Revolutionizing Pharmaceutical Intermediate Manufacturing with Scalable Metal-Free Triazole Synthesis Technology
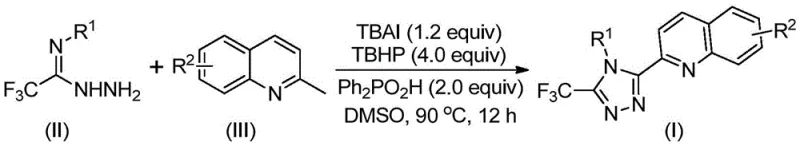
The recently granted Chinese patent CN113307790B introduces a groundbreaking synthetic route for producing structurally diverse 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This innovative methodology addresses critical limitations in existing production techniques by eliminating the need for transition metal catalysts while operating under ambient atmospheric conditions without requiring anhydrous environments. The process leverages commercially accessible starting materials including tetrabutylammonium iodide and tert-butyl peroxide to facilitate an oxidative cyclization reaction that achieves superior yields compared to conventional multi-step approaches. Crucially, this patent provides a versatile platform for synthesizing variously substituted triazole derivatives through strategic modification of aryl groups on both reaction components, thereby expanding the structural diversity available for drug discovery programs. The methodology's operational simplicity and compatibility with standard laboratory equipment make it particularly attractive for implementation across global pharmaceutical manufacturing facilities seeking reliable intermediate supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for quinolyl-substituted triazoles have historically relied on quinoline-2-formic acid as the primary building block, necessitating a complex five-step reaction sequence that operates under severe reaction conditions including high temperatures and extended reaction times. This conventional approach suffers from a critically low overall yield of only seventeen percent due to multiple purification challenges and intermediate instability throughout the synthetic pathway. The requirement for specialized anhydrous and oxygen-free environments significantly increases operational complexity while demanding substantial capital investment in specialized equipment and stringent process controls. Furthermore, these methods often incorporate transition metal catalysts that introduce persistent contamination risks requiring extensive post-reaction purification steps to meet pharmaceutical quality standards. The cumulative effect of these limitations renders traditional approaches economically unviable for large-scale commercial production despite the growing demand for quinoline-containing triazole scaffolds in modern drug development pipelines.
The Novel Approach
The patented methodology represents a paradigm shift by utilizing readily available 2-methylquinoline and trifluoroethylimine hydrazide as starting materials in a single-step oxidative cyclization process that operates efficiently under ambient atmospheric conditions without requiring anhydrous environments. This innovative approach eliminates all transition metal catalysts entirely while achieving significantly higher yields across diverse substrate combinations as demonstrated in examples one through fifteen of the patent documentation. The reaction proceeds through a well-defined mechanism involving in situ generation of quinoline aldehyde intermediates followed by condensation and oxidative cyclization steps that can be readily scaled from laboratory to manufacturing volumes. Crucially, the process maintains exceptional functional group tolerance across various aryl substitutions while producing high-purity products suitable for direct use in pharmaceutical applications without extensive additional purification requirements.
Mechanistic Insights into Oxidative Cyclization with TBAI/TBHP
The reaction mechanism begins with tetrabutylammonium iodide-mediated oxidation of the methyl group on the quinoline ring to form a reactive aldehyde intermediate under the influence of tert-butyl peroxide as the oxidizing agent. This aldehyde then undergoes condensation with trifluoroethylimine hydrazide to form a hydrazone intermediate through nucleophilic addition followed by dehydration. Subsequent oxidative iodination generates an electrophilic species that undergoes intramolecular electrophilic substitution at the quinoline ring position adjacent to nitrogen, forming the triazole core structure through cyclization and aromatization steps. The diphenyl phosphoric acid additive plays a critical role in facilitating proton transfer during the cyclization phase while stabilizing key intermediates throughout the reaction sequence. This mechanism operates efficiently under mild thermal conditions between eighty and one hundred degrees Celsius without requiring inert atmosphere protection due to the stability of the radical intermediates involved.
Impurity control is achieved through multiple built-in mechanisms within this synthetic pathway that prevent common side reactions observed in traditional triazole syntheses. The absence of transition metals eliminates metal-derived impurities that typically require complex chelation or chromatographic removal processes in conventional manufacturing. The reaction's inherent chemoselectivity minimizes formation of regioisomers due to the specific positioning requirements for electrophilic substitution on the quinoline ring system. Careful optimization of reagent stoichiometry—particularly the precise molar ratio of tetrabutylammonium iodide to tert-butyl peroxide to diphenyl phosphoric acid at one point two to four to two—ensures complete conversion while suppressing overoxidation byproducts. The final purification step using standard column chromatography effectively removes any residual starting materials or minor side products, consistently delivering products meeting pharmaceutical-grade purity specifications as confirmed by comprehensive NMR and HRMS characterization data.
How to Synthesize Quinolyl Trifluoromethyl Triazoles Efficiently
This patented synthetic route represents a significant advancement in the production of quinoline-containing triazole compounds by providing a streamlined alternative to traditional multi-step methodologies that have plagued pharmaceutical manufacturers with low yields and complex processing requirements. The methodology leverages commercially available reagents under practical operating conditions that can be readily implemented in standard manufacturing facilities without requiring specialized equipment or environmental controls typically associated with air-sensitive chemistry. Detailed analysis of the reaction parameters reveals optimal conditions that maximize both yield and purity while maintaining operational simplicity across diverse substrate combinations. The following standardized synthesis protocol provides step-by-step guidance for successful implementation of this innovative manufacturing approach in industrial settings.
- Combine tetrabutylammonium iodide (1.2 equiv), tert-butyl peroxide aqueous solution (4.0 equiv), diphenyl phosphoric acid (2.0 equiv), trifluoroethylimine hydrazide (II), and 2-methylquinoline (III) in DMSO solvent within a Schlenk tube.
- Heat the reaction mixture to 90°C under ambient atmosphere and stir continuously for twelve hours to complete the oxidative cyclization process.
- Perform post-treatment by filtration through silica gel followed by column chromatography purification to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional production methods for complex heterocyclic intermediates required in pharmaceutical development programs. The elimination of specialized environmental controls significantly reduces facility qualification requirements while enhancing operational flexibility across global manufacturing networks. The use of commercially available starting materials creates multiple sourcing options that mitigate supply chain disruption risks commonly encountered with specialized reagents or catalysts required in conventional syntheses. These advantages collectively strengthen procurement strategies by providing greater resilience against market volatility while ensuring consistent access to high-quality intermediates essential for drug development timelines.
- Cost Reduction in Manufacturing: The complete elimination of transition metal catalysts removes substantial expenses associated with catalyst procurement, handling, and subsequent removal processes required to meet regulatory standards for elemental impurities in pharmaceutical products; this simplification also reduces solvent consumption during purification stages while minimizing waste treatment costs through fewer processing steps compared to conventional multi-step syntheses.
- Enhanced Supply Chain Reliability: Utilization of widely available starting materials including tetrabutylammonium iodide and tert-butyl peroxide creates multiple sourcing options that significantly reduce dependency on single suppliers; the ambient condition operation eliminates specialized storage requirements for moisture-sensitive reagents while enabling flexible production scheduling across different geographic locations without environmental control investments.
- Scalability and Environmental Compliance: The reaction's demonstrated scalability from gram-scale laboratory experiments to potential commercial production volumes ensures seamless technology transfer without reoptimization; the absence of heavy metals simplifies waste stream management while meeting increasingly stringent environmental regulations governing industrial chemical processes across global manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN113307790B's experimental data and implementation parameters; these insights have been compiled specifically to support procurement decisions and supply chain planning for pharmaceutical intermediate acquisition.
Q: How does this metal-free synthesis overcome the limitations of traditional quinolyl triazole production methods?
A: The conventional five-step process using quinoline-2-formic acid operates under severe conditions with only 17% overall yield, whereas this patent eliminates anhydrous/anaerobic requirements and toxic heavy metal catalysts through a single-step oxidative cyclization using tetrabutylammonium iodide and tert-butyl peroxide.
Q: What supply chain advantages does the elimination of heavy metal catalysts provide for pharmaceutical manufacturing?
A: By avoiding transition metal catalysts entirely, this method removes costly purification steps for metal residue removal while ensuring regulatory compliance with stringent ICH Q3D guidelines for elemental impurities in active pharmaceutical ingredients.
Q: Can this synthesis maintain high purity during commercial scale-up from laboratory to manufacturing volumes?
A: The process demonstrates consistent high purity across gram-scale to pilot-scale reactions as evidenced by NMR and HRMS data in examples 1 through 15, with the absence of metal catalysts preventing common scale-up challenges associated with catalyst deactivation or heterogeneous mixing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolyl Trifluoromethyl Triazole Supplier
Our patented technology represents a significant advancement in heterocyclic compound manufacturing that directly addresses critical challenges in pharmaceutical intermediate production through innovative green chemistry principles; NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation for comprehensive quality verification. This capability ensures seamless transition from laboratory development to full-scale manufacturing without compromising product quality or regulatory compliance requirements essential for global pharmaceutical supply chains.
We invite you to request our Customized Cost-Saving Analysis tailored to your specific production needs; our technical procurement team stands ready to provide detailed COA data and route feasibility assessments upon inquiry to demonstrate how our innovative synthesis platform can optimize your intermediate supply strategy while ensuring reliable access to high-purity quinolyl trifluoromethyl triazoles.
