Revolutionizing Iodixanol Production: Advanced Intermediate Synthesis for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value contrast agents like Iodixanol, commercially known as Visipaque 320. A pivotal advancement in this domain is detailed in patent CN103058880A, which discloses a novel preparation method for a critical synthetic intermediate, specifically 1,3-bis(acetamino)-N,N'-bis[3,5-bis(chloroformyl)-2,4,6-triiodo-phenyl]-2-propanol acetate. This technical breakthrough addresses long-standing inefficiencies in the final dimerization condensation reactions that have historically plagued the manufacturing of this non-ionic, water-soluble X-ray contrast agent. By fundamentally restructuring the synthetic sequence, this method effectively avoids the problematic dimerization of 5-acetamino-N,N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-isophthalamide as the terminal step. For R&D Directors and Procurement Managers seeking a reliable pharmaceutical intermediates supplier, understanding this shift is crucial as it directly correlates to enhanced product quality and process economics. The innovation ensures that the content of Iodixanol in the final product is effectively improved, setting a new benchmark for industrial production standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Visipaque 320 has been hindered by the reliance on Compound A as the starting material for the final dimerization reaction, a method documented in various prior arts including EP108638 and CN1340042. In these conventional routes, the conversion of Compound A to Visipaque 320 typically achieves only 40% to 60% efficiency, resulting in a crude product purity of merely about 80%. More critically, these processes generate inexpungible impurities exceeding 2%, which necessitates rigorous and costly purification measures such as preparative liquid chromatography or repeated crystallization cycles up to five times. This reliance on complex purification not only escalates the operational expenditure but also introduces significant bottlenecks in the supply chain, making the commercial scale-up of complex pharmaceutical intermediates challenging. The high purification cost and material loss during these extensive cleaning steps represent a major defect in the industry's preparation of Visipaque 320, limiting the ability to achieve cost reduction in pharmaceutical manufacturing.
The Novel Approach
In stark contrast, the novel approach outlined in the patent data introduces a strategic synthesis of a specific chloroformyl intermediate prior to the final amidation, fundamentally altering the impurity profile of the reaction. By synthesizing the dimeric structure earlier in the sequence and functionalizing it as an acid chloride (Formula I), the process achieves a crude product purity greater than 90%, with the highest content of any single impurity remaining below 2%. This substantial improvement in crude quality means that the difficulty of purifying the product is drastically reduced, often requiring only simple methanol eddy crystallization rather than complex chromatographic separation. 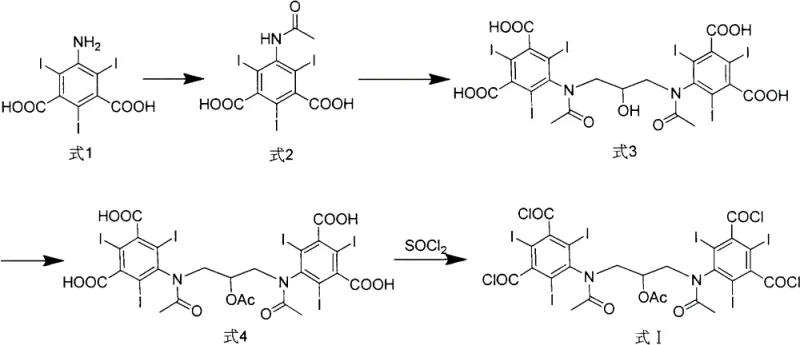 This shift allows for a more streamlined workflow where intermediate products can be used in the next step without further extensive purification, thereby reducing product loss and lowering the requirements for production units. For supply chain heads, this translates to a more predictable and efficient manufacturing timeline, significantly enhancing the reliability of the supply chain for high-purity pharmaceutical intermediates.
This shift allows for a more streamlined workflow where intermediate products can be used in the next step without further extensive purification, thereby reducing product loss and lowering the requirements for production units. For supply chain heads, this translates to a more predictable and efficient manufacturing timeline, significantly enhancing the reliability of the supply chain for high-purity pharmaceutical intermediates.
Mechanistic Insights into the Chloroformyl Intermediate Synthesis
The core of this technological advancement lies in the precise control of the acylation and chlorination mechanisms that generate the reactive Formula I intermediate. The process begins with the acylation of 5-amino-2,4,6-triiodo isophthalic acid using acetic anhydride at controlled temperatures between 60°C and 80°C, ensuring complete protection of the amino group. Subsequently, the dimerization is conducted under strictly alkaline conditions with a pH of 11.5 to 12.5, utilizing reagents like 1,3-dichloro-2-propanol or epichlorohydrin to bridge the aromatic rings. The critical mechanistic step involves the conversion of the carboxylic acid groups into acid chlorides using thionyl chloride in the presence of an organic base catalyst like DMF. This activation creates a highly reactive species that facilitates the subsequent amidation with amino-glycerol under mild conditions, typically between -5°C and 5°C. 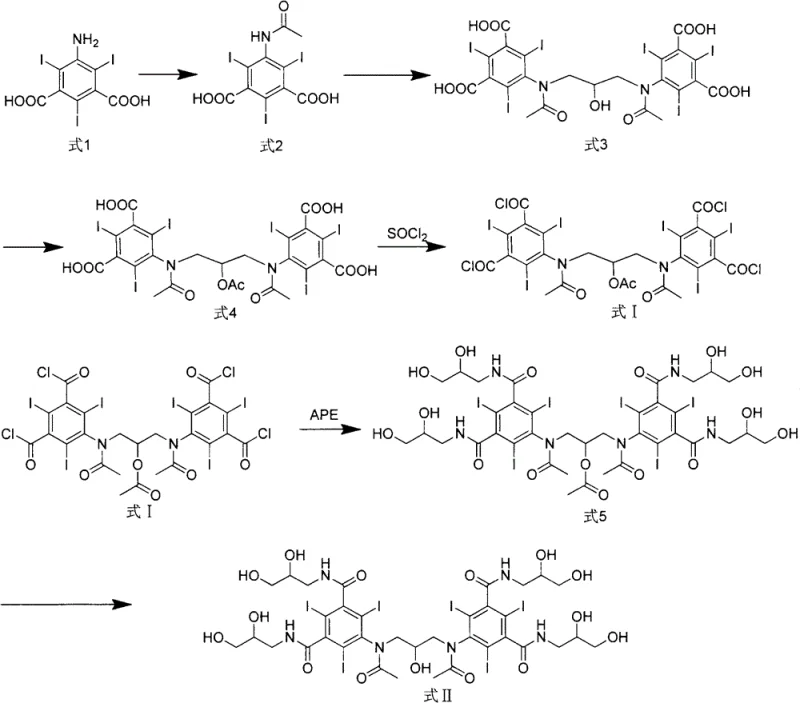 By managing the reactivity through the acid chloride intermediate, the reaction avoids the harsh conditions often required for direct amide coupling, thereby minimizing side reactions and thermal degradation that lead to impurities.
By managing the reactivity through the acid chloride intermediate, the reaction avoids the harsh conditions often required for direct amide coupling, thereby minimizing side reactions and thermal degradation that lead to impurities.
Furthermore, the impurity control mechanism is inherently built into the sequence of functional group transformations. By establishing the dimeric backbone before introducing the sensitive hydroxypropyl side chains, the process isolates the bulk of the molecular weight early, allowing for easier removal of unreacted monomers. The acetylation step protects the secondary amines during the chlorination, preventing unwanted side reactions at the nitrogen centers. This orthogonal protection strategy ensures that when the final deprotection and hydrolysis occur, the resulting Visipaque 320 crude product contains less than 1% of other single impurities. Such rigorous control over the impurity spectrum is vital for R&D Directors focused on purity and impurity profiles, as it simplifies the regulatory filing process by providing a cleaner drug substance. The ability to achieve these specifications through crystallization rather than chromatography underscores the robustness of the chemical design.
How to Synthesize Iodixanol Intermediate Efficiently
The synthesis of this critical intermediate requires adherence to specific reaction parameters to ensure reproducibility and safety on a commercial scale. The process involves a sequence of acylation, dimerization, acetylation, and chlorination, each demanding precise temperature and stoichiometric control to maximize yield and minimize hazardous byproducts. Operators must maintain strict pH levels during the dimerization phase and ensure anhydrous conditions during the chlorination step to prevent hydrolysis of the acid chloride. The detailed standardized synthetic steps, including specific solvent choices like 1-methoxy-2-propanol and catalyst loading rates, are essential for technology transfer.
- Acylation of 5-amino-2,4,6-triiodo isophthalic acid with acetic anhydride at 60°C to 80°C.
- Dimerization using 1,3-dichloro-2-propanol in alkaline conditions (pH 11.5 to 12.5) at 10°C to 20°C.
- Acetylation and subsequent chlorination using thionyl chloride to generate the reactive acid chloride intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers profound advantages that extend beyond mere chemical yield. The primary benefit lies in the drastic simplification of the purification process, which directly correlates to substantial cost savings in manufacturing operations. By eliminating the need for preparative liquid chromatography and reducing the number of crystallization cycles, the consumption of solvents and stationary phases is significantly reduced, leading to a lower environmental footprint and reduced waste disposal costs. This efficiency gain allows for a more competitive pricing structure without compromising on the stringent quality standards required for injectable contrast agents. The process is designed to be economically viable for large-scale production, addressing the critical need for cost reduction in pharmaceutical manufacturing while maintaining high margins.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in chromatographic purification steps lead to a streamlined cost structure. By achieving high crude purity directly from the reaction, the need for resource-intensive cleaning processes is minimized, resulting in significant operational expenditure savings. The ability to use intermediates directly in the next step without extensive isolation further reduces labor and equipment time, enhancing overall plant throughput. This qualitative improvement in process efficiency ensures that the final product can be offered at a more competitive price point in the global market.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route, characterized by wide operating windows for temperature and pH, ensures consistent batch-to-batch quality. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the risk of batch failures and production delays. The use of readily available raw materials and standard reagents like thionyl chloride and acetic anhydride further secures the supply chain against raw material shortages. Consequently, partners can rely on a steady flow of materials to meet the demanding schedules of downstream formulation and packaging.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, moving seamlessly from kilogram to multi-ton production without significant re-optimization. The reduction in solvent usage and waste generation aligns with modern environmental compliance standards, making it easier to obtain necessary regulatory approvals for production facilities. The simplified work-up procedures reduce the load on waste treatment plants, contributing to a more sustainable manufacturing profile. This scalability ensures that the supply can grow in tandem with market demand for Visipaque 320, securing long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Iodixanol intermediates based on the patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities. Understanding these details helps stakeholders make informed decisions regarding technology adoption and procurement strategies.
Q: How does this new route improve Iodixanol purity compared to conventional methods?
A: Conventional methods relying on the dimerization of Compound A as the final step typically yield crude products with approximately 80% purity and over 2% impurities. The novel route described in patent CN103058880A shifts the dimerization earlier and utilizes a chloroformyl intermediate, achieving crude purity greater than 90% with single impurities below 2%, significantly simplifying downstream purification.
Q: What are the critical reaction conditions for the dimerization step?
A: The dimerization step requires precise control of alkaline conditions, specifically maintaining a pH between 11.5 and 12.5 using sodium or potassium hydroxide. The reaction temperature is critically kept between 10°C and 20°C over a period of 24 to 48 hours to ensure optimal coupling without degradation.
Q: Why is the chloroformyl intermediate (Formula I) significant for supply chain stability?
A: Formula I serves as a stable, high-purity checkpoint in the synthesis. By isolating this intermediate before the final amidation with amino-glycerol, manufacturers can verify quality early, reducing the risk of batch failure in the final expensive steps and ensuring consistent supply of the active pharmaceutical ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iodixanol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving diagnostic agents like Visipaque 320. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Iodixanol intermediate meets the highest industry standards. We are committed to leveraging advanced synthetic technologies, such as the route described in CN103058880A, to deliver superior value to our global partners.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through our specialized expertise. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Let us be your partner in advancing the availability and affordability of high-quality pharmaceutical intermediates.
