Advanced Catalytic Synthesis of Camptothecin Analogues for Commercial Scale-Up
Advanced Catalytic Synthesis of Camptothecin Analogues for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of complex anti-tumor agents, particularly camptothecin and its derivatives which serve as critical precursors for topoisomerase I inhibitors. Patent CN101824038B introduces a groundbreaking synthetic strategy that addresses long-standing inefficiencies in the construction of the pentacyclic quinoline skeleton inherent to these bioactive molecules. This proprietary technology leverages a synergistic combination of palladium on carbon (Pd/C) catalytic oxidation and molecular iodine-mediated cyclization to achieve superior yields and purity profiles. By shifting away from stoichiometric metal oxidants and harsh acidic conditions, this route offers a streamlined pathway that is exceptionally well-suited for the demands of a reliable camptothecin intermediate supplier aiming to optimize their manufacturing footprint. The structural versatility of the final product, denoted as Formula (I), allows for various substitutions at the R1 and R2 positions, facilitating the rapid generation of diverse analogue libraries for drug discovery programs.
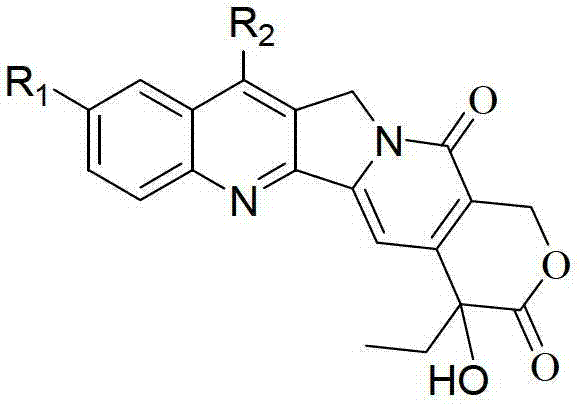
The core innovation lies in the precise control of oxidation states and protecting group manipulations, ensuring that the sensitive lactone ring and the E-ring stereochemistry are preserved throughout the synthesis. This technical advancement not only enhances the chemical efficiency but also aligns with modern green chemistry principles by minimizing three-waste pollution. For procurement and supply chain stakeholders, the implications are profound, as the ability to recycle the heterogeneous Pd/C catalyst multiple times directly translates to reduced raw material consumption and lower operational expenditures. Furthermore, the mild reaction temperatures ranging from 20°C to 90°C across the different stages mitigate thermal degradation risks, thereby ensuring consistent batch-to-batch quality essential for regulatory compliance in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the crucial ABC ring system of camptothecin has been plagued by significant technical hurdles that impede efficient commercial scale-up. Traditional protocols often relied on stoichiometric amounts of metal Lewis acids such as cupric chloride or neutralized verdigris, which generated substantial quantities of heavy metal waste that required complex and costly disposal procedures. Additionally, earlier methods utilizing Raney nickel or copper-chromium alloys for oxidation steps were characterized by severe reaction conditions and cumbersome work-up procedures that negatively impacted overall throughput. The subsequent formation of the quinoline ring via Friedlander condensation was typically catalyzed by p-toluenesulfonic acid in toluene/ethanol systems, necessitating prolonged azeotropic water removal for 5 to 10 hours, which frequently resulted in low reaction yields and poor product purity. Alternative approaches employing trimethylsilyl chloride, while improving yields slightly, suffered from the excessive consumption of reagents, often requiring 4 to 5 equivalents relative to the substrate, rendering the process economically unviable for large-scale production.
The Novel Approach
In stark contrast, the methodology disclosed in CN101824038B presents a paradigm shift by utilizing catalytic amounts of reagents under remarkably mild conditions to drive the transformation forward. The novel route initiates with a Pd/C catalyzed oxidation of the tetrahydropyrano-indolizine precursor (Compound II) using molecular oxygen, eliminating the need for toxic stoichiometric oxidants. This is followed by a highly efficient ketal deprotection step mediated by catalytic iodine in acetone, which proceeds smoothly at moderate temperatures of 50°C to 60°C. The final cyclization employs the same iodine catalyst to facilitate the condensation with substituted o-aminobenzaldehydes in polar aprotic solvents like DMF or DMAc, achieving completion within 6 to 10 hours without the need for complex water removal setups. This integrated approach not only simplifies the operational workflow but also drastically improves the atom economy of the synthesis, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
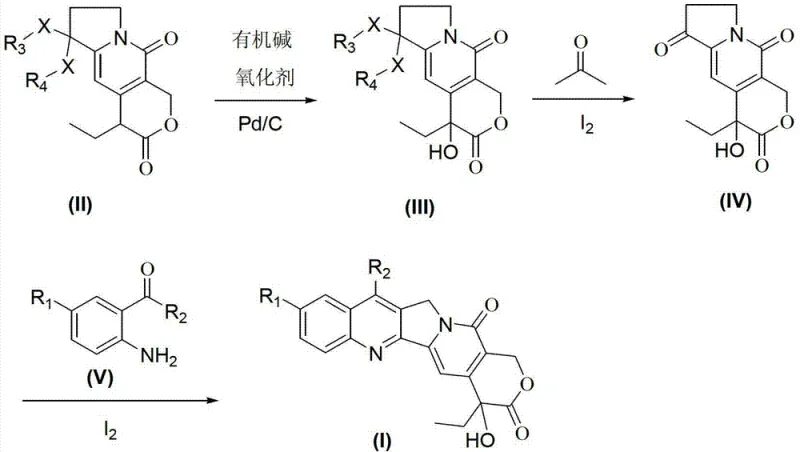
Mechanistic Insights into Pd/C-Catalyzed Oxidation and Iodine-Mediated Cyclization
The mechanistic elegance of this synthesis begins with the heterogeneous catalytic oxidation of the indolizine derivative, where the Pd/C surface facilitates the activation of molecular oxygen to selectively oxidize the allylic or benzylic positions without over-oxidizing sensitive functional groups. The presence of a catalytic amount of organic base, such as triethylamine, plays a crucial role in neutralizing acidic byproducts and maintaining the optimal pH environment for the catalyst to function efficiently at temperatures between 20°C and 30°C. Kinetic studies suggest that the reaction follows a zero-order dependence on the substrate at higher concentrations, indicating that the surface adsorption of oxygen is the rate-determining step, which can be optimized by controlling the oxygen flow rate and stirring speed. Importantly, the heterogeneous nature of the Pd/C catalyst allows for easy filtration and recovery, with experimental data confirming that the catalyst retains its activity for at least three consecutive cycles, thereby minimizing the leaching of palladium into the product stream and ensuring high purity specifications for the downstream intermediates.
Following the oxidation, the iodine-catalyzed steps demonstrate a unique dual functionality that streamlines the protection and cyclization logic. In the deprotection phase, molecular iodine acts as a soft Lewis acid to coordinate with the acetal oxygen atoms, facilitating the cleavage of the ketal group to reveal the reactive ketone functionality required for the subsequent condensation. In the final Friedlander condensation, iodine continues to serve as an effective catalyst by activating the carbonyl group of the aldehyde and enhancing the nucleophilicity of the amine, promoting the formation of the imine intermediate and subsequent cyclization to form the quinoline ring. This mechanism avoids the harsh acidic conditions associated with traditional Brønsted acid catalysts, which often lead to the hydrolysis of the sensitive lactone ring or polymerization of the reactants. The use of polar aprotic solvents further stabilizes the transition states, allowing the reaction to proceed at 80°C to 90°C with high regioselectivity and minimal formation of side products, thus ensuring a clean impurity profile.
How to Synthesize Camptothecin Analogues Efficiently
The execution of this synthetic route requires careful attention to reaction parameters to maximize the benefits of the catalytic systems described. The process is divided into three distinct stages: the initial aerobic oxidation, the deprotection of the ketal moiety, and the final condensation to close the quinoline ring. Each step has been optimized to balance reaction rate with selectivity, ensuring that the intermediate compounds are generated with high purity before proceeding to the next stage. Operators should note that the choice of solvent and the precise molar ratios of the catalysts are critical variables that influence the overall yield and quality of the final camptothecin analogue. For a detailed breakdown of the specific operational parameters, including solvent volumes, temperature ramps, and work-up procedures, please refer to the standardized synthesis guide below.
- Oxidize the starting indolizine derivative using Pd/C catalyst and oxygen in the presence of an organic base like triethylamine at 20-30°C.
- Perform ketal deprotection on the oxidized intermediate using catalytic molecular iodine in acetone at 50-60°C to generate the trioxo compound.
- Execute the final Friedlander condensation with substituted o-aminobenzaldehyde using iodine catalysis in DMF or DMAc at 80-90°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this catalytic methodology offers substantial benefits that extend beyond mere chemical yield improvements. The elimination of stoichiometric heavy metal oxidants removes a significant bottleneck in waste management and regulatory compliance, thereby reducing the environmental burden and associated disposal costs for manufacturing facilities. The ability to recycle the Pd/C catalyst multiple times without regeneration represents a direct reduction in the cost of goods sold (COGS), as precious metal consumption is a major cost driver in fine chemical synthesis. Furthermore, the mild reaction conditions reduce the energy load on the production plant, as there is no need for extreme heating or cooling, contributing to a lower carbon footprint and enhanced sustainability metrics which are increasingly important for global supply chains.
- Cost Reduction in Manufacturing: The transition from stoichiometric metal reagents to catalytic systems fundamentally alters the cost structure of the synthesis by minimizing raw material expenses. By utilizing molecular oxygen as the terminal oxidant and recyclable Pd/C, the process avoids the procurement of expensive and hazardous chemical oxidants. Additionally, the high yields achieved in each step, often exceeding 80%, mean that less starting material is required to produce the same amount of final product, effectively amplifying the throughput of existing reactor capacity. The simplified work-up procedures, which involve basic filtration and washing rather than complex chromatographic separations, further reduce labor and solvent costs, leading to significant overall economic efficiency.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more stable and predictable supply of high-purity camptothecin intermediates. The use of readily available and stable reagents such as iodine and triethylamine mitigates the risk of supply disruptions often associated with specialized or hazardous chemicals. Moreover, the tolerance of the reaction to minor variations in conditions enhances process reliability, reducing the likelihood of batch failures that can delay deliveries to downstream API manufacturers. This stability is crucial for maintaining continuous production schedules and meeting the stringent delivery timelines required by multinational pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The absence of exothermic runaway risks associated with strong oxidants and the use of standard solvents like ethanol and acetone simplify the engineering controls required for large-scale reactors. Environmentally, the reduction in heavy metal waste and the use of greener oxidation methods align with strict international environmental regulations, facilitating easier permitting and operation in regulated jurisdictions. This compliance advantage future-proofs the supply chain against tightening environmental laws, ensuring long-term viability of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of adopting this technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their current manufacturing workflows.
Q: What are the key advantages of the Pd/C catalytic system in this synthesis?
A: The Pd/C catalyst allows for mild oxidation conditions (20-30°C) and can be recycled at least three times without loss of activity, significantly reducing heavy metal waste and catalyst costs compared to stoichiometric metal oxidants.
Q: How does the iodine-catalyzed Friedlander condensation improve yield?
A: Unlike traditional p-TsOH methods requiring azeotropic water removal which often result in low yields, the iodine-catalyzed method in polar aprotic solvents like DMF achieves yields of 70-80% with higher product purity under milder thermal conditions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process features simple operations, avoids hazardous stoichiometric reagents, and utilizes recyclable catalysts, making it highly scalable and environmentally compliant for commercial manufacturing of anti-tumor intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Camptothecin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of life-saving oncology therapies. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and robust. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging advanced catalytic technologies like the one described in CN101824038B, we can offer our partners a competitive edge through superior quality and consistent supply continuity.
We invite you to collaborate with us to explore how this innovative synthesis can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can support your next breakthrough in cancer treatment development.
