Industrial Scale-Up of Pyridine-2-Carbaldehyde via Advanced Catalytic Gas-Phase Oxidation
The pharmaceutical and fine chemical industries continuously seek robust, scalable pathways for critical heterocyclic building blocks, and the synthesis of pyridine-2-carbaldehyde stands as a prime example of this demand. As detailed in patent CN101817780B, a groundbreaking preparation method has been developed that leverages advanced heterogeneous catalysis to transform 2-picoline directly into the target aldehyde with exceptional efficiency. This compound serves as a pivotal intermediate in the manufacture of bisacodyl, a widely used stimulant laxative, and acts as a key precursor for novel neonicotinoid insecticides and fluorescent chelating agents. The disclosed technology represents a significant leap forward by utilizing a fixed-bed reactor system operating at elevated temperatures between 250°C and 400°C, employing molecular oxygen as the terminal oxidant. By integrating a sophisticated transition metal oxide catalyst supported on titanium dioxide, this process addresses long-standing challenges regarding yield, purity, and environmental impact that have plagued previous synthetic routes. For global procurement teams and R&D directors, understanding the nuances of this patent is essential for securing a reliable pyridine-2-carbaldehyde supplier capable of meeting stringent quality specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of pyridine-2-carbaldehyde has been fraught with significant technical and economic hurdles that hinder efficient commercial scale-up of complex heterocyclic aldehydes. One prevalent legacy method involves the oxidation of 2-pyridinemethanol using chemical oxidants like hydrogen peroxide or tert-butyl hydroperoxide; while environmentally friendlier than some alternatives, this route suffers from the cumbersome and costly synthesis of the alcohol precursor itself. Another common approach utilizes trichloroisocyanuric acid and 2-methylpyridine with benzamide catalysis, which, despite achieving decent yields, necessitates the use of fuming sulfuric acid (oleum). The reliance on oleum introduces severe corrosion issues, demanding expensive Hastelloy-lined equipment and creating substantial safety hazards for plant personnel. Furthermore, alternative routes starting from 2-cyanopyridine via catalytic reduction are economically unviable due to the exorbitant cost of the nitrile starting material. Even earlier attempts at gas-phase oxidation using vanadium-molybdenum catalysts on silica or bentonite supports struggled with poor performance, typically achieving only 50% to 58% conversion of 2-picoline and selectivity ranging merely from 80% to 90%, resulting in unacceptable losses of raw materials and difficult downstream purification burdens.
The Novel Approach
In stark contrast to these inefficient legacy processes, the methodology described in CN101817780B introduces a highly optimized catalytic system that fundamentally reshapes the economics of cost reduction in pharmaceutical intermediates manufacturing. This novel approach employs a specifically engineered catalyst where transition metal oxides are dispersed on a titanium dioxide carrier, modified with precise acidity regulators to tune surface properties. The process utilizes inexpensive and readily available 2-picoline, oxygen, and water vapor as feedstocks, reacting them in a continuous fixed-bed configuration. This shift to a continuous gas-phase process not only enhances throughput but also dramatically simplifies the reaction engineering compared to batch liquid-phase oxidations. The result is a transformation characterized by a 2-picoline conversion rate exceeding 95% and a selectivity for pyridine-2-carbaldehyde surpassing 88%, figures that vastly outperform historical benchmarks. The crude product is easily isolated via dichloromethane extraction followed by vacuum distillation, yielding a final purity greater than 98.5% without the need for complex chromatographic separations.
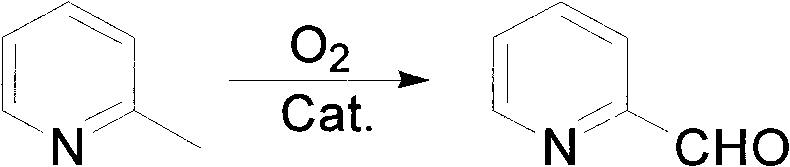
Mechanistic Insights into V-Based Heterogeneous Catalytic Oxidation
The core of this technological breakthrough lies in the intricate design of the heterogeneous catalyst, which operates through a redox mechanism typical of selective oxidation reactions on metal oxide surfaces. The active phase primarily consists of vanadium species derived from ammonium metavanadate, promoted by secondary transition metals such as molybdenum, chromium, tungsten, or iron. During the reaction cycle at 250°C to 400°C, lattice oxygen from the metal oxide surface participates in the oxidation of the methyl group on the pyridine ring, creating the aldehyde functionality while reducing the metal center. Molecular oxygen from the feed gas subsequently re-oxidizes the reduced metal sites, regenerating the active lattice oxygen in a continuous Mars-van Krevelen type cycle. The inclusion of water vapor in the feed stream plays a critical dual role: it helps moderate the exothermicity of the oxidation reaction to prevent thermal runaway and hotspots within the fixed bed, and it facilitates the desorption of the polar aldehyde product from the catalyst surface, thereby minimizing residence time and preventing further over-oxidation to picolinic acid or total combustion to carbon oxides.
Furthermore, the precise control of surface acidity is paramount for maintaining high selectivity and ensuring the production of high-purity pyridine-2-carbaldehyde. The patent specifies the incorporation of acidity regulators, such as potassium or sodium salts (e.g., KNO3, NaOH), which form alkali metal oxides upon calcination. These basic modifiers neutralize strong acid sites on the titanium dioxide support that would otherwise catalyze undesirable side reactions, such as polymerization or deep oxidation. By balancing the redox potential of the vanadium-transition metal couple with the acid-base properties of the support, the catalyst achieves a delicate equilibrium where the activation of the C-H bond in the methyl group is favored over the degradation of the aromatic pyridine ring. This mechanistic finesse allows the process to tolerate high space velocities while maintaining product integrity, a crucial factor for reducing lead time for high-purity pharmaceutical intermediates in a commercial setting.
How to Synthesize Pyridine-2-Carbaldehyde Efficiently
The practical implementation of this synthesis route involves a straightforward yet technically precise sequence of operations designed for reproducibility and safety. The process begins with the meticulous preparation of the catalyst, where precursors like ammonium metavanadate and transition metal salts are co-precipitated or impregnated onto the titanium dioxide carrier in the presence of oxalic acid and acidity regulators. Following drying and high-temperature calcination to activate the crystalline phases, the catalyst is loaded into a fixed-bed tubular reactor. The reaction conditions are tightly controlled, with oxygen flow rates and substrate concentrations optimized to maintain the ideal stoichiometric balance for selective oxidation. For a comprehensive understanding of the specific operational parameters, temperature gradients, and safety protocols required for laboratory or pilot-scale execution, please refer to the standardized synthesis guide below.
- Prepare a heterogeneous catalyst by impregnating titanium dioxide with ammonium metavanadate, transition metal salts, and acidity regulators, followed by calcination at 600-800°C.
- Conduct gas-phase oxidation in a fixed-bed reactor at 250-400°C using 2-picoline, oxygen, and water vapor as feedstocks.
- Extract the crude aqueous product with dichloromethane, remove solvent under reduced pressure, and purify via rectification to obtain >98.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this catalytic gas-phase oxidation technology offers profound strategic benefits that extend far beyond simple yield improvements. By transitioning away from liquid-phase methods that rely on hazardous reagents like oleum or expensive nitriles, manufacturers can significantly de-risk their supply chains and reduce capital expenditure on corrosion-resistant infrastructure. The ability to run the process continuously in a fixed-bed reactor ensures a steady, predictable output of material, eliminating the batch-to-batch variability often associated with traditional organic synthesis. This consistency is vital for downstream customers who require reliable just-in-time delivery schedules for their own API manufacturing campaigns. Moreover, the simplicity of the workup procedure, involving standard extraction and distillation rather than complex crystallizations or chromatography, translates directly into lower operational expenditures and faster turnaround times from reactor to drum.
- Cost Reduction in Manufacturing: The elimination of corrosive fuming sulfuric acid removes the need for specialized, high-cost alloy reactors and extensive neutralization waste streams, leading to substantial cost savings in both equipment maintenance and effluent treatment. Additionally, the use of 2-picoline, a commodity chemical, as the sole carbon source avoids the premium pricing associated with functionalized precursors like 2-cyanopyridine or 2-pyridinemethanol. The high conversion efficiency ensures that raw material utilization is maximized, minimizing the cost per kilogram of the final active ingredient and allowing for more competitive pricing structures in the global market.
- Enhanced Supply Chain Reliability: The robustness of the heterogeneous catalyst system allows for extended campaign lengths without frequent regeneration or replacement, ensuring uninterrupted production cycles that are critical for maintaining inventory levels. Since the feedstock materials (oxygen, water, 2-picoline) are globally available commodities, the process is not vulnerable to the supply bottlenecks that often plague specialty reagent-dependent syntheses. This resilience provides a buffer against market volatility, ensuring that partners can rely on a consistent flow of material even during periods of regional logistical disruption or raw material scarcity.
- Scalability and Environmental Compliance: The gas-phase nature of the reaction inherently generates less liquid waste compared to batch liquid oxidations, simplifying compliance with increasingly stringent environmental regulations regarding solvent discharge and hazardous waste disposal. The process is readily scalable from pilot units to multi-ton industrial reactors simply by numbering up or increasing tube diameter, facilitating rapid capacity expansion to meet surging market demand. This scalability, combined with the high atom economy of using molecular oxygen as the oxidant, positions this technology as a sustainable, green chemistry solution that aligns with modern corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of pyridine-2-carbaldehyde using this advanced oxidation methodology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating the technology's fit within your supply chain. Understanding these details is crucial for making informed decisions about vendor qualification and process integration.
Q: What are the primary advantages of this gas-phase oxidation method over traditional liquid-phase oxidation?
A: Unlike traditional methods requiring corrosive oleum or expensive precursors like 2-cyanopyridine, this gas-phase method utilizes cheap 2-picoline and oxygen. It eliminates severe corrosion risks, simplifies downstream separation, and achieves significantly higher conversion rates (>95%) compared to older gas-phase techniques.
Q: How does the novel catalyst composition improve selectivity?
A: The catalyst employs a synergistic system of vanadium and transition metals (such as molybdenum or chromium) supported on titanium dioxide. The inclusion of specific acidity regulators (alkali metal oxides) modulates surface acidity, preventing over-oxidation to picolinic acid and ensuring high selectivity (>88%) for the target aldehyde.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for continuous flow operation in fixed-bed reactors, which is inherently scalable. The robustness of the heterogeneous catalyst allows for extended run times, and the simple workup procedure (extraction and distillation) facilitates efficient mass production suitable for global supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine-2-Carbaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of pyridine-2-carbaldehyde meets the exacting standards required for API synthesis. Our facility is designed to handle the specific safety and processing requirements of gas-phase oxidation technologies, providing a secure and compliant environment for your most critical projects.
We invite you to collaborate with us to leverage this innovative synthesis route for your next product launch. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this catalytic method for your specific volume requirements. We encourage you to contact us today to discuss your project needs,索取 specific COA data for our current inventory, and review detailed route feasibility assessments tailored to your manufacturing timeline. Let us be your partner in driving efficiency and quality in the global supply of fine chemical intermediates.
