Advanced Crystallization Technology for High-Purity Iodixanol Manufacturing and Commercial Scale-Up
Advanced Crystallization Technology for High-Purity Iodixanol Manufacturing and Commercial Scale-Up
The pharmaceutical industry's demand for non-ionic X-ray contrast agents has driven intense innovation in process chemistry, specifically targeting the purification bottlenecks associated with complex iodinated molecules. Patent CN101830826B represents a pivotal advancement in this domain, detailing a sophisticated crystallization protocol for Iodixanol (Visipaque 320) that fundamentally alters the economic and technical landscape of its production. This technology addresses the critical challenge of purifying crude Iodixanol, which typically emerges from dimerization reactions with purity levels between 75% and 90%, containing persistent impurities such as unreacted Compound A and various side products. By leveraging a precise ternary solvent system comprising water, methanol, and isopropanol (Virahol), this method achieves pharmacopeia-compliant purity exceeding 98% without resorting to costly chromatographic techniques. For R&D directors and supply chain strategists, this patent offers a blueprint for transforming a traditionally low-yield, high-cost purification step into a robust, scalable unit operation that enhances overall process efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Iodixanol has been plagued by significant technical and economic inefficiencies inherent to earlier synthetic routes described in patents such as EP 108638. The primary bottleneck in these conventional processes was the reliance on preparative liquid phase chromatography to isolate the final drug substance from the reaction mixture. While chromatography is effective for analytical separation, its application on an industrial manufacturing scale introduces prohibitive capital expenditures for column hardware and substantial ongoing operational costs related to stationary phase replacement and solvent consumption. Furthermore, chromatographic processes are inherently batch-limited and slow, often requiring multiple cycles to achieve the necessary purity, which drastically extends the total production lead time. The complexity of managing large volumes of eluents and the difficulty in recycling solvents further exacerbate the environmental footprint, making these legacy methods unsustainable for modern, high-volume API manufacturing where cost-per-kilogram is a decisive competitive factor.
The Novel Approach
The innovative methodology disclosed in CN101830826B circumvents these historical limitations by replacing chromatography with a highly optimized crystallization process driven by thermodynamic solubility control. Instead of separating components based on adsorption affinity, this approach exploits the differential solubility of Iodixanol and its impurities in a specific mixture of water, methanol, and isopropanol. The process begins with the crude product obtained from the dimerization of Compound A, as illustrated in the reaction scheme below, which highlights the structural complexity necessitating such rigorous purification.
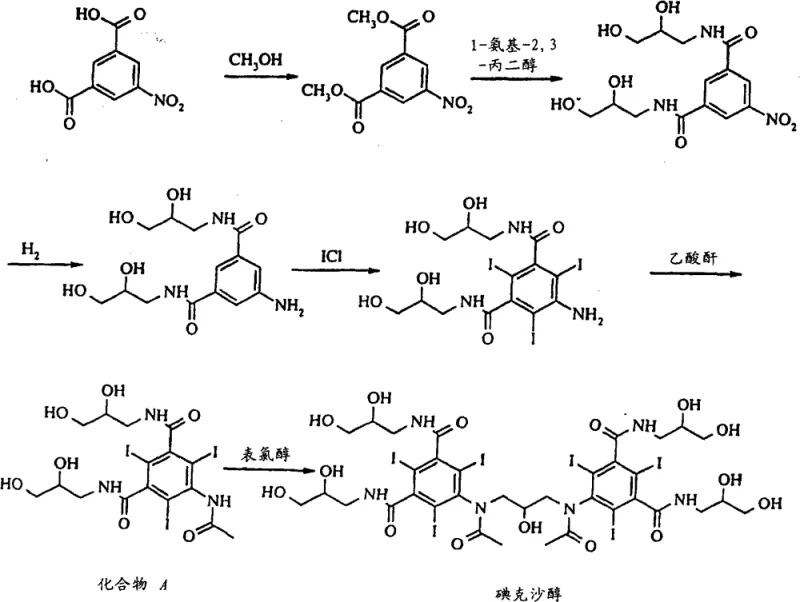
By carefully adjusting the water content and sequentially adding methanol followed by a controlled, gradual introduction of isopropanol, the process induces a state of supersaturation that favors the nucleation and growth of pure Iodixanol crystals. This strategy effectively excludes impurities like Compound A and Schering AG byproducts into the mother liquor, allowing for their removal via simple filtration. The result is a dramatic simplification of the downstream processing workflow, eliminating the need for expensive chromatography resins and reducing the total processing time to merely 1 to 4 days. This shift from a separation-intensive model to a crystallization-driven model represents a paradigm shift in the manufacturing of high-value contrast media, offering a pathway to significantly lower production costs and higher throughput.
Mechanistic Insights into Solvent-Mediated Crystallization
The success of this purification strategy hinges on a deep understanding of the solubility dynamics of polyhydroxylated, triiodinated aromatic compounds in mixed solvent systems. Iodixanol possesses multiple hydrophilic hydroxypropyl groups alongside hydrophobic iodine-substituted aromatic rings, creating a complex solvation shell that is highly sensitive to solvent polarity. The mechanism operates on the principle of anti-solvent crystallization, where methanol acts as a primary co-solvent to maintain the drug substance in solution at elevated temperatures, while isopropanol serves as the anti-solvent to reduce solubility upon cooling or concentration. Crucially, the patent emphasizes the management of water content; since water significantly increases the solubility of Iodixanol, failing to reduce water levels prior to the addition of organic solvents would prevent the system from reaching the necessary supersaturation point. The gradual addition of isopropanol allows for the controlled growth of crystals rather than rapid, chaotic precipitation, which is essential for excluding occluded impurities and ensuring the formation of large, filterable crystals with high lattice purity.
Furthermore, the mechanistic advantage extends to the handling of specific impurities generated during the upstream dimerization of Compound A. The solvent mixture is tuned such that the solubility of the target Iodixanol drops precipitously while the solubility of key impurities remains relatively high, effectively partitioning them into the liquid phase. This selectivity is achieved through the specific ratio of methanol to isopropanol (approximately 1:1 to 1:2 by volume relative to the drug mass), which creates a dielectric environment unfavorable for the dissolution of the highly symmetric Iodixanol molecule but favorable for the more polar or less symmetric byproducts. This precise control over the crystallization trajectory ensures that the final solid form meets stringent quality specifications without the need for recrystallization loops, thereby maximizing the overall yield of the process which can reach 85% to 90% relative to the crude input.
How to Synthesize Iodixanol Efficiently
The implementation of this crystallization technology requires strict adherence to the defined solvent addition profiles and temperature controls to replicate the high purity and yield demonstrated in the patent examples. The process transforms a complex purification challenge into a standardized unit operation that can be readily integrated into existing GMP manufacturing facilities. Operators must focus on the precise metering of isopropanol to maintain the supersaturation curve within the metastable zone, avoiding both under-saturation which yields no product and over-saturation which leads to oiling out or amorphous precipitation. The detailed standardized synthesis steps, including specific flow rates and temperature ramps, are outlined in the technical guide below.
- Perform dimerization of 5-acetamido-N,N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-isophthalamide (Compound A) using a dimerization agent like epichlorohydrin in a non-aqueous solvent.
- Adjust the water content in the crude aqueous solution to a specific concentration range to optimize solubility parameters prior to solvent addition.
- Add methanol followed by a gradual, controlled addition of isopropanol (Virahol) to induce supersaturation and crystal growth without trapping impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this crystallization technology translates directly into tangible improvements in cost structure and supply reliability. By eliminating the dependency on preparative liquid chromatography, manufacturers can remove a major capital expenditure barrier and significantly reduce the variable costs associated with stationary phase materials and high-volume solvent waste disposal. The simplification of the process flow also reduces the risk of batch failures associated with complex chromatographic separations, leading to a more predictable and stable supply of the active pharmaceutical ingredient. This stability is crucial for maintaining continuous production schedules for contrast media, which are essential diagnostic tools in global healthcare systems.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete removal of chromatographic purification steps, which are notoriously expensive due to the cost of resin and the energy required for pumping large solvent volumes. By switching to a crystallization-based purification, the process utilizes commodity solvents like methanol and isopropanol, which are inexpensive and easily recoverable through distillation. This fundamental shift in the separation mechanism drastically lowers the cost of goods sold (COGS) per kilogram of Iodixanol, allowing for more competitive pricing in the generic contrast media market while preserving healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: Traditional chromatography processes are prone to column fouling and resin degradation, which can cause unplanned downtime and disrupt supply continuity. The robust nature of the solvent-based crystallization method described in the patent minimizes these operational risks, as the equipment involved (reactors, filters, dryers) is standard and less prone to failure. Additionally, the shorter processing time of 1 to 4 days compared to multi-day chromatographic runs accelerates the manufacturing cycle, enabling faster response times to market demand fluctuations and reducing the inventory holding costs associated with work-in-progress materials.
- Scalability and Environmental Compliance: Scaling up chromatographic processes is technically challenging and often requires linear increases in column size and solvent usage, leading to massive waste streams. In contrast, crystallization scales efficiently with vessel volume, and the solvents used in this process (methanol and isopropanol) are well-established in the industry with mature recovery and recycling infrastructure. This facilitates a greener manufacturing profile by reducing the E-factor (mass of waste per mass of product) and simplifying compliance with increasingly stringent environmental regulations regarding solvent emissions and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this crystallization technology, derived directly from the patent's experimental data and claims. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for commercial production. The answers reflect the specific advantages of the water-methanol-isopropanol system over legacy purification methods.
Q: Why is the new crystallization method superior to preparative liquid chromatography?
A: Conventional methods relying on preparative liquid chromatography are prohibitively expensive and time-consuming for industrial scale. The novel solvent-based crystallization eliminates the need for complex chromatography columns, significantly reducing operational costs and processing time while achieving pharmacopeia-grade purity.
Q: What is the critical role of water content in this crystallization process?
A: Water content is a critical parameter because even small amounts can drastically increase the solubility of Iodixanol in the mother liquor, preventing effective crystallization. The process strictly controls water levels before adding organic anti-solvents to ensure maximum yield and purity.
Q: Can this process handle crude product with varying purity levels?
A: Yes, the method is robust enough to purify crude Iodixanol with purity levels ranging from 75% to 90%. The specific solvent mixture of water, methanol, and isopropanol effectively separates the target API from impurities like Compound A and Schering AG byproducts in a single crystallization step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iodixanol Supplier
The technological breakthroughs detailed in CN101830826B underscore the immense potential for optimizing the production of high-value contrast media intermediates and APIs. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such complex laboratory-scale innovations into reliable, commercial-scale manufacturing realities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate solvent controls and crystallization parameters required for Iodixanol are meticulously managed. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the exacting standards required for parenteral administration, providing our partners with absolute confidence in product quality.
We invite global pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain needs. By leveraging our advanced crystallization capabilities, we can help you reduce the total cost of ownership for your contrast media portfolio while ensuring uninterrupted supply. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering excellence can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
