Scaling High-Performance BAPO Photoinitiators: A Technical Breakthrough in UV Curing Technology
The landscape of UV-curable formulations is undergoing a significant transformation driven by the demand for higher performance photoinitiators with superior stability and reduced migration. Patent CN1324037C introduces a groundbreaking methodology for the synthesis of dimeric and multimeric forms of bisacylphosphine oxides (BAPO) and monoacylphosphine oxides (MAPO). Unlike traditional monomeric initiators, these novel polymeric structures offer enhanced thermal properties and compatibility within complex resin matrices. The invention details a robust chemical pathway utilizing dimetalated phosphines reacted with polycarboxylic acid halides, enabling the precise construction of symmetric and asymmetric architectures. This technical advancement addresses critical pain points in the coatings and adhesives industry, particularly regarding the volatility and odor associated with lower molecular weight initiators. By leveraging this proprietary synthesis route, manufacturers can access a new class of high-efficiency photoinitiators tailored for demanding industrial applications.
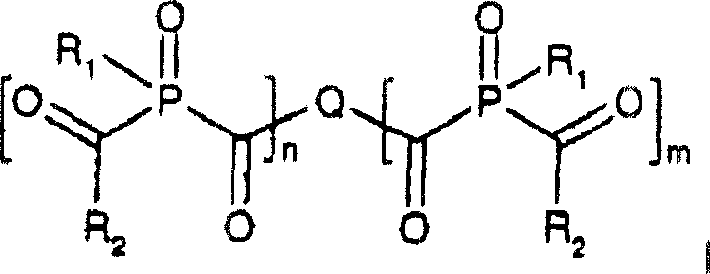
Historically, the synthesis of acylphosphine oxide dimers relied on limited methodologies that restricted structural diversity. For instance, European patent publication EP-A 0601413 described the reaction of arene-diacid chlorides with alkoxy-diphenyl-phosphines. A major limitation of this conventional approach was the inherent symmetry it imposed on the final product, forcing residues R5 and R6 to be identical, or requiring one to be strictly alkoxy. This lack of flexibility hindered the optimization of photophysical properties for specific curing profiles. Furthermore, while US Patent Publication 2001/0031898 broadly encompassed dimeric forms within its definitions, it failed to provide explicit disclosure or working examples for their preparation, leaving a significant gap in practical manufacturing knowledge. The reliance on specific alkoxy reactants in older methods also constrained the range of substituents available on the phosphorus atom, limiting the ability to fine-tune solubility and reactivity.
In stark contrast, the novel approach disclosed in CN1324037C utilizes a versatile dimetalated phosphine intermediate, denoted as R1P(M)2, where M represents lithium, sodium, or potassium. This strategy allows for the reaction with a wide array of di- or polycarboxylic acid halides to form a stable intermediate of Formula III. Crucially, this intermediate can then be divergently processed: reacting with additional acid halides yields symmetric or asymmetric BAPO dimers, while reaction with alkyl or aryl halides produces MAPO variants. This modularity permits the creation of structures where R1 and R3 are distinctly different, a feat difficult to achieve with prior art. The ability to incorporate diverse arylene residues (Q) derived from commercially available phthalic, isophthalic, or naphthalene derivatives further expands the chemical space, enabling the design of initiators with customized absorption spectra and curing speeds.
Mechanistic Insights into Dimetalated Phosphine Acylation
The core of this synthetic breakthrough lies in the controlled nucleophilic attack of the dimetalated phosphine on the carbonyl carbon of the acid halide. The process begins with the generation of the highly reactive species R1P(M)2, typically achieved by treating a phosphine halide with an excess of alkali metal in an ether solvent like tetrahydrofuran at temperatures between -60°C and +120°C. This metalation step is critical, as the resulting dianion possesses sufficient nucleophilicity to displace halide ions from di- or tri-acid chlorides efficiently. The reaction stoichiometry is precisely managed, with approximately one equivalent of metalated phosphine used per equivalent of acyl chloride to favor the formation of the desired intermediate without excessive oligomerization. Maintaining an excess of phosphine during the dropwise addition of the acid chloride is a key operational parameter that ensures high selectivity for the target intermediate structure.
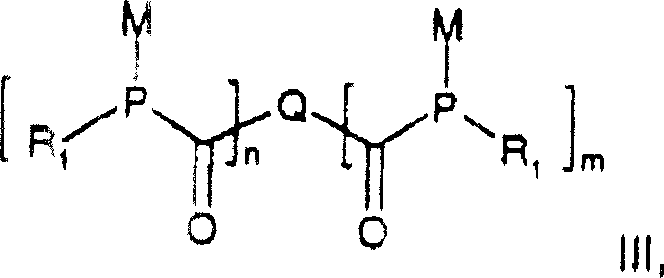
Following the formation of the acylphosphine intermediate (Formula III), the pathway diverges based on the desired final product. For BAPO synthesis, a second acylation step introduces the second acyl group, whereas MAPO synthesis involves alkylation or arylation. The final oxidation step converts the trivalent phosphorus species into the pentavalent phosphine oxide, which is the active photoinitiating moiety. This oxidation can be conveniently performed using common oxidants such as hydrogen peroxide, air, or pure oxygen, avoiding the need for expensive or toxic heavy metal oxidants. The stability of the intermediate Formula III is noteworthy, as it can exist in solution for several weeks under inert gas, providing a significant operational window for process optimization. This mechanistic robustness ensures consistent impurity profiles and high batch-to-batch reproducibility, which are essential metrics for R&D directors evaluating new supply chains.
How to Synthesize Dimeric BAPO Compounds Efficiently
The synthesis of these advanced photoinitiators follows a streamlined sequence that is amenable to scale-up. The process initiates with the preparation of the metalated phosphine precursor, followed by the controlled addition of polycarboxylic acid halides to generate the key acylated intermediate. Subsequent functionalization and oxidation steps yield the final high-purity product. Detailed standard operating procedures regarding specific molar ratios, temperature ramps, and workup protocols are critical for maximizing yield and minimizing byproduct formation. The following guide outlines the standardized synthesis steps derived from the patent data.
- Prepare dimetalated phosphine R1P(M)2 by reacting phosphine halides with alkali metals like lithium in THF at low temperatures.
- React the metalated phosphine with di- or polycarboxylic acid halides to form the key acylphosphine intermediate of Formula III.
- Treat the intermediate with further acid halides or alkyl halides, followed by oxidation using hydrogen peroxide or air to yield the final phosphine oxide.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthesis route offers substantial strategic benefits regarding cost structure and supply security. The reliance on widely available commodity chemicals such as phenylphosphine, various phthalic acid chlorides, and common solvents like THF and toluene mitigates the risk of raw material shortages. Unlike processes requiring exotic catalysts or specialized reagents, this method utilizes standard organic synthesis infrastructure, allowing for production across multiple geographic locations without significant capital expenditure on new equipment. The elimination of transition metal catalysts in the oxidation step further simplifies the downstream purification process, removing the need for expensive heavy metal scavenging resins and reducing the overall environmental footprint of the manufacturing operation.
- Cost Reduction in Manufacturing: The synthetic pathway significantly lowers production costs by utilizing inexpensive starting materials and avoiding complex catalytic systems. The ability to perform the reaction in common ether solvents at moderate temperatures reduces energy consumption compared to high-pressure or cryogenic alternatives. Furthermore, the stability of the intermediate allows for flexible batch scheduling, optimizing plant utilization rates. By removing the necessity for costly purification steps associated with metal catalyst removal, the overall cost of goods sold is drastically improved, offering a competitive pricing advantage in the global photoinitiator market.
- Enhanced Supply Chain Reliability: The use of commercially abundant precursors such as isophthaloyl chloride and terephthaloyl chloride ensures a resilient supply chain less susceptible to geopolitical disruptions. Since the synthesis does not depend on single-source proprietary catalysts, procurement managers can diversify their supplier base more effectively. The robustness of the chemistry means that production can be scaled rapidly to meet surging demand in the coatings and electronics sectors without the long lead times typically associated with commissioning new catalytic processes. This reliability is crucial for maintaining continuous production lines in downstream formulation facilities.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory to pilot scales using standard reactor configurations. The waste stream is primarily composed of inorganic salts and organic solvents which can be readily recovered and recycled, aligning with stringent environmental regulations. The avoidance of heavy metals simplifies wastewater treatment requirements, reducing compliance costs. Additionally, the high purity of the final product minimizes the need for extensive recrystallization, thereby reducing solvent waste volumes and enhancing the overall sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the operational parameters and product capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating these new initiators into existing formulations.
Q: What is the primary advantage of dimeric BAPO compounds over monomeric forms?
A: Dimeric and multimeric forms offer enhanced thermal stability and reduced volatility compared to monomers, making them superior for high-performance UV curing applications where migration and odor are critical concerns.
Q: Can this synthesis method produce asymmetric MAPO compounds?
A: Yes, the patented process specifically enables the preparation of asymmetric monoacylphosphine oxides where substituents R1 and R3 are different, providing greater flexibility in tuning photophysical properties.
Q: What are the typical reaction conditions for the metalation step?
A: The metalation is typically conducted in inert ether solvents like tetrahydrofuran (THF) at temperatures ranging from -60°C to +120°C, preferably using lithium as the metalating agent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BAPO Photoinitiator Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent technologies into commercial reality. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical expertise to manufacture these advanced dimeric BAPO compounds with stringent purity specifications. Our rigorous QC labs ensure that every batch meets the exacting standards required for high-performance UV curing applications, guaranteeing consistent reactivity and color stability. We understand the critical nature of supply continuity in the polymer additives sector and have established robust logistics networks to serve global markets efficiently.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their photoinitiator supply chains. By leveraging our manufacturing capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your next project. Let us help you unlock the full potential of next-generation photoinitiators for your coating and adhesive formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
