Advanced Iodixanol Manufacturing: Technical Breakthroughs in Dimerization and Purification for Commercial Scale
The pharmaceutical industry continuously seeks robust manufacturing pathways for high-value contrast agents, and the technical disclosures within patent CN102079716A represent a significant leap forward in the synthesis of Iodixanol. This specific intellectual property outlines a refined methodology that addresses the longstanding challenges of yield optimization and impurity control in the production of this non-ionic X-ray contrast medium. By integrating a sophisticated buffer system during the critical dimerization phase, the process effectively mitigates the formation of undesirable alkoxy by-products that typically plague conventional synthesis routes. The strategic implementation of boron-containing acidic substances allows for precise pH modulation, creating a reaction environment that favors the transformation of the precursor Compound A into the target dimer with exceptional efficiency. Furthermore, the subsequent purification strategy replaces costly and complex preparative liquid chromatography with a scalable macroporous adsorbent resin technique. This shift not only enhances the economic viability of the production line but also ensures that the final bulk drug substance meets the stringent purity requirements necessary for direct intravascular injection. For stakeholders evaluating the commercial potential of this technology, the combination of high conversion rates and streamlined purification offers a compelling value proposition for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Iodixanol has been hindered by synthetic routes that suffer from low overall recovery and excessive purification costs. Traditional methods, such as those described in earlier patents like EP 0108638, often rely on preparative liquid chromatography to achieve the necessary pharmaceutical grade purity. While effective in a laboratory setting, this technique is notoriously difficult to translate into high-volume commercial production due to its high operational expenditure and technical complexity. Additionally, conventional dimerization reactions frequently lack precise pH control, leading to significant side reactions such as excessive alkylation and the generation of alkoxy impurities. These side products not only reduce the yield of the desired Iodixanol but also complicate the downstream purification process, requiring multiple crystallization steps that extend the production cycle significantly. The reliance on harsh chlorinating agents in some prior art routes further introduces safety and environmental concerns, adding another layer of difficulty to the manufacturing process. Consequently, the total cost of goods for Iodixanol produced via these legacy methods remains prohibitively high, limiting accessibility and profitability for manufacturers operating in competitive global markets.
The Novel Approach
The innovative approach detailed in the patent data introduces a paradigm shift by focusing on reaction condition optimization and efficient solid-phase purification. Central to this new methodology is the use of a buffer solution formed by neutralizing excess alkali with boron-containing acids, which maintains the reaction pH within a narrow window of 10 to 11. This precise control drastically suppresses the formation of side products, resulting in a crude reaction mixture that already contains 85-90% Iodixanol. By improving the quality of the crude input, the burden on the purification stage is significantly reduced, allowing for the use of macroporous adsorbent resin chromatography instead of expensive liquid chromatography. This resin-based purification effectively removes inorganic salts and organic impurities, elevating the purity to over 97% with a total recovery rate of 90-95%. The final recrystallization step, utilizing a mixed solvent system of 2-methoxyethanol and alcohols, polishes the product to exceed 99% purity. This holistic improvement in both synthesis and purification creates a streamlined workflow that is inherently more suitable for industrial scale-up and cost-effective manufacturing.
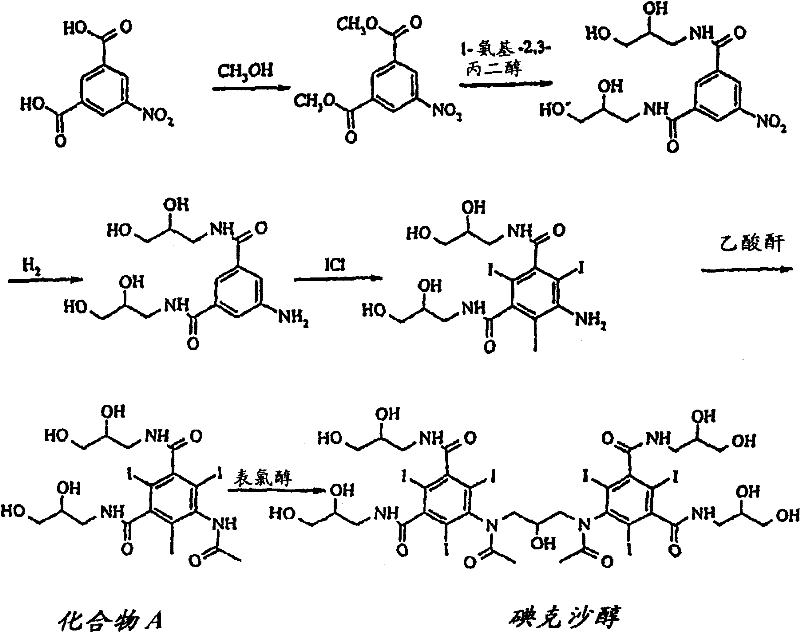
Mechanistic Insights into Buffered Dimerization and Resin Purification
The core chemical innovation lies in the dimerization of 5-acetamido-N,N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-isophthalamide, commonly referred to as Compound A. In this reaction, the control of nucleophilic substitution is paramount to preventing the formation of mono-alkylated or over-alkylated species. The introduction of a boron-based buffer system serves as a kinetic regulator, ensuring that the concentration of reactive hydroxide ions remains optimal for the coupling with dimerization reagents like Epichlorohydrin. By maintaining the pH between 10 and 11, the reaction environment discourages the attack of alkoxide ions on the epoxide ring in uncontrolled manners, which would otherwise lead to alkoxy impurities. This mechanistic precision ensures that the transformation ratio of Compound A to Iodixanol is maximized, with unreacted starting material minimized to less than 5%. The use of water or aqueous alcohol mixtures as solvents further facilitates heat dissipation and mass transfer, contributing to the reproducibility of the reaction on a large scale. This level of mechanistic control is essential for R&D directors who prioritize impurity profile management and batch-to-batch consistency in API production.
Following the synthesis, the purification mechanism leverages the differential adsorption properties of macroporous resins to separate the target molecule from contaminants. Unlike ion-exchange resins that rely on charge interactions, macroporous adsorbent resins such as Amberlite XAD-16 operate through hydrophobic interactions and pore size exclusion. This allows the resin to retain organic impurities and the target Iodixanol while allowing inorganic salts to pass through during the initial water wash. Subsequent elution with diluted alcohol solutions selectively recovers the Iodixanol, leaving behind higher molecular weight by-products or more polar impurities. The ability to recycle the mother liquor through the resin column further enhances the overall yield, recovering valuable product that would otherwise be lost in traditional crystallization processes. This multi-stage purification logic ensures that the final crude product entering the recrystallization phase is of exceptionally high quality, thereby guaranteeing that the final API meets the rigorous standards required for injectable medications without the need for repetitive, yield-eroding purification cycles.
How to Synthesize Iodixanol Efficiently
The synthesis of Iodixanol via this patented route requires careful attention to reagent stoichiometry and temperature control to replicate the high yields reported in the technical data. The process begins with the dissolution of Compound A in an aqueous alkali solution, followed by the critical addition of the boron-based buffer to establish the correct pH environment before introducing the dimerization agent. Operators must monitor the reaction progress closely, typically using HPLC, to ensure that the conversion of Compound A reaches completion within the expected timeframe, usually indicated by the residual starting material dropping below 5%. Once the reaction is quenched and neutralized, the crude mixture is subjected to the macroporous resin column, where elution gradients must be precisely managed to separate the product from salts and impurities effectively. The detailed standardized synthesis steps see the guide below.
- Dissolve Compound A in an aqueous alkali solution and cool the mixture to below 20°C to prepare for the dimerization reaction.
- Add a boron-containing acidic substance to form a buffer solution, adjusting the pH to between 10 and 11 to inhibit side reactions.
- Introduce the dimerization reagent such as Epichlorohydrin, monitor the reaction until Compound A conversion is complete, and proceed to purification.
- Purify the crude reaction liquid using a macroporous adsorbent resin chromatography column to remove inorganic salts and organic impurities.
- Perform recrystallization on the purified crude product using a mixed solvent system containing 2-methoxyethanol to achieve over 99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented manufacturing process translates into tangible operational efficiencies and risk mitigation. The elimination of preparative liquid chromatography removes a significant bottleneck associated with high capital equipment costs and complex maintenance requirements, thereby lowering the barrier to entry for large-scale production. The robust nature of the buffer-controlled synthesis reduces the variability in crude product quality, which in turn stabilizes the supply chain by minimizing the risk of batch failures or off-specification results. Furthermore, the high recovery rates achieved through the resin purification and recrystallization steps mean that raw material utilization is optimized, directly contributing to a reduction in the cost of goods sold. The use of readily available solvents and reagents, such as 2-methoxyethanol and common alkalis, ensures that the supply chain is not dependent on exotic or hard-to-source chemicals that could introduce procurement delays. This combination of process stability, material efficiency, and equipment simplification creates a resilient manufacturing framework capable of meeting high-volume demand without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The transition from preparative liquid chromatography to macroporous adsorbent resin chromatography represents a substantial decrease in operational expenditures. Preparative HPLC systems require high-pressure pumps, specialized columns, and significant energy consumption, all of which drive up the cost per kilogram of the final product. By replacing this with a resin-based column that operates under lower pressure and utilizes reusable matrices, the manufacturing process becomes significantly more economical. Additionally, the improved conversion efficiency in the dimerization step reduces the amount of expensive iodinated starting material that is wasted as by-products. This qualitative improvement in material efficiency means that less raw material is required to produce the same amount of API, leading to direct cost savings that can be passed on to customers or retained as margin. The simplified purification workflow also reduces labor hours and solvent consumption, further enhancing the overall cost-effectiveness of the production line.
- Enhanced Supply Chain Reliability: The robustness of the buffered synthesis method ensures consistent batch quality, which is critical for maintaining a reliable supply of pharmaceutical intermediates. Variability in chemical synthesis often leads to production delays as teams work to troubleshoot off-spec batches, but the precise pH control mechanism described in the patent minimizes this risk. By stabilizing the reaction output, manufacturers can adhere to stricter delivery schedules and reduce the safety stock required to buffer against production uncertainties. The use of common industrial solvents and reagents also mitigates the risk of supply disruptions caused by the scarcity of specialized chemicals. This reliability is essential for downstream formulators who depend on a steady stream of high-purity Iodixanol to manufacture finished contrast agent injections. A stable and predictable manufacturing process allows supply chain planners to optimize inventory levels and reduce the lead time associated with procurement and production planning.
- Scalability and Environmental Compliance: The design of this synthesis and purification process is inherently scalable, moving seamlessly from laboratory validation to multi-ton commercial production. The use of macroporous resins and standard crystallization equipment avoids the need for specialized high-pressure infrastructure that is difficult to scale. From an environmental perspective, the process reduces the volume of hazardous waste generated by eliminating the need for large quantities of chromatography solvents associated with HPLC. The ability to recover and recycle unreacted starting materials and mother liquors through the resin column further minimizes waste discharge. This alignment with green chemistry principles not only reduces disposal costs but also ensures compliance with increasingly stringent environmental regulations. Manufacturers adopting this technology can position themselves as sustainable partners, appealing to global clients who prioritize environmental responsibility in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Iodixanol synthesis technology. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and technical feasibility for potential partners. Understanding these details is crucial for making informed decisions about integrating this process into existing manufacturing portfolios. The insights provided here reflect the consensus of technical experts who have analyzed the patent data for industrial applicability.
Q: How does the buffered dimerization method improve Iodixanol purity compared to conventional methods?
A: The patented method utilizes a boron-based buffer system to strictly control the reaction pH between 10 and 11. This specific control effectively inhibits excessive alkylation and the formation of alkoxy by-products, which are common issues in traditional synthesis routes. By minimizing these side reactions at the source, the crude product purity is significantly elevated to 85-90%, reducing the burden on downstream purification steps and ensuring a higher quality final API.
Q: What are the advantages of using macroporous adsorbent resin over preparative liquid chromatography for purification?
A: Traditional preparative liquid chromatography is often cost-prohibitive and difficult to scale for industrial production due to high operational complexity and expense. The use of macroporous adsorbent resin, such as polystyrene or polyacrylic ester matrices, offers a robust alternative that efficiently separates inorganic salts and organic impurities. This method allows for high recovery rates of 90-95% and achieves crude purity levels exceeding 97% without the economic and logistical constraints associated with high-pressure liquid chromatography systems.
Q: Why is the recrystallization solvent system critical for meeting pharmacopeia standards?
A: The final purity required for injectable contrast agents like Iodixanol is extremely high, often exceeding 99%. The patented recrystallization process employs a specific mixed solvent system containing 2-methoxyethanol and a solubility promoter like propanol. This combination ensures the effective removal of trace impurities and residual starting materials that survive the resin purification step. The controlled crystallization kinetics in this solvent system yield a product with a clean impurity profile that strictly adheres to rigorous pharmacopeia specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iodixanol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, leveraging advanced technologies like the buffered dimerization process to deliver superior pharmaceutical intermediates. Our expertise extends beyond simple synthesis; we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Iodixanol meets the highest industry standards. By combining our technical capabilities with a commitment to quality, we provide a secure and reliable source for high-value contrast agent intermediates that supports your long-term product development goals.
We invite you to explore how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall procurement costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate the tangible benefits of partnering with us. Let us collaborate to drive innovation and efficiency in your pharmaceutical production pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
