Optimizing Gefitinib Production: A Novel Purification Strategy for High-Purity API Manufacturing
Optimizing Gefitinib Production: A Novel Purification Strategy for High-Purity API Manufacturing
The pharmaceutical industry constantly seeks robust methodologies to enhance the purity and yield of critical oncology therapeutics, and the recent technological advancements detailed in patent CN111533703B represent a significant leap forward in the manufacturing of Gefitinib. This selective Epidermal Growth Factor Receptor (EGFR) tyrosine kinase inhibitor is a cornerstone treatment for non-small cell lung cancer (NSCLC), necessitating production processes that guarantee exceptional safety profiles and chemical consistency. The disclosed innovation introduces a sophisticated three-stage purification protocol involving salt-forming salting-out, alkali washing, and ethanol recrystallization, which collectively address the longstanding challenges of impurity removal and solvent efficiency. By shifting away from energy-intensive cryogenic crystallization and unstable solvate formations, this process offers a streamlined pathway to achieving purity levels exceeding 99.9 percent. For global supply chain stakeholders, understanding the mechanistic underpinnings of this refinement is crucial for securing a reliable gefitinib supplier capable of meeting stringent regulatory standards. The following analysis dissects the technical merits of this approach, highlighting its potential to redefine cost structures and operational reliability in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Gefitinib has been plagued by processes that are either economically inefficient or technically unstable, creating bottlenecks for large-scale commercial production. For instance, earlier methodologies described in documents like CN103012290A relied heavily on reflux dissolution in ethanol followed by cooling to extreme temperatures of minus 5 degrees Celsius to induce crystallization. While effective to a degree, this approach suffers from excessive ethanol consumption, generating substantial volumes of waste liquid that complicate environmental compliance and increase disposal costs. Furthermore, maintaining such low crystallization temperatures on an industrial scale requires significant energy input and specialized cryogenic equipment, which inherently limits the scalability and throughput of the manufacturing facility. Other attempts, such as those found in CN102584720A, utilized formamide solvent mixtures to create solvates, but these methods often resulted in incomplete crystallization and difficult solvent recovery issues. The instability of formamide solvates and the complexity of the deamidation step in ethyl acetate introduced variability in yield and purity, making these routes unsuitable for the rigorous demands of modern pharmaceutical supply chains where consistency is paramount.
The Novel Approach
In stark contrast to these legacy techniques, the novel purification process outlined in CN111533703B employs a clever sequence of chemical transformations that leverage solubility differences rather than extreme thermal conditions. The core innovation lies in the initial salt-forming salting-out step, where dilute hydrochloric acid and sodium chloride are introduced to the reaction mixture at room temperature. This induces the selective precipitation of Gefitinib hydrochloride, effectively separating the desired product from soluble impurities through a salting-out effect that enhances system saturation without the need for freezing. Following this, a targeted alkali wash with sodium bicarbonate neutralizes the hydrochloride salt back to the free base while simultaneously dissolving residual inorganic salts like sodium chloride. The final stage involves a controlled recrystallization in ethanol with activated carbon decolorization, which polishes the product to ultra-high purity levels. This multi-step refinement not only simplifies the operational workflow but also drastically reduces the reliance on hazardous or difficult-to-recover solvents, presenting a much more viable option for cost reduction in API manufacturing.
Mechanistic Insights into Salt-Forming Salting-Out and Recrystallization
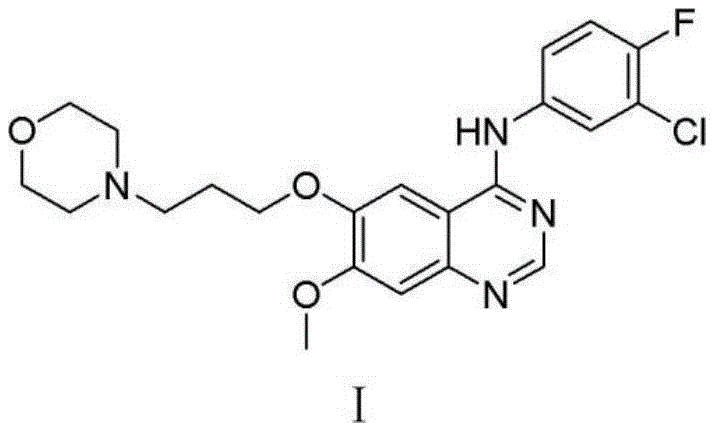
The chemical efficacy of this purification route is rooted in the precise manipulation of ionic equilibria and solubility parameters during the synthesis and isolation phases. The synthesis begins with the nucleophilic substitution reaction between the gefitinib intermediate, specifically 4-(3-chloro-4-fluoroaniline)-7-methoxyquinazoline-6-alcohol, and N-(3-chloropropyl)morpholine. This reaction is catalyzed by potassium carbonate and potassium iodide in N,N-dimethylformamide (DMF) at a moderate temperature of 70 degrees Celsius, facilitating the formation of the ether linkage that characterizes the final drug molecule. 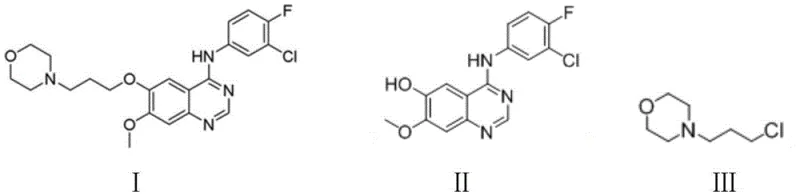 Once the reaction is complete, the addition of dilute hydrochloric acid protonates the quinazoline amine nitrogen, converting the organic base into its water-soluble hydrochloride salt form. However, the subsequent addition of sodium chloride increases the ionic strength of the solution significantly, triggering the common ion effect and salting-out phenomenon. This forces the Gefitinib hydrochloride to precipitate out of the DMF solution while leaving many organic impurities and unreacted starting materials in the supernatant, thereby achieving a primary level of purification before the solid is even isolated.
Once the reaction is complete, the addition of dilute hydrochloric acid protonates the quinazoline amine nitrogen, converting the organic base into its water-soluble hydrochloride salt form. However, the subsequent addition of sodium chloride increases the ionic strength of the solution significantly, triggering the common ion effect and salting-out phenomenon. This forces the Gefitinib hydrochloride to precipitate out of the DMF solution while leaving many organic impurities and unreacted starting materials in the supernatant, thereby achieving a primary level of purification before the solid is even isolated.
Following the isolation of the hydrochloride salt, the process transitions to a neutralization and polishing phase designed to eliminate trace contaminants and ensure crystal perfection. The filter cake containing the hydrochloride salt is treated with a sodium bicarbonate solution, which acts as a mild base to deprotonate the amine, regenerating the free base form of Gefitinib. This step is critical not only for obtaining the correct chemical form of the API but also for washing away the excess sodium chloride and inorganic byproducts generated during the salting-out phase. The final recrystallization from ethanol serves as the ultimate purification barrier; here, the use of activated carbon adsorbs colored impurities and high-molecular-weight byproducts, while the controlled cooling rate of 5 to 10 degrees Celsius per hour ensures the growth of large, well-defined crystals. Slow cooling is mechanistically superior to rapid precipitation because it allows the crystal lattice to form orderly, rejecting impurity molecules that do not fit sterically or electronically, thus resulting in the reported purity of over 99.9 percent.
How to Synthesize Gefitinib Efficiently
Implementing this purification strategy requires strict adherence to the stoichiometric ratios and thermal profiles defined in the patent to maximize yield and quality. The process initiates by combining the quinazoline alcohol intermediate with potassium carbonate and potassium iodide in DMF, followed by the addition of the morpholine derivative at 70 degrees Celsius for a reaction time of approximately 5 to 7 hours. Upon completion, the reaction mixture is treated with dilute hydrochloric acid and sodium chloride to induce crystallization of the hydrochloride salt, which is then filtered and washed. The detailed standardized synthetic steps, including specific molar ratios and washing protocols, are provided in the technical guide below to ensure reproducibility across different manufacturing scales.
- React gefitinib intermediate with N-(3-chloropropyl)morpholine in DMF using potassium carbonate and iodide at 70°C.
- Perform salt-forming salting-out by adding dilute hydrochloric acid and sodium chloride to crystallize the hydrochloride salt.
- Wash the filter cake with sodium bicarbonate solution and recrystallize the free base in ethanol with activated carbon decolorization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology translates directly into enhanced operational resilience and significant economic benefits. The elimination of cryogenic cooling requirements removes a major energy cost center and reduces the dependency on specialized refrigeration infrastructure, which often constitutes a bottleneck in batch processing. Furthermore, the simplified solvent system, which relies primarily on DMF and ethanol rather than complex formamide mixtures or vast quantities of ethyl acetate, streamlines solvent recovery and recycling operations. This reduction in solvent complexity lowers the total cost of ownership for the manufacturing process and minimizes the environmental footprint associated with waste disposal, aligning with increasingly strict global sustainability mandates. By optimizing the crystallization kinetics through salting-out, the process also improves the physical handling characteristics of the intermediate solids, reducing filtration times and improving overall plant throughput.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the substantial reduction in solvent consumption and energy usage compared to traditional low-temperature crystallization methods. By avoiding the need to cool reaction masses to minus 5 degrees Celsius, manufacturers can eliminate the capital and operational expenditures associated with chillers and cryogenic fluids. Additionally, the salting-out technique allows for higher concentrations of reactants, meaning smaller reactor volumes can be used to produce the same amount of product, effectively increasing asset utilization. The use of common, inexpensive reagents like sodium chloride and sodium bicarbonate further drives down the raw material costs, ensuring that the final API is produced with a highly competitive cost basis that supports margin preservation in generic drug markets.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this purification method ensures consistent batch-to-batch quality, which is critical for maintaining regulatory approval and avoiding costly production delays. The process is less sensitive to minor fluctuations in ambient temperature compared to cryogenic methods, making it more reliable for production facilities in diverse geographic locations. Moreover, the reliance on commercially available and stable reagents reduces the risk of supply disruptions caused by the scarcity of specialized solvents or catalysts. This stability allows for more accurate production planning and inventory management, enabling suppliers to meet tight delivery windows and maintain continuous supply lines for downstream formulation partners who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The scalability of this process is evidenced by its straightforward unit operations, which can be easily transferred from pilot plants to multi-ton commercial reactors without complex engineering modifications. The avoidance of formamide solvates eliminates the need for difficult deamidation steps that often pose safety risks and yield losses at large scales. Environmentally, the process generates less hazardous waste due to the efficient recovery of ethanol and the use of aqueous washes that are easier to treat in standard wastewater facilities. This alignment with green chemistry principles not only reduces compliance costs but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for environmentally conscious pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific Gefitinib purification technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear view of how this method outperforms legacy techniques in terms of purity and yield. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does the salt-forming salting-out method improve purity compared to direct crystallization?
A: The process utilizes the solubility difference between the product hydrochloride and impurity hydrochlorides. By adding sodium chloride, the system achieves a salting-out effect that selectively precipitates gefitinib hydrochloride, effectively removing soluble impurities before the final neutralization and recrystallization steps.
Q: What are the critical parameters for the ethanol recrystallization step?
A: Critical parameters include heating the ethanol solution to 65-75°C for complete dissolution, using activated carbon for decolorization, and controlling the cooling rate to 5-10°C/h. Slow cooling is essential to minimize impurity inclusion within the crystal lattice, ensuring the final purity exceeds 99.9%.
Q: Why is this purification process considered more scalable than previous methods?
A: Unlike prior art requiring extreme low temperatures (-5°C) or unstable formamide solvates, this method operates at manageable temperatures (room temp to 70°C) and uses standard solvents like DMF and ethanol. The robust salting-out mechanism ensures consistent crystallization yields without the need for energy-intensive cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gefitinib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven scaling capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of the salt-forming and recrystallization steps are perfectly translated to industrial reactors. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify the absence of genotoxic impurities and residual solvents, guaranteeing that every batch of Gefitinib meets the highest international pharmacopeial standards. Our commitment to quality assurance means that we do not just supply chemicals; we deliver validated solutions that integrate seamlessly into your existing supply chain.
We invite you to engage with our technical procurement team to discuss how this advanced purification process can optimize your specific sourcing requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic impact of switching to this more efficient manufacturing route. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate concretely how our technical prowess can drive value and security for your oncology drug portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
