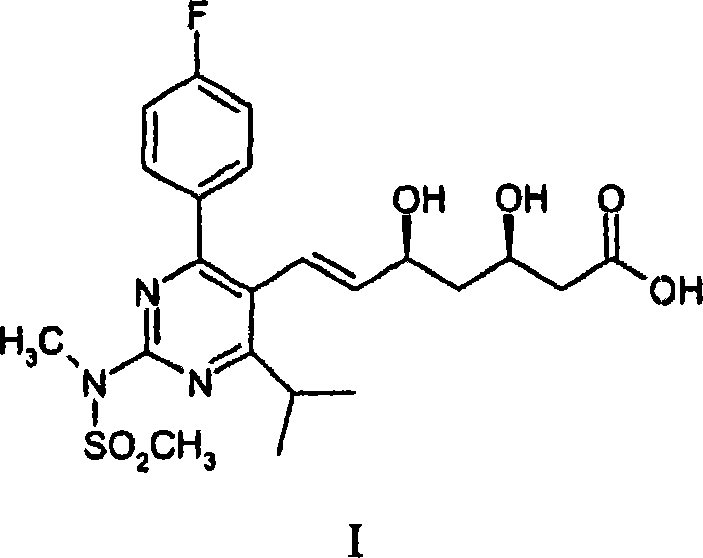
Scalable Manufacturing of Rosuvastatin Intermediates Using Novel Palladium-Catalyzed Heck Coupling Technology
Scalable Manufacturing of Rosuvastatin Intermediates Using Novel Palladium-Catalyzed Heck Coupling Technology
The pharmaceutical industry continuously seeks robust and scalable pathways for high-volume statin production, and patent CN101084197A presents a significant technological leap in this domain. This intellectual property discloses a highly efficient method for forming the core structure of Rosuvastatin, a potent HMG-CoA reductase inhibitor, through a palladium-catalyzed Heck reaction. Unlike traditional approaches that often rely on phosphorus ylides or complex condensation reactions requiring stringent anhydrous conditions, this novel process leverages the power of cross-coupling chemistry to join a functionalized pyrimidine ring with a chiral side-chain precursor. The technical breakthrough lies in the successful application of Heck coupling to fully substituted, sterically hindered six-membered pyrimidine systems, a feat previously considered challenging due to electronic and steric deactivation. By utilizing specific ligand-catalyst combinations, the method achieves high convergence, allowing for the late-stage assembly of the molecule which simplifies the overall synthetic tree and enhances the purity profile of the final active pharmaceutical ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Rosuvastatin and its analogues has relied heavily on Wittig-type olefination or Horner-Wadsworth-Emmons reactions to construct the critical carbon-carbon double bond linking the heterocyclic core to the heptanoic acid side chain. These conventional methodologies, while effective on a laboratory scale, present substantial hurdles when translated to industrial manufacturing environments. The use of phosphorus-based reagents often generates stoichiometric amounts of phosphine oxide byproducts, which are notoriously difficult to remove from the final product stream, thereby complicating purification and increasing waste disposal costs. Furthermore, these reactions frequently require cryogenic temperatures and strictly anhydrous solvents to maintain reagent stability and control stereochemistry, leading to high energy consumption and operational complexity. The multi-step protection and deprotection sequences often associated with these older routes also result in lower overall yields and increased material throughput, creating bottlenecks in supply chains that struggle to meet the global demand for cardiovascular medications.
The Novel Approach
In stark contrast, the methodology outlined in CN101084197A introduces a convergent strategy centered on a palladium-catalyzed Heck coupling reaction. This approach fundamentally shifts the bond disconnection logic, enabling the direct coupling of a halogenated pyrimidine intermediate with a vinyl-functionalized side chain. The novelty is not merely in the reaction type but in the specific adaptation to the sterically crowded pyrimidine scaffold. By employing bulky trialkylphosphine ligands, specifically tri-tert-butylphosphine, the catalyst system overcomes the steric hindrance that typically inhibits oxidative addition in such substrates. This allows the reaction to proceed under relatively mild thermal conditions, often in mixed solvent systems containing water, which significantly reduces the reliance on volatile organic compounds. The result is a streamlined process that minimizes byproduct formation, simplifies downstream processing, and offers a more environmentally benign manufacturing footprint, addressing both economic and regulatory pressures faced by modern pharmaceutical producers.
Mechanistic Insights into Palladium-Catalyzed Heck Coupling
The success of this synthetic route hinges on the intricate mechanistic cycle of the palladium catalyst within a sterically demanding environment. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-halogen bond of the pyrimidine intermediate, a step that is traditionally rate-limiting for electron-deficient and hindered heterocycles. The use of bis(tri-tert-butylphosphine)palladium(0) is critical here, as the bulky ligands create a highly active, electron-rich metal center capable of facilitating this insertion even on the deactivated pyrimidine ring. Following oxidative addition, the coordination and subsequent migratory insertion of the alkene side chain occur, establishing the new carbon-carbon bond with high regioselectivity. The cycle concludes with beta-hydride elimination to regenerate the double bond and reductive elimination to release the product and restore the active palladium(0) catalyst. This catalytic turnover is remarkably efficient, allowing for low catalyst loadings while maintaining high conversion rates, which is essential for minimizing residual metal impurities in the final drug substance.
Impurity control is another critical aspect managed by this mechanistic pathway. Traditional condensation methods often lead to isomeric mixtures or over-alkylation byproducts that are structurally similar to the target molecule, making chromatographic separation difficult and costly. The Heck reaction, being catalytic and highly selective, inherently suppresses many of these side reactions. The specific choice of base, such as N,N-dicyclohexylmethylamine, further tunes the reaction environment to favor the desired coupling over potential homocoupling of the halide or polymerization of the alkene. Moreover, the ability to perform the reaction in aqueous mixtures helps in solubilizing inorganic salts formed during the neutralization of the hydrogen halide byproduct, preventing them from precipitating and fouling the reaction mixture or the catalyst surface. This clean reaction profile translates directly into a simpler impurity spectrum, reducing the burden on analytical quality control and ensuring that the final API meets stringent pharmacopeial standards with less intensive purification efforts.
How to Synthesize Rosuvastatin Intermediate Efficiently
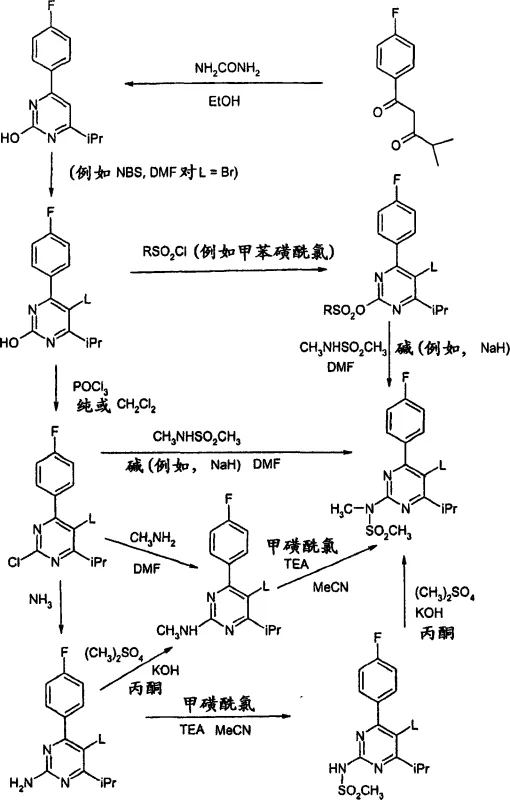
The practical execution of this synthesis involves preparing the pyrimidine halide precursor, typically a 5-bromo or 5-chloro derivative, and reacting it with a protected vinyl side chain under inert atmosphere. The process requires careful control of temperature, typically between 30°C and 110°C, and the use of specific solvent blends like water and DMF to optimize solubility and reaction kinetics. Detailed standard operating procedures for catalyst activation, reagent addition rates, and workup protocols are essential to reproduce the high yields reported in the patent literature. For a comprehensive breakdown of the specific experimental conditions and stoichiometry required to implement this technology safely and effectively, please refer to the standardized synthesis guide below.
- Prepare the pyrimidine halide intermediate (Formula II) where L is a leaving group such as bromine or chlorine, and X is a precursor to the N-methyl-N-methanesulfonylamino group.
- React Formula II with the protected side-chain alkene (Formula III) using a catalytically effective amount of palladium catalyst, specifically bis(tri-tert-butylphosphine)palladium(0).
- Conduct the reaction in a solvent system such as water/DMF mixtures with a base like N,N-dicyclohexylmethylamine at temperatures between 30-110°C, followed by deprotection and salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Heck coupling technology represents a strategic opportunity to optimize the cost structure and reliability of Rosuvastatin sourcing. The shift from stoichiometric phosphorus reagents to a catalytic palladium system fundamentally alters the raw material cost equation, reducing the volume of expensive reagents required per kilogram of product. Additionally, the simplified purification process means less solvent consumption and reduced waste treatment costs, which are significant factors in the total cost of ownership for pharmaceutical intermediates. The robustness of the reaction conditions also implies a more stable supply chain, as the process is less sensitive to minor fluctuations in raw material quality or environmental conditions compared to moisture-sensitive alternatives.
- Cost Reduction in Manufacturing: The transition to a catalytic process eliminates the need for stoichiometric amounts of expensive phosphorus ylides and the associated waste disposal costs for phosphine oxides. By utilizing water-containing solvent systems, the process reduces the dependency on large volumes of pure organic solvents, leading to substantial savings in solvent procurement and recovery operations. Furthermore, the high selectivity of the reaction minimizes the loss of valuable chiral side-chain precursors to byproduct formation, maximizing the yield of the desired intermediate and lowering the effective cost per unit of active ingredient produced.
- Enhanced Supply Chain Reliability: The intermediates generated through this pathway, such as the novel pyrimidine derivatives described in the patent, are chemically stable and can be stockpiled without significant degradation, providing a buffer against supply disruptions. The use of commercially available palladium catalysts and common bases ensures that the supply chain is not dependent on exotic or single-source reagents that could become bottlenecks. This diversification of the supply base enhances resilience, allowing manufacturers to maintain consistent production schedules even when facing market volatility for specific chemical feedstocks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plant to commercial scale without significant re-engineering. The ability to operate in aqueous mixtures aligns with green chemistry principles, reducing the environmental impact and simplifying compliance with increasingly stringent environmental regulations regarding volatile organic compound emissions. This sustainability advantage not only mitigates regulatory risk but also appeals to end-users who are prioritizing environmentally responsible suppliers in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of this supply source. The answers are derived directly from the technical disclosures and experimental data provided within the patent documentation.
Q: What is the key advantage of the Heck reaction method for Rosuvastatin described in CN101084197A?
A: The key advantage is the ability to couple fully substituted, sterically hindered 6-membered pyrimidines with alkenes efficiently, avoiding the harsh conditions and complex purification associated with traditional Wittig-type condensations.
Q: Which palladium catalyst is preferred for this synthesis?
A: The patent specifically highlights bis(tri-tert-butylphosphine)palladium(0) as particularly suitable due to its ability to handle the steric bulk of the pyrimidine substrate effectively.
Q: Can this process be scaled for commercial production?
A: Yes, the process utilizes robust reagents and solvent systems like water/DMF mixtures, which are amenable to large-scale manufacturing and offer improved environmental profiles compared to older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global pharmaceutical market. Our technical team has extensively evaluated the Heck coupling route described in CN101084197A and possesses the expertise to scale this complex chemistry from gram-scale optimization to multi-ton commercial production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. Our facilities are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to monitor every step of the synthesis, guaranteeing that our products meet stringent purity specifications and comply with all relevant international regulatory standards.
We invite you to collaborate with us to leverage this innovative technology for your Rosuvastatin supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing processes can enhance your operational efficiency and reduce your overall production costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
