Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. A significant breakthrough in this domain is detailed in patent CN112480015B, which discloses a novel multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. Quinazolinone compounds represent a vital class of condensed nitrogen-containing six-membered heterocycles found in numerous bioactive molecules, exhibiting a wide spectrum of pharmacological activities including antifungal, antibacterial, antiviral, anti-inflammatory, and anticancer properties. The introduction of a trifluoromethyl group into these scaffolds is particularly strategic, as fluorine atoms significantly enhance physicochemical properties such as metabolic stability, lipophilicity, and bioavailability, thereby optimizing the drug-like characteristics of the parent molecule. This technological advancement addresses the growing demand for reliable pharmaceutical intermediate suppliers who can deliver high-purity, fluorinated heterocycles efficiently.
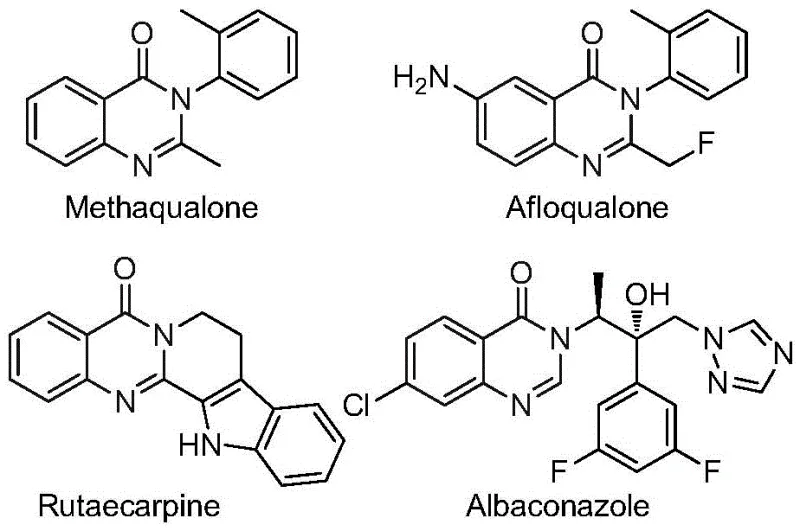
The structural diversity of quinazolinones is exemplified by market-known drugs such as methaqualone and afloqualone, highlighting the commercial importance of this chemical class. The ability to introduce a trifluoromethyl group at the 2-position opens new avenues for medicinal chemistry campaigns aimed at improving the efficacy and safety profiles of lead compounds. By leveraging this patented methodology, manufacturers can access a versatile library of derivatives that were previously difficult or costly to produce, positioning themselves as a preferred partner for cost reduction in API manufacturing where complex fluorinated motifs are required.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives has been plagued by significant operational and economic challenges that hinder large-scale adoption. Conventional synthetic strategies often rely on harsh reaction conditions, such as high-pressure carbon monoxide atmospheres, which necessitate specialized and expensive reactor equipment, thereby increasing capital expenditure and safety risks. Furthermore, traditional methods frequently utilize precious metal catalysts like ruthenium or platinum, which not only drive up raw material costs but also introduce complexities in downstream processing due to the stringent requirements for removing trace heavy metal impurities to meet regulatory standards. Many existing protocols also require pre-activated substrates, such as 2-bromoformylaniline or acid anhydrides, which are more expensive and less stable than simple nitro compounds. These factors collectively result in lower overall yields, narrow substrate scope, and a cumbersome workflow that is ill-suited for the rapid iteration needed in modern drug discovery and the efficiency demanded by commercial supply chains.
The Novel Approach
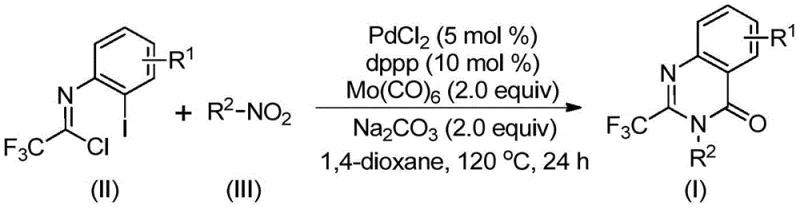
In stark contrast, the method described in patent CN112480015B offers a transformative solution by utilizing a palladium-catalyzed carbonylation cascade reaction that operates under much milder and more practical conditions. This innovative approach employs cheap and readily available nitro compounds and trifluoroethylimidoyl chloride as the primary starting materials, effectively bypassing the need for pre-activation or hazardous high-pressure gas cylinders. The reaction proceeds efficiently in common organic solvents like 1,4-dioxane at a moderate temperature of 120°C, utilizing molybdenum hexacarbonyl as a safe and solid carbon monoxide surrogate. This shift in strategy not only simplifies the operational procedure to a straightforward one-pot process but also dramatically expands the functional group tolerance, allowing for the synthesis of diverse substituted derivatives without compromising yield. The result is a highly practical and scalable protocol that significantly lowers the barrier to entry for producing these valuable heterocycles.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The success of this synthesis relies on a sophisticated yet elegant catalytic cycle driven by a palladium complex. The reaction initiates with the reduction of the nitro compound to the corresponding amine by molybdenum hexacarbonyl, which simultaneously serves as the source of carbon monoxide. This in situ generated amine then undergoes a base-promoted intermolecular carbon-nitrogen bond coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine derivative. Subsequently, the palladium catalyst, coordinated with the dppp ligand, inserts into the carbon-iodine bond of the intermediate to form a divalent palladium species. The carbon monoxide released from the molybdenum complex then inserts into the carbon-palladium bond, generating an acyl-palladium intermediate. This key step is followed by an intramolecular cyclization facilitated by the base, forming a seven-membered cyclic palladium intermediate. Finally, reductive elimination releases the desired 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst, completing the cycle. This mechanistic pathway ensures high atom economy and minimizes the formation of unwanted byproducts.
From a quality control perspective, the choice of ligands and additives plays a crucial role in suppressing side reactions and ensuring a clean impurity profile. The use of 1,3-bis(diphenylphosphino)propane (dppp) as the ligand optimizes the electronic and steric environment around the palladium center, promoting the specific insertion and elimination steps required for quinazolinone formation while discouraging alternative pathways that could lead to polymeric tars or dehalogenated impurities. Furthermore, the use of sodium carbonate as a mild base helps to neutralize acidic byproducts like HCl generated during the coupling step without causing hydrolysis of the sensitive imidoyl chloride or the final product. This precise control over the reaction environment allows for the production of high-purity pharmaceutical intermediates that meet the rigorous specifications required for subsequent drug substance manufacturing, reducing the burden on purification teams.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The experimental procedure outlined in the patent provides a clear roadmap for executing this transformation with high reproducibility. The process involves charging a reaction vessel with the palladium catalyst, ligand, base, carbon monoxide source, and the two primary organic substrates in an aprotic solvent. The mixture is then heated to facilitate the cascade reaction, after which standard workup procedures involving filtration and chromatography yield the pure product. This streamlined workflow eliminates the need for multiple isolation steps between the reduction, coupling, and cyclization events, saving both time and solvent. For detailed operational parameters and stoichiometry, please refer to the standardized guide below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow the carbonylation cascade reaction to proceed to completion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl substituted quinazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that directly impact the bottom line and operational resilience. The shift from expensive, pre-activated substrates to commodity chemicals like nitro compounds represents a fundamental change in the cost structure of the synthesis. Nitro compounds are ubiquitous in the chemical industry, produced on a massive scale for various applications, which ensures their availability and price stability even during periods of market volatility. By anchoring the supply chain on such robust raw materials, manufacturers can mitigate the risk of shortages that often plague specialized reagents. Additionally, the elimination of high-pressure carbon monoxide gas removes the need for specialized gas handling infrastructure and safety protocols, further reducing the overhead costs associated with facility maintenance and regulatory compliance.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the substitution of costly catalysts and substrates with more affordable alternatives. Traditional methods often require stoichiometric amounts of activating agents or expensive noble metals like ruthenium, whereas this method uses catalytic amounts of palladium with a simple phosphine ligand. The use of molybdenum hexacarbonyl as a solid CO source avoids the logistical costs and safety hazards associated with storing and transporting high-pressure gas cylinders. Furthermore, the high reaction efficiency and yields reported in the patent mean that less raw material is wasted, and the throughput per batch is maximized. These factors combine to deliver substantial cost savings in the manufacturing of complex fluorinated heterocycles, making the final API more competitive in the global marketplace.
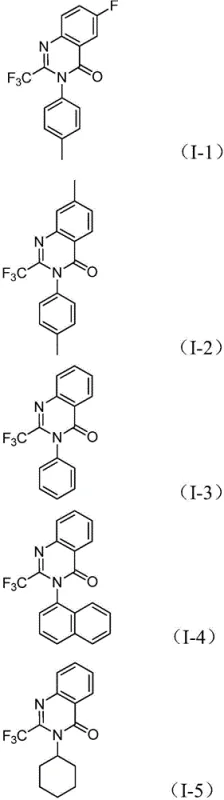
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for pharmaceutical production, and this method enhances reliability by relying on widely available starting materials. The substrates, including various substituted nitrobenzenes and trifluoroethylimidoyl chlorides, are commercially accessible from multiple vendors, reducing dependency on single-source suppliers. The robustness of the reaction conditions, which tolerate a wide range of functional groups as demonstrated by the successful synthesis of derivatives with methyl, halogen, and alkoxy substituents, means that the process is less sensitive to minor variations in raw material quality. This flexibility allows procurement teams to source materials based on best value without compromising the integrity of the synthesis, ensuring a steady flow of intermediates to downstream production lines.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often introduces unforeseen challenges, but this one-pot method is inherently designed for scalability. The reaction does not generate excessive waste streams associated with multi-step protection-deprotection sequences, and the use of standard solvents like 1,4-dioxane simplifies solvent recovery and recycling programs. The simplified workup procedure, involving basic filtration and chromatography, reduces the consumption of silica gel and eluents compared to more complex purifications. From an environmental standpoint, the avoidance of toxic gases and the use of lower catalyst loadings contribute to a greener manufacturing footprint. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the process attractive for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing portfolios. The answers reflect the specific conditions and outcomes observed during the development of this process, providing a realistic view of its capabilities and limitations in a production setting.
Q: What are the key advantages of this new synthesis method over conventional routes?
A: This method utilizes cheap and readily available nitro compounds and trifluoroethylimidoyl chloride as starting materials, avoiding the need for harsh high-pressure carbon monoxide conditions or expensive pre-activated substrates often required in traditional ruthenium or platinum-catalyzed processes. It offers high reaction efficiency, broad substrate compatibility, and simplified operation suitable for industrial scale-up.
Q: What catalyst system is employed in this carbonylation reaction?
A: The reaction employs a palladium catalyst system consisting of palladium chloride (PdCl2) and 1,3-bis(diphenylphosphino)propane (dppp) as the ligand. Molybdenum hexacarbonyl (Mo(CO)6) serves as both the carbon monoxide source and a reducing agent for the nitro group, facilitating the cascade transformation under mild thermal conditions.
Q: Can this process be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the patent explicitly states that the method can be expanded to the gram level and provides convenience for large-scale application in industrial production. The use of standard organic solvents like 1,4-dioxane and commercially available reagents ensures that the process is robust and adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and securing supply chains. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like the one described in CN112480015B can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-trifluoromethyl quinazolinone meets the highest international standards. Our commitment to quality and consistency makes us a trusted partner for multinational pharmaceutical companies seeking to optimize their API manufacturing processes.
We invite you to explore how this innovative synthesis route can benefit your specific projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions. Let us collaborate to bring your next generation of therapeutics to market faster and more efficiently.
