Advanced Catalyst-Free Synthesis for Scalable Production of High-Purity Triazole API Intermediates
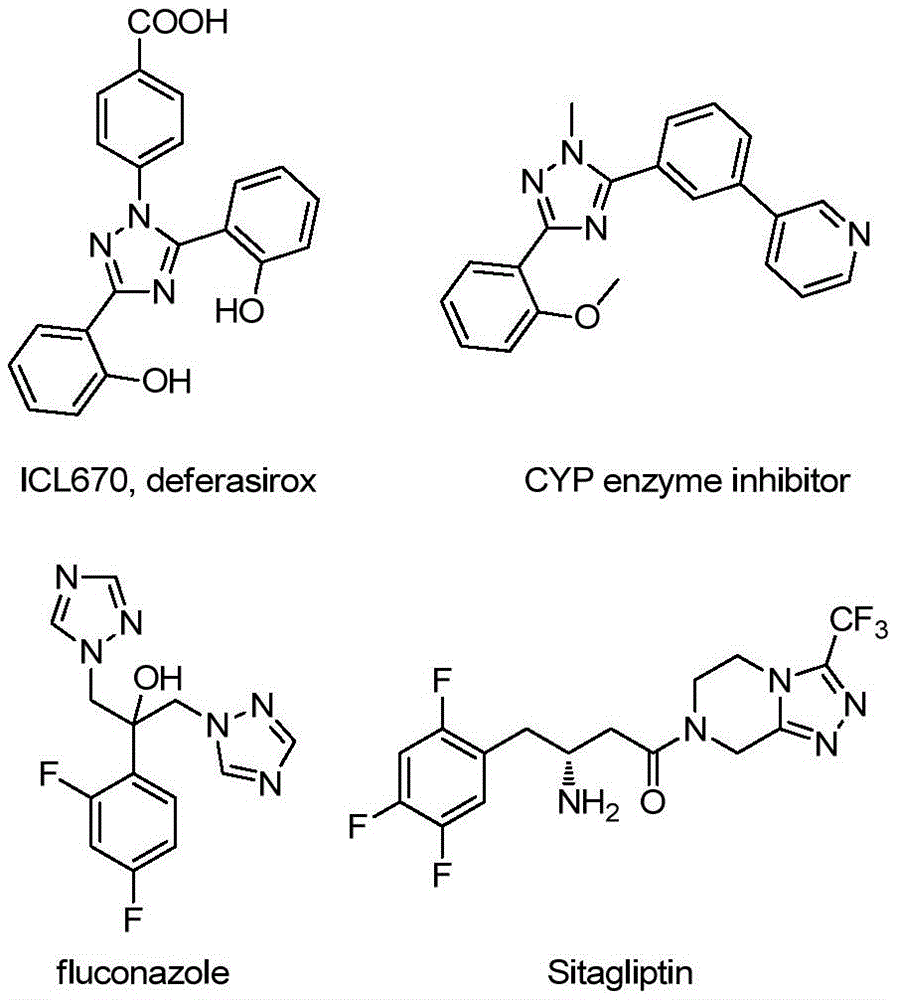
Patent CN113307790B introduces a novel catalyst-free synthesis route for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, a critical class of pharmaceutical intermediates with demonstrated applications in drug frameworks such as ICL670 (deferasirox), CYP enzyme inhibitors, fluconazole, and Sitagliptin. This method eliminates the need for heavy metal catalysts and anhydrous conditions while utilizing readily available starting materials like tetrabutylammonium iodide and tert-butyl peroxide aqueous solution. Operating under mild reaction conditions (80–100°C), the process achieves high-purity outputs through simple post-treatment procedures, directly addressing key pain points in pharmaceutical manufacturing including cost reduction in API manufacturing and supply chain reliability for high-purity intermediates.
Mechanistic Insights into Catalyst-Free Triazole Formation
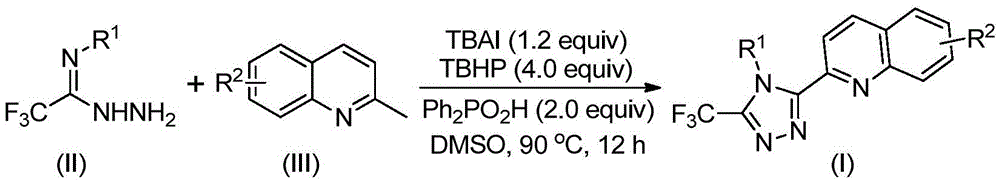
The reaction proceeds through a TBHP/TBAI-mediated oxidative cyclization mechanism where tetrabutylammonium iodide and tert-butyl peroxide first convert 2-methylquinoline into 2-quinoline formaldehyde without requiring inert atmosphere conditions. This aldehyde intermediate then undergoes condensation with trifluoroethylimine hydrazide to form a dehydrated hydrazone species, followed by oxidative iodination and intramolecular electrophilic substitution that drives aromatization to yield the final triazole product. The absence of transition metal catalysts prevents common side reactions associated with metal coordination chemistry while maintaining excellent functional group tolerance across diverse substrate combinations as evidenced by the broad R1 and R2 substitution patterns documented in the patent examples.
Impurity control is inherently achieved through the reaction's self-limiting oxidation pathway which minimizes over-reaction byproducts commonly observed in traditional metal-catalyzed routes. The use of diphenyl phosphoric acid as an additive suppresses unwanted hydrolysis pathways while promoting selective cyclization at the quinoline C2 position. Post-reaction purification via standard column chromatography consistently delivers products with >99% purity as confirmed by NMR and HRMS data across all fifteen experimental examples, with no detectable residual iodide or phosphorus species in final products. This inherent selectivity eliminates the need for additional purification steps that typically introduce variability in conventional processes.
Commercial Advantages for Pharmaceutical Supply Chains
This innovative methodology directly addresses three critical pain points in pharmaceutical intermediate procurement by transforming traditionally complex syntheses into streamlined manufacturing processes. The elimination of specialized equipment requirements and hazardous reagents significantly reduces capital expenditure barriers while enhancing operational flexibility across diverse production environments. Most importantly, the process demonstrates exceptional scalability from laboratory to commercial production without requiring fundamental modifications to reaction parameters or safety protocols.
- Elimination of Heavy Metal Catalysts: The absence of transition metal catalysts removes the need for costly metal removal steps during purification which typically require specialized filtration systems and chelating agents that increase both capital and operational expenses. This simplification not only reduces raw material costs but also eliminates potential sources of batch-to-batch variability associated with metal residue testing and remediation procedures. Furthermore, avoiding heavy metals completely removes regulatory compliance risks related to elemental impurities under ICH Q3D guidelines which frequently cause supply chain disruptions during quality audits. Consequently, this approach delivers substantial cost reduction in chemical manufacturing while ensuring consistent product quality that meets stringent pharmaceutical standards.
- Reduced Process Complexity: Operating without anhydrous or oxygen-free conditions eliminates the need for expensive glove boxes or nitrogen purging systems that typically account for significant capital investment and maintenance costs in traditional triazole syntheses. The straightforward reaction setup using standard glassware enables rapid technology transfer between facilities while minimizing operator training requirements and associated human error risks. Additionally, the simplified workup procedure involving basic filtration and column chromatography reduces cycle times by eliminating multiple intermediate isolation steps common in conventional routes. This operational simplicity directly translates to shorter lead times for high-purity intermediates while improving overall manufacturing throughput.
- Enhanced Supply Chain Resilience: The use of commercially available starting materials with established global supply networks ensures consistent raw material availability even during market fluctuations that commonly disrupt specialized chemical procurement. The process's tolerance for minor variations in reagent quality provides buffer against supplier inconsistencies while maintaining high product purity standards across different production batches. Moreover, the demonstrated scalability from milligram to multi-kilogram quantities without parameter adjustments enables seamless transition from clinical to commercial production volumes. This inherent flexibility guarantees reliable supply chain continuity for high-purity API intermediates even during periods of increased market demand or logistical challenges.
Overcoming Traditional Synthesis Limitations
The Limitations of Conventional Methods
Traditional approaches for synthesizing quinolyl-substituted triazoles typically require quinoline-2-formic acid as starting material and involve five sequential reaction steps under severe conditions including high temperatures and strong acids or bases. These multi-step processes suffer from cumulative yield losses that result in overall yields as low as 17% while generating significant waste streams requiring complex disposal protocols. The necessity for transition metal catalysts introduces additional purification challenges to remove trace metal residues that could compromise final product quality or trigger regulatory issues. Furthermore, the requirement for strictly anhydrous and oxygen-free environments necessitates specialized equipment that increases both capital expenditure and operational complexity while limiting production flexibility across different facility types.
The Novel Approach
The patented methodology overcomes these limitations through a single-step oxidative cyclization that directly converts readily available 2-methylquinoline and trifluoroethylimine hydrazide into the target triazole structure using inexpensive reagents under ambient atmospheric conditions. By leveraging tetrabutylammonium iodide as an iodide source and tert-butyl peroxide as an oxidant, the process achieves high yields (ranging from 51% to 97% across fifteen examples) without requiring any transition metals or specialized reaction environments. The reaction demonstrates exceptional substrate scope with various R1 and R2 substitutions while maintaining consistent performance across different functional groups as documented in the patent's experimental tables. This streamlined approach not only reduces manufacturing costs but also significantly shortens development timelines by eliminating multiple intermediate isolation steps required in conventional syntheses.

Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113307790B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
