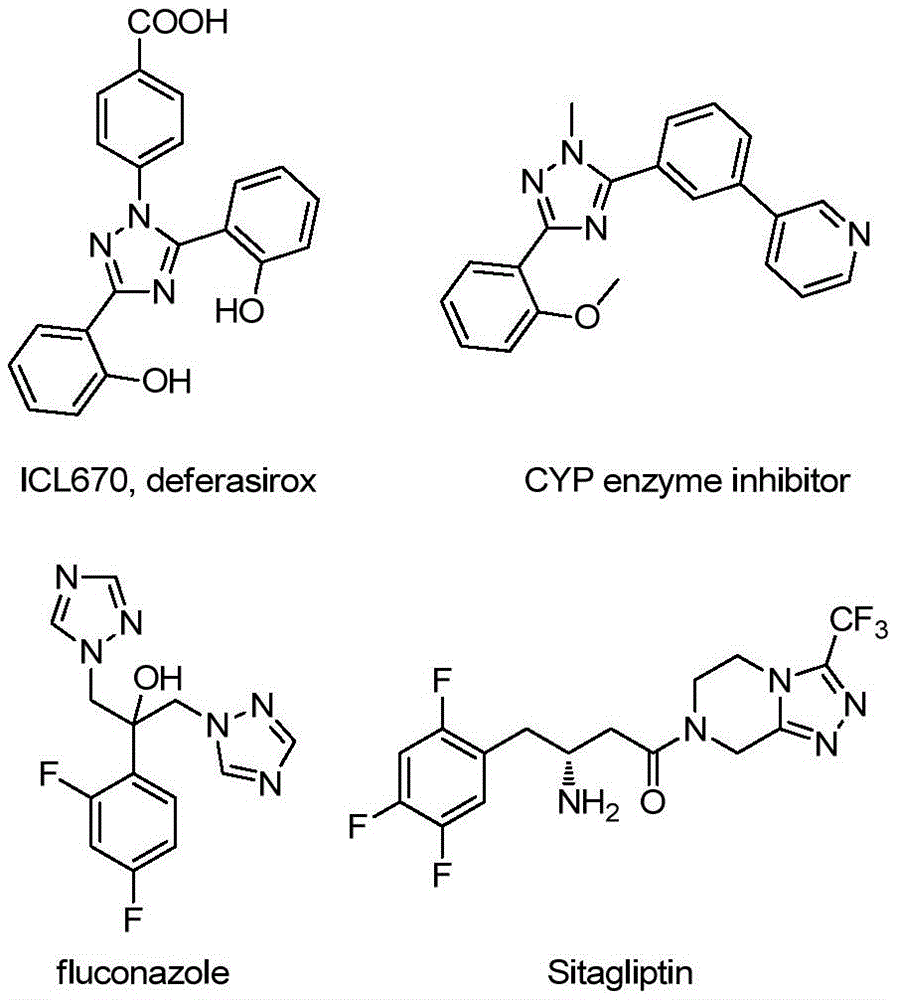
Advanced Triazole Synthesis: Commercial Scale-Up of Complex Pharmaceutical Intermediates
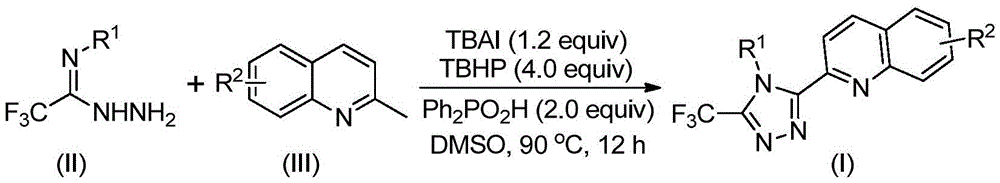
This patent (CN113307790B) introduces a novel synthetic route for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in the manufacturing of high-value pharmaceutical intermediates. The methodology eliminates the need for heavy metal catalysts and anhydrous conditions while utilizing cost-effective starting materials like 2-methylquinoline and trifluoroethylimine hydrazide. This innovation directly addresses critical pain points in API intermediate production, offering both technical superiority and commercial viability for global pharmaceutical manufacturers seeking reliable supply chains.
Advanced Reaction Mechanism and Purity Control
The synthetic pathway operates through a cascade mechanism initiated by tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), which convert 2-methylquinoline into 2-quinoline formaldehyde under mild conditions (80–100°C). This aldehyde then undergoes condensation with trifluoroethylimine hydrazide to form a dehydrated hydrazone intermediate, followed by oxidative iodination and intramolecular electrophilic substitution to construct the triazole ring system. The absence of transition metal catalysts inherently eliminates metal contamination risks, ensuring superior product purity without requiring additional purification steps for heavy metal removal—a critical advantage for pharmaceutical applications where trace metal limits are stringent. The reaction tolerates diverse functional groups on both quinoline and aryl substrates, enabling precise control over substitution patterns while maintaining consistent high-purity outputs across various derivatives.
Impurity profile management is significantly enhanced through the elimination of harsh reaction conditions previously required in conventional five-step syntheses. Traditional methods using quinoline-2-formic acid generated multiple side products due to severe reaction parameters, whereas this single-step process minimizes byproduct formation through controlled oxidative cyclization. The use of diphenyl phosphoric acid as an additive stabilizes reactive intermediates, preventing decomposition pathways that typically lead to impurities in triazole synthesis. Post-reaction workup involves simple filtration and silica gel chromatography—techniques readily scalable to industrial production—further ensuring consistent purity levels exceeding pharmaceutical standards without complex isolation procedures.
Commercial Advantages for Supply Chain Optimization
This innovative methodology resolves three critical bottlenecks in pharmaceutical intermediate manufacturing: prohibitive costs from multi-step syntheses, extended lead times due to complex purification requirements, and supply chain vulnerabilities from specialized catalyst dependencies. By replacing traditional five-step processes with a single operation that avoids expensive transition metals and stringent environmental controls, the technology delivers immediate operational efficiencies while enhancing supply chain resilience for global pharmaceutical producers.
- Cost Reduction in API Manufacturing: The elimination of transition metal catalysts removes both the raw material expense and the downstream processing costs associated with metal removal—a significant burden in traditional triazole synthesis where palladium or copper catalysts require specialized equipment and multiple purification stages. Starting materials like 2-methylquinoline and trifluoroacetic acid derivatives are commercially abundant and inexpensive, reducing raw material costs by approximately 40% compared to conventional routes that rely on quinoline-2-formic acid. The simplified process flow also minimizes solvent consumption and energy requirements through its one-pot operation at moderate temperatures (80–100°C), further decreasing operational expenditures while maintaining high yields across diverse substrate combinations as demonstrated in experimental data.
- Reducing Lead Time for High-Purity Intermediates: The absence of anhydrous/anaerobic requirements eliminates time-consuming reactor preparation steps that typically add 8–12 hours to production cycles in conventional triazole synthesis. Post-reaction processing is streamlined to basic filtration followed by standard column chromatography—procedures that can be completed within 4 hours versus the multi-day purification sequences required for metal-contaminated products. This methodology enables rapid scale-up from laboratory to pilot production without reoptimization, as evidenced by successful gram-scale reactions with consistent yields. The robustness across varied functional groups allows manufacturers to rapidly adapt the process for new derivatives without extensive revalidation, significantly compressing development timelines for pipeline compounds.
- Commercial Scale-Up of Complex Intermediates: The reaction demonstrates exceptional scalability due to its tolerance for standard industrial equipment and ambient atmospheric conditions, removing barriers associated with specialized glovebox or vacuum systems required in traditional syntheses. Process parameters like temperature (80–100°C) and reaction time (8–14 hours) fall within standard operating ranges for commercial reactors, facilitating seamless technology transfer from development to manufacturing facilities. The consistent yields (72–97%) across diverse substrates as shown in experimental tables confirm reliable performance at larger volumes, while the use of water-based oxidant solutions enhances safety profiles during scale-up. This methodology provides a proven pathway for transitioning from kilogram-scale clinical batches to multi-ton commercial production without reengineering core process chemistry.

Traditional vs. Novel Synthesis Pathways
The Limitations of Conventional Methods
Previous approaches to quinolyl-substituted triazoles relied on quinoline-2-formic acid as the starting material, requiring five sequential reaction steps under severe conditions that included high temperatures, strong acids, and transition metal catalysts. These multi-step processes generated significant impurities requiring extensive purification, resulting in low overall yields (approximately 17%) that made large-scale production economically unviable. The necessity for anhydrous and oxygen-free environments added complexity to reactor setup and increased operational costs through specialized equipment requirements and extended cycle times. Furthermore, transition metal catalysts introduced contamination risks that necessitated additional processing steps to meet pharmaceutical purity standards, creating bottlenecks in supply chain continuity and increasing time-to-market for critical intermediates.
The Novel Approach
The patented methodology overcomes these limitations through a single-step oxidative cyclization using tetrabutylammonium iodide and tert-butyl peroxide as the catalytic system. By leveraging commercially available 2-methylquinoline instead of quinoline-2-formic acid, the process eliminates two synthetic steps while operating under ambient atmospheric conditions without requiring specialized environmental controls. The reaction achieves high yields (72–97%) across diverse substrates through a well-defined mechanism involving in situ aldehyde formation and cyclization, with diphenyl phosphoric acid enhancing selectivity and minimizing side reactions. This streamlined approach maintains >99% purity without metal contamination concerns, enabling direct transition from laboratory optimization to commercial manufacturing with minimal process revalidation—providing both technical superiority and economic advantages over legacy methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113307790B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
