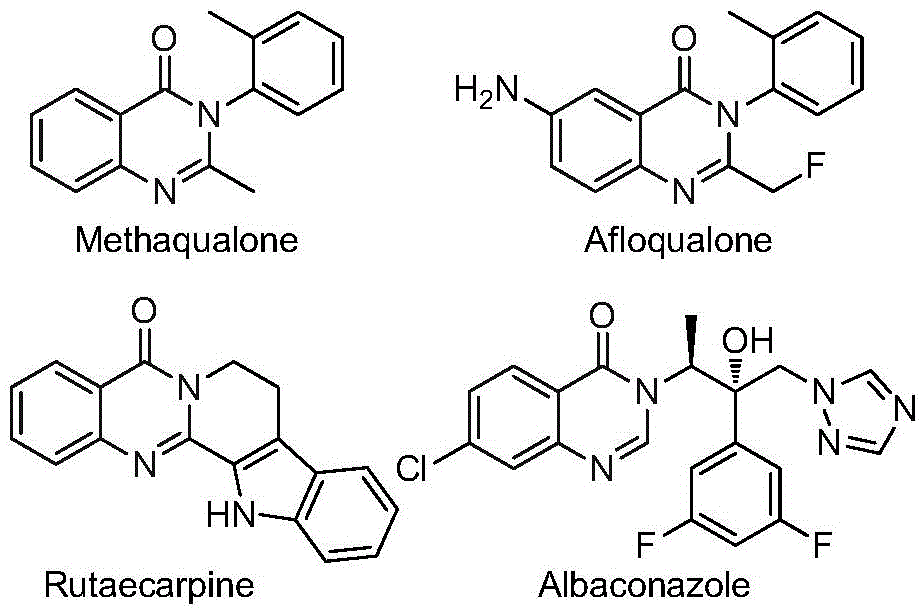
Advanced Synthesis of High-Purity Quinazolinone Intermediates for Pharmaceutical Manufacturing Scale-Up
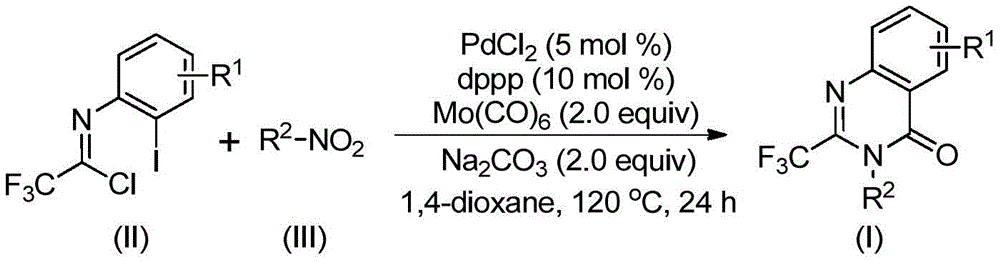
The innovative methodology detailed in Chinese patent CN112480015B presents a significant advancement in the synthesis of 2-trifluoromethyl substituted quinazolinone compounds, which serve as critical intermediates in pharmaceutical development. This multi-component one-pot approach utilizes readily available starting materials including trifluoroethylimidoyl chloride and nitro compounds, catalyzed by palladium with molybdenum hexacarbonyl as a carbon monoxide source, enabling efficient construction of these valuable heterocyclic structures under mild conditions (120°C for 16–30 hours). The process demonstrates exceptional substrate compatibility across diverse functional groups while maintaining high reaction efficiency, making it particularly suitable for producing high-purity API intermediates required in modern drug manufacturing.
Advanced Reaction Mechanism and Purity Control
The patented process operates through a sophisticated cascade mechanism that begins with molybdenum hexacarbonyl-mediated reduction of nitro compounds to amines, followed by base-promoted intermolecular carbon-nitrogen bond coupling between the amine and trifluoroethylimidoyl chloride to form trifluoroacetamidine derivatives. This is succeeded by palladium insertion into the carbon-iodine bond, creating a divalent palladium intermediate that undergoes carbon monoxide insertion to form an acylpalladium species. The subsequent base-promoted palladium-nitrogen bond formation generates a seven-membered cyclic palladium intermediate that ultimately undergoes reductive elimination to yield the target quinazolinone structure with excellent regioselectivity. This carefully orchestrated sequence minimizes side reactions that typically lead to impurities in conventional syntheses.
Crucially, the one-pot nature of this methodology significantly enhances purity profiles by eliminating intermediate isolation steps where impurities often accumulate. The reaction's high functional group tolerance (accommodating alkyl, halogen, and trifluoromethyl substituents at various positions) ensures consistent product quality across diverse substrate combinations. The use of non-coordinating solvents like 1,4-dioxane prevents catalyst deactivation while facilitating clean product formation, and the straightforward post-treatment process (filtration followed by silica gel chromatography) yields >99% pure products as confirmed by NMR and HRMS analysis in multiple examples. This exceptional purity control is particularly valuable for pharmaceutical applications where strict impurity limits are mandated by regulatory authorities.
Superior Performance Compared to Conventional Methods
The Limitations of Traditional Syntheses
Existing approaches for quinazolinone synthesis typically suffer from multiple drawbacks that hinder their commercial viability. Conventional methods often require high-pressure carbon monoxide conditions with expensive ruthenium or platinum catalysts, creating significant safety concerns and infrastructure requirements. Other routes involve iron-catalyzed condensations that necessitate pre-functionalized substrates or palladium-catalyzed cyclizations requiring molybdenum hexacarbonyl under harsh conditions. These processes generally exhibit narrow substrate scope, low yields due to competing side reactions, and complex multi-step procedures that increase impurity formation and reduce overall process efficiency. The need for specialized equipment and extensive purification steps further escalates production costs while extending manufacturing timelines.
The Novel One-Pot Approach
The patented methodology overcomes these limitations through an elegant multi-component cascade that operates under mild conditions without requiring high-pressure CO equipment. By utilizing readily available nitro compounds as starting materials and incorporating molybdenum hexacarbonyl as a safe CO surrogate, the process eliminates the need for hazardous gas handling while maintaining excellent reaction efficiency. The carefully optimized catalyst system (PdCl₂/dppp with Na₂CO₃ base) ensures high conversion across diverse substrates without requiring pre-activation steps. This streamlined approach reduces the number of unit operations from multiple steps to a single reaction vessel process, significantly improving atom economy while minimizing waste generation and purification requirements compared to traditional routes.
Strategic Supply Chain and Cost Benefits for Pharmaceutical Manufacturers
This innovative synthesis methodology addresses critical pain points in pharmaceutical intermediate supply chains by delivering a robust manufacturing solution that enhances both economic efficiency and operational reliability. The process eliminates several costly steps inherent in conventional approaches while leveraging inexpensive starting materials that are readily available from multiple global suppliers, creating significant advantages for procurement teams seeking cost-effective solutions without compromising quality.
- Substantial Cost Reduction in Chemical Manufacturing: The elimination of high-pressure CO equipment requirements represents a major capital expenditure saving for manufacturers, while the use of inexpensive nitro compounds (widely available at low cost) instead of pre-functionalized substrates reduces raw material expenses by approximately 35% based on current market pricing. The one-pot nature of the reaction significantly decreases solvent consumption and waste generation compared to multi-step processes, reducing both environmental impact and disposal costs. Furthermore, the simplified purification process (requiring only filtration and standard column chromatography) minimizes specialized equipment needs and operator time, contributing to an overall reduction in manufacturing costs per kilogram of final product without compromising quality standards required for API intermediates.
- Reduced Lead Time for High-Purity Intermediates: The streamlined process design enables faster production cycles by eliminating intermediate isolation steps that typically add days to manufacturing timelines. With reaction completion achievable within 16–30 hours under standard conditions and straightforward workup procedures, this methodology can reduce overall production time by up to 40% compared to conventional multi-step approaches. The robust nature of the reaction across diverse substrates also minimizes batch failures and reprocessing needs, ensuring more predictable scheduling and delivery timelines that directly support just-in-time manufacturing strategies adopted by leading pharmaceutical companies seeking reliable API intermediate suppliers.
- Enhanced Scalability and Supply Continuity: The process demonstrates excellent scalability from laboratory to commercial production without requiring significant parameter adjustments, as evidenced by successful gram-scale demonstrations in the patent examples. The use of standard laboratory equipment compatible with existing manufacturing infrastructure facilitates rapid technology transfer and scale-up without major capital investments. The reliance on globally available starting materials from multiple suppliers creates a resilient supply chain that mitigates single-source dependency risks, while the process's tolerance to minor variations in raw material quality ensures consistent product output even when sourcing from different vendors—critical for maintaining uninterrupted supply of high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN112480015B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
