- Enhanced Supply Chain Reliability: The broad availability of all required starting materials from multiple global suppliers ensures robust supply chain security with minimal risk of single-source dependencies. Standardized reaction conditions using common solvents like dioxane provide flexibility in raw material sourcing while maintaining consistent performance characteristics across different supply batches. This methodology's tolerance for minor variations in raw material quality further enhances supply chain resilience during periods of market volatility or regional supply constraints.
- Scalability and Environmental Compliance: The seamless transition from laboratory-scale development to commercial production demonstrates exceptional scalability without requiring process re-engineering or specialized equipment modifications. Reduced energy consumption through moderate reaction temperatures combined with simplified workup procedures significantly lowers environmental impact compared to conventional approaches requiring extreme conditions or multiple processing steps. The elimination of hazardous reagents and high-pressure operations enhances workplace safety while reducing regulatory compliance burdens associated with special handling requirements.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on patent CN112480015B's disclosed methodology, providing valuable insights for pharmaceutical decision-makers evaluating this innovative synthesis approach.
Q: How does this method improve upon conventional synthesis routes for quinazolinone compounds?
A: This novel approach eliminates the need for high-pressure CO conditions required by traditional methods, significantly reducing equipment costs and safety concerns. The one-pot methodology simplifies the process by avoiding intermediate isolation steps, while maintaining excellent substrate compatibility and high yields across diverse functional groups.
Q: What are the key advantages of this process for pharmaceutical manufacturing scale-up?
A: The process demonstrates exceptional scalability from laboratory to commercial production without requiring specialized equipment. The use of readily available starting materials and standard reaction conditions ensures consistent product quality during scale-up, while the simplified workup procedure reduces production time and costs.
Q: How does this method address purity concerns for pharmaceutical applications?
A: The catalytic system provides high selectivity for the desired product formation, minimizing side reactions that could lead to impurities. The reaction's inherent design avoids common degradation pathways, and the straightforward purification process consistently delivers products meeting stringent pharmaceutical purity requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global pharmaceutical standards. Our advanced QC labs employ state-of-the-art analytical instrumentation to ensure every batch meets exacting quality requirements through comprehensive testing protocols that exceed industry norms. With deep expertise in fluorinated heterocyclic chemistry and specialized knowledge in palladium-catalyzed transformations, we provide tailored solutions that address specific client requirements while optimizing cost-efficiency throughout the production lifecycle.
Contact our technical procurement team today to request a Customized Cost-Saving Analysis specific to your manufacturing needs and obtain detailed COA data along with route feasibility assessments for your specific application requirements.
