Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as the backbone of modern therapeutics. Patent CN112125856A introduces a groundbreaking methodology for the preparation of 2-trifluoromethyl substituted quinazolinone derivatives, addressing critical challenges in safety and efficiency. Quinazolinones are privileged structures found in numerous bioactive molecules exhibiting anti-inflammatory, antiviral, antifungal, and anticancer properties. The strategic incorporation of a trifluoromethyl group at the 2-position further enhances these pharmacological profiles by improving metabolic stability and lipophilicity. This technical insight report analyzes the novel palladium-catalyzed carbonylation tandem reaction described in the patent, highlighting its potential to revolutionize the supply chain for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl quinazolinones has been plagued by significant operational hazards and inefficiencies. Traditional protocols often rely on the direct use of carbon monoxide gas, a highly toxic and colorless substance that requires specialized high-pressure equipment and rigorous safety protocols, thereby inflating capital expenditure and operational risk. Alternative methods involving cyclization with ethyl trifluoroacetate or trifluoroacetic anhydride frequently suffer from harsh reaction conditions, necessitating extreme temperatures or strong acids that limit functional group tolerance. Furthermore, many existing routes utilize expensive or unstable precursors, such as unstable trifluoroacetamides, which complicate storage and logistics. These factors collectively result in lower overall yields, narrow substrate scopes, and increased production costs, making large-scale commercialization difficult for many contract development and manufacturing organizations.
The Novel Approach
The methodology disclosed in CN112125856A represents a paradigm shift by employing a transition metal palladium-catalyzed carbonylation tandem reaction that utilizes a solid carbon monoxide substitute. Instead of handling dangerous CO gas, the process leverages 1,3,5-tricarboxylate phenol ester (TFBen), which decomposes under heating conditions to release carbon monoxide in situ. This innovation drastically simplifies the reactor setup and enhances workplace safety. The reaction couples readily available o-iodoanilines with trifluoroethylimidoyl chlorides in the presence of a palladium catalyst and a phosphine ligand. As illustrated in the reaction scheme below, this approach allows for the efficient construction of the quinazolinone core under relatively mild conditions.
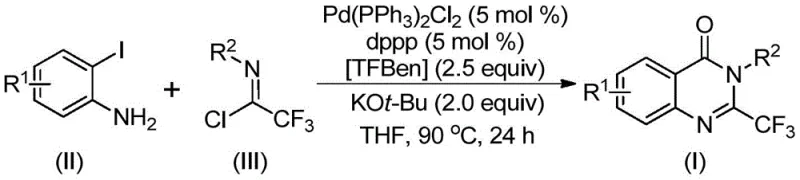
This novel route not only mitigates safety risks but also expands the chemical space accessible to medicinal chemists. The use of stable, commercially available starting materials ensures a reliable supply chain, while the mild reaction conditions preserve sensitive functional groups that might be degraded in traditional acidic or high-temperature environments. Consequently, this method offers a superior balance of safety, cost-effectiveness, and chemical versatility.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
Understanding the mechanistic pathway is crucial for optimizing process parameters and ensuring consistent product quality. The reaction likely initiates with an intermolecular carbon-nitrogen bond coupling promoted by the base, potassium tert-butoxide, generating a trifluoroacetamidine derivative intermediate. Subsequently, the palladium catalyst, specifically bis(triphenylphosphine)palladium(II) dichloride coordinated with 1,3-bis(diphenylphosphino)propane (dppp), undergoes oxidative addition into the carbon-iodine bond of the aniline substrate. This forms a key divalent palladium intermediate. Upon heating, the solid CO surrogate TFBen decomposes, releasing carbon monoxide which then inserts into the carbon-palladium bond to form an acyl palladium species. The base facilitates the formation of a palladium-nitrogen bond, leading to a seven-membered ring palladium intermediate. Finally, reductive elimination occurs to release the final 2-trifluoromethyl substituted quinazolinone derivative and regenerate the active catalyst. This intricate cycle ensures high atom economy and selectivity.
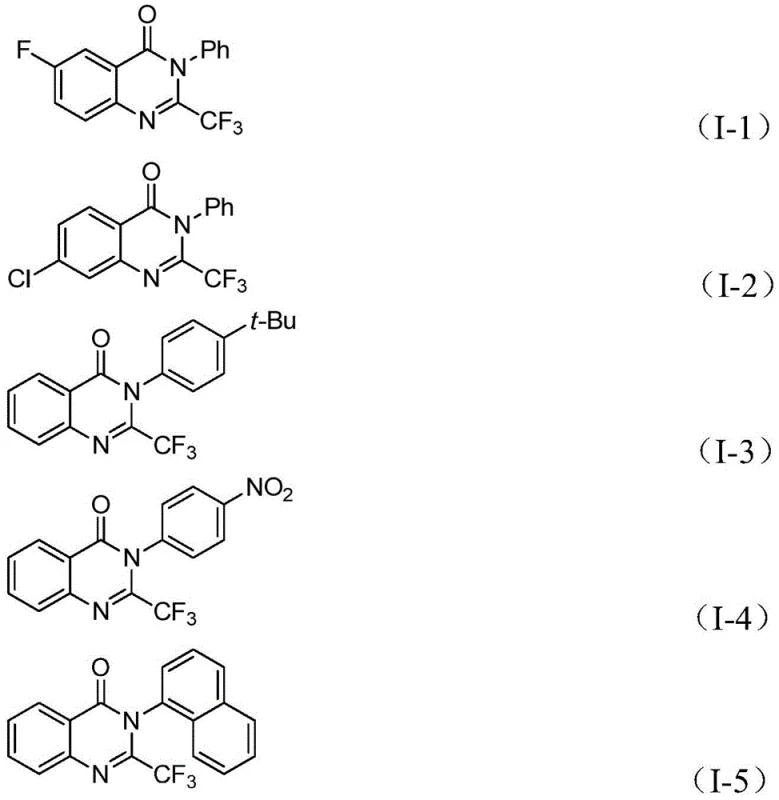
From an impurity control perspective, the mild nature of this catalytic cycle is advantageous. Harsh conditions often lead to decomposition of the trifluoromethyl group or polymerization of reactive intermediates. By maintaining the reaction at 90°C in tetrahydrofuran (THF), the process minimizes thermal degradation pathways. The specificity of the palladium insertion and the controlled release of CO from TFBen prevent the formation of over-carbonylated byproducts or homocoupling impurities often seen in uncontrolled gas-phase carbonylations. The broad substrate tolerance, accommodating electron-withdrawing groups like nitro and halogens as well as electron-donating alkyl groups, further confirms the robustness of the catalytic system against electronic variations in the substrate.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical implementation of this synthesis involves a straightforward procedure suitable for standard laboratory and pilot plant equipment. The process begins by charging a reaction vessel with the palladium catalyst, ligand, base, solid CO source, and the two primary organic substrates in an aprotic solvent. The mixture is then heated to facilitate the tandem reaction sequence. Detailed standardized operating procedures regarding stoichiometry, addition rates, and work-up protocols are essential for reproducibility. For a comprehensive step-by-step guide on executing this synthesis with optimal yield and purity, please refer to the technical documentation provided below.
- Combine palladium catalyst, ligand, solid CO substitute (TFBen), base, trifluoroethylimidoyl chloride, and o-iodoaniline in an organic solvent.
- Heat the reaction mixture to 90°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Perform post-treatment including filtration and silica gel purification to isolate the final 2-trifluoromethyl substituted quinazolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical curiosity. The shift away from hazardous gases and expensive activated substrates directly impacts the total cost of ownership and supply reliability. By adopting this method, manufacturers can mitigate regulatory hurdles associated with toxic gas handling and reduce dependency on niche, high-cost reagents. The following points detail how this technology translates into commercial value.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide infrastructure significantly lowers capital expenditure requirements for production facilities. Furthermore, the use of cheap and readily available starting materials, such as substituted o-iodoanilines and trifluoroethylimidoyl chlorides, reduces raw material costs compared to specialized cyclic precursors. The avoidance of pre-activation steps simplifies the process flow, reducing labor hours and energy consumption associated with multi-step sequences. This streamlined approach inherently drives down the cost per kilogram of the final active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: The starting materials for this reaction are commodity chemicals or easily synthesized derivatives that are widely available in the global market. Unlike methods relying on unstable intermediates that require immediate use or cold chain logistics, the reagents here are stable solids or liquids with long shelf lives. This stability ensures consistent inventory management and reduces the risk of production stoppages due to reagent degradation. The robustness of the reaction across various substituents also means that supply chains are less vulnerable to shortages of specific, narrowly defined precursors.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is facilitated by the use of standard batch reactors without the need for specialized gas containment systems. The use of a solid CO surrogate aligns with green chemistry principles by minimizing the release of toxic gases into the environment. Waste streams are easier to manage as they primarily consist of organic solvents and solid residues rather than complex gas scrubbing byproducts. This environmental profile simplifies permitting processes and supports corporate sustainability goals, making the technology attractive for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent literature, providing clarity for technical decision-makers evaluating this route for adoption.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: This method utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide substitute, effectively eliminating the need for handling toxic and hazardous carbon monoxide gas directly.
Q: What types of substituents are compatible with this palladium-catalyzed reaction?
A: The reaction demonstrates excellent substrate compatibility, tolerating various groups such as halogens (F, Cl, Br), alkyl groups, trifluoromethyl groups, and nitro groups on both the aniline and imidoyl chloride components.
Q: Why is the 2-trifluoromethyl group significant in quinazolinone drug design?
A: Introducing a trifluoromethyl group significantly enhances the physicochemical properties of the parent molecule, including improved metabolic stability, lipophilicity, and bioavailability, which are critical for drug efficacy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
The technological advancements presented in patent CN112125856A underscore the evolving landscape of pharmaceutical intermediate manufacturing, where safety and efficiency are paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is evidenced by our stringent purity specifications and rigorous QC labs, ensuring that every batch of 2-trifluoromethyl quinazolinone meets the exacting standards required for downstream drug synthesis. We possess the technical expertise to adapt this palladium-catalyzed protocol to meet specific client needs, ensuring a seamless transition from development to full-scale manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer, more efficient method. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your project requirements, ensuring a reliable partnership for your critical pharmaceutical intermediates.
