Advanced Catalytic Strategies For Commercial Vitamin K3 Production And Scalability
Advanced Catalytic Strategies For Commercial Vitamin K3 Production And Scalability
The pharmaceutical and fine chemical industries are constantly seeking more efficient and environmentally sustainable pathways for the production of essential vitamins and intermediates. Patent CN115805096A introduces a groundbreaking methodology for the synthesis of Vitamin K3, also known as menadione, utilizing a heterogeneous nitrogen-doped carbon material-supported cobalt catalyst. This innovation addresses critical pain points in traditional manufacturing, such as the reliance on toxic heavy metals and the difficulty in catalyst recovery. By leveraging a novel oxidative dehydrogenation process, this technology offers a robust solution for producing high-purity menadione with significantly reduced environmental impact. The strategic implementation of this catalytic system allows for a streamlined production workflow that aligns with modern green chemistry principles while maintaining the rigorous quality standards required for pharmaceutical applications. This report analyzes the technical merits and commercial implications of adopting this advanced synthetic route for large-scale vitamin manufacturing.
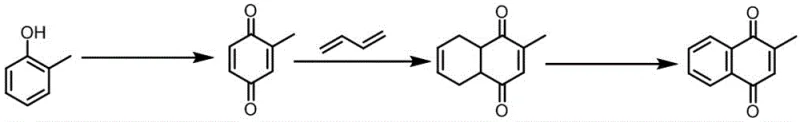
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Vitamin K3 has relied heavily on oxidation processes involving chromium-based reagents, such as chromium trioxide or sodium dichromate, in acidic media. While effective in terms of conversion, these traditional methods generate substantial quantities of hazardous waste containing hexavalent chromium, a known carcinogen that poses severe environmental and regulatory challenges. The disposal of such toxic byproducts requires complex and costly treatment protocols, which significantly inflate the overall operational expenditure for manufacturers. Furthermore, alternative laboratory-scale methods utilizing hydrogen peroxide and expensive transition metals like rhenium or vanadium often suffer from low yields and selectivity issues, making them economically unviable for commercial scale-up. The reliance on homogeneous catalysts in some existing routes also complicates the downstream processing, as separating the catalyst from the final product often requires energy-intensive purification steps that can degrade product quality. These inherent inefficiencies create a pressing need for a more sustainable and cost-effective catalytic system that can deliver high purity without the associated environmental liabilities.
The Novel Approach
The innovative process disclosed in the patent circumvents these traditional bottlenecks by employing a heterogeneous nitrogen-doped carbon material-supported cobalt catalyst, designated as Co@CN. This catalyst is synthesized through a controlled sintering process involving glucosamine hydrochloride, melamine, and cobalt acetate, resulting in a highly dispersed active phase that is robust and reusable. Unlike the toxic chromium routes, this method utilizes molecular oxygen or air as the terminal oxidant, producing water as the primary byproduct and thereby drastically reducing the chemical oxygen demand of the waste stream. The heterogeneous nature of the catalyst facilitates straightforward separation via filtration, allowing for multiple reuse cycles without significant loss of activity, which is a critical factor for long-term process economics. Additionally, the reaction conditions are milder, often operating at temperatures between 0°C and 40°C, which reduces energy consumption and enhances safety profiles compared to high-temperature oxidation processes. This novel approach represents a paradigm shift towards greener manufacturing, offering a viable pathway for cost reduction in vitamin K3 manufacturing while ensuring compliance with increasingly stringent environmental regulations.
Mechanistic Insights into Co@CN-Catalyzed Oxidative Dehydrogenation
The core of this technological advancement lies in the unique mechanistic pathway enabled by the Co@CN catalyst during the oxidative dehydrogenation of 2-methyl-1,4-tetrahydronaphthoquinone. The nitrogen-doped carbon support plays a pivotal role in stabilizing the cobalt active sites, preventing agglomeration and ensuring high surface area availability for the substrate. During the reaction, the cobalt species facilitate the activation of molecular oxygen, generating reactive oxygen species that selectively abstract hydrogen atoms from the tetrahydronaphthoquinone ring system. This selective dehydrogenation is crucial for avoiding over-oxidation or ring-opening side reactions that typically plague less selective catalytic systems. The synergy between the cobalt centers and the nitrogen functionalities in the carbon matrix enhances the electron transfer efficiency, leading to high conversion rates and exceptional selectivity towards the desired naphthoquinone structure. Understanding this mechanism is vital for R&D directors aiming to optimize reaction parameters such as solvent choice and oxygen pressure to maximize throughput and minimize impurity formation in a commercial setting.
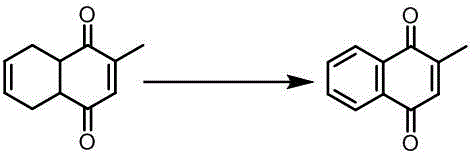
Furthermore, the upstream synthesis of the precursor, 2-methyl-1,4-tetrahydronaphthoquinone, is also optimized in this patent through a metal-chelating ionic liquid catalyzed Diels-Alder reaction and a TEMPO-mediated oxidation of o-cresol. The use of a specific metal-chelating ionic liquid allows for the efficient cycloaddition of o-methylbenzoquinone and 1,3-butadiene under mild conditions, avoiding the high pressures and safety hazards associated with traditional methods. Complementing this, the initial oxidation of o-cresol to o-methylbenzoquinone utilizes a metal-free TEMPO catalytic system with nitrite and protic acid co-catalysts, eliminating the need for transition metals in the first step entirely. This multi-step cascade ensures that the impurity profile of the final intermediate is tightly controlled before it even reaches the final dehydrogenation stage. The cumulative effect of these mechanistic refinements is a process that delivers high-purity menadione with a significantly reduced burden on downstream purification units, thereby enhancing the overall process mass intensity and sustainability metrics of the production line.
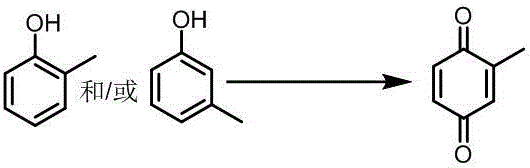
How to Synthesize Vitamin K3 Efficiently
Implementing this synthetic route requires precise adherence to the catalyst preparation and reaction conditions outlined in the patent to ensure reproducibility and optimal performance. The preparation of the Co@CN catalyst involves a specific ratio of glucosamine hydrochloride to melamine and cobalt acetate, followed by a two-stage sintering process under inert gas to establish the correct crystal structure and nitrogen doping levels. Once the catalyst is prepared, the oxidative dehydrogenation reaction is conducted in an alcohol solvent, such as methanol or ethanol, with the continuous introduction of oxygen or air at controlled pressures. The reaction mixture is stirred at ambient or slightly elevated temperatures to facilitate the conversion, after which the solid catalyst is recovered by filtration for regeneration and reuse. The detailed standardized synthesis steps see the guide below.
- Prepare the heterogeneous nitrogen-doped carbon material-supported cobalt catalyst by sintering glucosamine hydrochloride, melamine, and cobalt acetate under inert atmosphere.
- Conduct the oxidative dehydrogenation of 2-methyl-1,4-tetrahydronaphthoquinone using the prepared catalyst and oxygen as the oxidant in an alcohol solvent.
- Recover the catalyst via filtration and purify the crude Vitamin K3 product through distillation to achieve pharmaceutical grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology translates into tangible strategic benefits that extend beyond mere technical performance. The elimination of hexavalent chromium and expensive noble metals like palladium from the process chain directly correlates to a substantial reduction in raw material costs and waste disposal fees. By utilizing earth-abundant cobalt and organic precursors for the catalyst, the supply chain becomes less vulnerable to the price volatility associated with critical raw materials, ensuring more stable long-term pricing for the final product. The ease of catalyst recovery and reuse further amplifies these savings, as the effective catalyst consumption per kilogram of product is drastically lowered compared to single-use homogeneous systems. This efficiency gain allows for a more competitive cost structure, enabling suppliers to offer better pricing tiers without compromising on margin, which is a key consideration for large-scale procurement contracts in the pharmaceutical sector.
- Cost Reduction in Manufacturing: The transition to a heterogeneous cobalt-based catalyst system eliminates the need for costly heavy metal removal steps and reduces the consumption of expensive oxidants. By avoiding the use of chromium and palladium, manufacturers can bypass the significant regulatory and disposal costs associated with hazardous waste management. The ability to recycle the catalyst multiple times without significant loss of activity means that the catalyst cost per batch is amortized over a much larger production volume. This structural change in the cost base allows for significant cost savings in the overall manufacturing process, making the final Vitamin K3 product more price-competitive in the global market while maintaining high profitability.
- Enhanced Supply Chain Reliability: The reliance on readily available precursors such as glucosamine, melamine, and cobalt acetate ensures a robust and resilient supply chain that is not dependent on scarce or geopolitically sensitive materials. The simplified process flow, which operates at mild temperatures and pressures, reduces the risk of unplanned shutdowns due to equipment failure or safety incidents. Furthermore, the high stability of the catalyst ensures consistent production output, minimizing the variability in lead times that often plagues complex chemical syntheses. This reliability is crucial for supply chain heads who need to guarantee continuous availability of high-purity intermediates to downstream pharmaceutical customers, thereby strengthening the overall partnership and trust between supplier and buyer.
- Scalability and Environmental Compliance: The heterogeneous nature of the catalyst and the use of green oxidants like oxygen make this process inherently scalable from laboratory to industrial production without the need for major re-engineering. The reduction in toxic waste generation aligns perfectly with global environmental regulations, reducing the risk of compliance violations and associated fines. The process generates minimal hazardous byproducts, simplifying the environmental permitting process for new production facilities or the expansion of existing ones. This environmental compatibility not only future-proofs the manufacturing operation against tightening regulations but also enhances the brand reputation of the supplier as a responsible and sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Vitamin K3 synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure. The answers reflect the specific advantages of the Co@CN catalyst system and the associated process conditions.
Q: What are the advantages of the Co@CN catalyst over traditional chromium-based methods?
A: The Co@CN catalyst eliminates the use of toxic hexavalent chromium, significantly reducing environmental hazards and waste treatment costs while maintaining high catalytic efficiency and recyclability.
Q: Can the catalyst be reused in industrial scale production?
A: Yes, the heterogeneous nature of the Co@CN catalyst allows for easy recovery via filtration. Experimental data shows minimal loss in activity after multiple cycles, ensuring process stability.
Q: What is the purity level achievable with this new synthetic route?
A: The process described in the patent achieves product purity exceeding 98%, with specific examples demonstrating purity levels up to 99.3% after standard purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin K3 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the global pharmaceutical market. Our expertise as a CDMO partner allows us to leverage innovations like the Co@CN catalytic system to deliver Vitamin K3 with unparalleled quality and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless and efficient. Our rigorous QC labs and stringent purity specifications guarantee that every batch of menadione meets the highest international standards, providing our clients with the confidence they need to proceed with their own drug development and manufacturing programs. We are committed to being a strategic partner who not only supplies chemicals but also provides technical solutions that enhance your overall product lifecycle.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic advantages of switching to this greener manufacturing process. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs. Our team is ready to collaborate with you to engineer a supply solution that balances cost, quality, and sustainability, ensuring that your production of Vitamin K3 remains competitive and compliant in a rapidly changing regulatory landscape.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
