Advanced Synthesis of Anticancer Ursolic Acid Derivatives for Commercial Pharmaceutical Production
The pharmaceutical industry is constantly seeking novel scaffolds to overcome drug resistance and improve therapeutic indices in oncology treatments. Patent CN102260315A introduces a significant advancement in the field of anticancer agents by detailing a series of ursolic acid derivatives modified through a unique ethylenediamine linker system. This specific chemical architecture connects the C-28 carboxyl group of the natural triterpenoid ursolic acid with various natural alpha-amino acids, creating a new class of compounds with demonstrated antitumor activity. The innovation lies not just in the final molecule but in the strategic use of the diamine spacer, which alters the physicochemical properties of the parent compound. For R&D directors and procurement specialists, understanding the nuances of this synthesis is critical for evaluating its potential as a reliable pharmaceutical intermediate supplier solution. The patent data provides robust experimental evidence, including specific reaction conditions and biological assay results, confirming the viability of this route for developing high-purity oncology intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the modification of ursolic acid for medicinal purposes has often focused on direct esterification or simple amidation at the C-28 position without intermediate spacers. These conventional methods frequently encounter challenges related to poor water solubility and limited bioavailability, which hinder the clinical efficacy of the resulting drugs. Furthermore, direct coupling can sometimes lead to steric hindrance that prevents the active moiety from effectively binding to target proteins within cancer cells. Existing literature often reports complex purification processes required to separate unreacted starting materials from the desired product, leading to lower overall yields and increased production costs. In many cases, the lack of a flexible linker restricts the conformational freedom of the molecule, reducing its ability to interact optimally with biological receptors. These limitations necessitate a more sophisticated approach to molecular design that can enhance both the pharmacokinetic profile and the synthetic efficiency of ursolic acid-based therapeutics.
The Novel Approach
The methodology outlined in patent CN102260315A overcomes these historical barriers by introducing ethylenediamine as a bifunctional connecting arm. This novel approach allows for the formation of a stable amide bond while simultaneously introducing a secondary amine group that can be further functionalized with amino acids. By utilizing this spacer, the synthesis achieves a balance between lipophilicity and hydrophilicity, which is crucial for drug absorption and distribution. The process described involves a sequential activation strategy where the C-28 carboxyl is first activated using oxalyl chloride before reacting with the diamine, ensuring high conversion rates. This step-wise construction enables precise control over the stereochemistry and purity of the final derivatives. For supply chain heads, this structured approach implies a more predictable manufacturing process with fewer side reactions, thereby reducing the burden on downstream purification units and enhancing the overall cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Ethylenediamine-Mediated Amide Coupling
The core of this synthetic route relies on the efficient activation of the triterpenoid carboxylic acid and its subsequent nucleophilic attack by the diamine. Initially, the C-3 hydroxyl group of ursolic acid is protected via acetylation to prevent unwanted side reactions during the activation phase. The C-28 carboxyl group is then converted into an acid chloride using oxalyl chloride, a highly reactive species that facilitates rapid amide bond formation. When anhydrous ethylenediamine is introduced, it selectively reacts with the acid chloride to form the mono-amide intermediate, leaving the second amine group available for further conjugation. This mechanistic pathway is critical for maintaining the integrity of the ursolic acid skeleton while introducing the necessary functional handles for diversity-oriented synthesis. The use of Fmoc-protected amino acids in the subsequent step ensures that the amino group of the amino acid does not interfere prematurely, allowing for controlled coupling using carbodiimide reagents like EDCI.
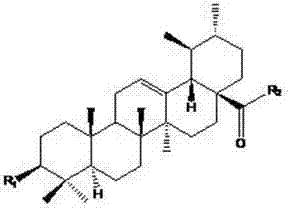
Following the initial coupling, the removal of protecting groups such as Fmoc and acetyl is performed under mild alkaline or acidic conditions to reveal the final active pharmacophore. The impurity profile of this reaction is generally clean, as the byproducts are primarily water-soluble salts or urea derivatives from the coupling reagents, which can be easily removed during aqueous workup. This high level of selectivity is paramount for meeting the stringent purity specifications required for clinical grade materials. The structural diversity is achieved by varying the natural alpha-amino acid component, such as glycine, methionine, or phenylalanine, each imparting different physicochemical properties to the final molecule. Understanding these mechanistic details allows R&D teams to predict the stability and reactivity of these intermediates during scale-up, ensuring that the commercial scale-up of complex pharmaceutical intermediates proceeds without unexpected chemical deviations.
How to Synthesize Ursolic Acid Derivatives Efficiently
The synthesis protocol described in the patent offers a reproducible pathway for generating these high-value anticancer intermediates. The process begins with the acetylation of ursolic acid in pyridine, followed by activation with oxalyl chloride in dichloromethane. The resulting acid chloride is then reacted with ethylenediamine to form the key linker intermediate. Finally, this intermediate is coupled with protected amino acids using standard peptide coupling conditions. Detailed standard operating procedures for each step, including specific molar ratios, temperature controls, and purification methods, are essential for maintaining batch-to-batch consistency. The patent examples demonstrate that recrystallization from methanol or ethanol can effectively purify the intermediates to high standards. For technical teams looking to implement this route, adhering to the specific reaction times and quenching procedures outlined in the documentation is vital for maximizing yield and minimizing impurity formation.
- Acetylation of ursolic acid at the C-3 position using acetic anhydride and pyridine to protect the hydroxyl group.
- Activation of the C-28 carboxyl group using oxalyl chloride followed by reaction with anhydrous ethylenediamine to form the linker intermediate.
- Coupling the intermediate with Fmoc-protected natural alpha-amino acids using EDCI, followed by deprotection and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthesis route offers significant strategic benefits regarding raw material availability and process safety. The starting materials, including ursolic acid, ethylenediamine, and common natural amino acids, are commercially available in bulk quantities from established chemical suppliers. This abundance ensures a stable supply chain and mitigates the risk of raw material shortages that can plague more exotic synthetic routes. Furthermore, the reaction conditions employed, such as room temperature stirring and standard solvent systems like dichloromethane and ethyl acetate, are compatible with existing manufacturing infrastructure. This compatibility reduces the need for specialized equipment investments, leading to substantial cost savings in facility upgrades. The elimination of toxic heavy metal catalysts, often used in cross-coupling reactions, simplifies the waste treatment process and aligns with increasingly strict environmental regulations.
- Cost Reduction in Manufacturing: The synthetic pathway avoids the use of expensive transition metal catalysts and precious ligands, which are often cost-prohibitive in large-scale production. Instead, it relies on organic reagents like oxalyl chloride and EDCI, which are cost-effective and widely sourced. The high yields reported in the patent examples, particularly for the final coupling steps, indicate efficient atom economy and reduced material waste. By minimizing the number of purification steps required, such as avoiding complex chromatographic separations in favor of recrystallization, the overall processing time and labor costs are significantly reduced. This efficiency translates directly into a more competitive pricing structure for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the synthesis ensures that the supply chain is robust against market fluctuations. Ursolic acid is a natural product extract with a stable supply base, and amino acids are produced globally in massive quantities for the food and feed industries. This diversification of raw material sources reduces the dependency on single suppliers and enhances the resilience of the procurement strategy. Additionally, the intermediates generated during the process are stable solids that can be stored and transported without special handling requirements, facilitating smoother logistics and inventory management. This stability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream drug manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are easily transferable from laboratory to pilot and commercial scales. The use of standard solvents allows for efficient recovery and recycling systems, further reducing the environmental footprint and operational costs. The absence of heavy metals simplifies the regulatory approval process for the manufacturing site, as there is no need for extensive testing for residual metal contaminants in the final product. This compliance advantage accelerates the time to market for new drug candidates utilizing these intermediates. The waste streams generated are primarily organic and can be treated using conventional incineration or biological treatment methods, ensuring adherence to global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these ursolic acid derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their product pipelines. The information covers aspects of chemical stability, biological activity, and manufacturing scalability, providing a comprehensive overview for decision-makers.
Q: What is the primary structural advantage of using ethylenediamine in this synthesis?
A: The ethylenediamine linker provides a flexible spacer between the ursolic acid core and the amino acid moiety, potentially enhancing bioavailability and interaction with cancer cell receptors compared to direct conjugation methods.
Q: Which cancer cell lines have shown sensitivity to these derivatives?
A: In vitro pharmacological tests indicate significant inhibitory activity against human liver cancer HepG2, colon cancer HT-29, gastric cancer AGS and BGC-823, and prostate cancer PC-3 cell lines.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the method utilizes standard organic synthesis techniques such as acetylation, acid chloride activation, and amide coupling, which are well-established and adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ursolic Acid Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing for complex pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from bench to plant. We understand the critical importance of stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest industry standards. Our expertise in amide coupling and triterpenoid chemistry positions us as an ideal partner for developing and supplying these advanced anticancer intermediates. We are committed to delivering high-quality materials that support your drug development timelines and regulatory filings.
We invite you to discuss your specific requirements with our technical procurement team to explore how we can optimize your supply chain. Request a Customized Cost-Saving Analysis to understand the economic benefits of partnering with us for your ursolic acid derivative needs. Our team is ready to provide specific COA data and route feasibility assessments tailored to your project's unique constraints. By collaborating with NINGBO INNO PHARMCHEM, you gain access to a reliable partner dedicated to driving innovation and efficiency in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
