Advanced Carbonylation Strategy for Scalable Thioester Manufacturing
Introduction to Next-Generation Thioester Synthesis
The landscape of organic synthesis for sulfur-containing motifs is undergoing a significant transformation, driven by the urgent need for safer, more efficient, and environmentally benign methodologies. Patent CN113004181B introduces a groundbreaking approach to preparing thioester compounds through a transition metal-catalyzed carbonylation reaction that fundamentally alters the traditional reliance on hazardous thiol reagents. This innovation leverages a sophisticated palladium-catalyzed system utilizing benzyl chloride compounds and sulfonyl chlorides as key starting materials, effectively bypassing the notorious olfactory and toxicity issues associated with mercaptans. For R&D directors and process chemists, this represents a pivotal shift towards more robust synthetic routes that offer superior functional group tolerance and operational simplicity. The methodology not only streamlines the synthetic pathway but also enhances the overall safety profile of the manufacturing process, making it an attractive candidate for the production of high-value pharmaceutical intermediates and fine chemicals where purity and regulatory compliance are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has predominantly relied on the direct acylation of thiols with carboxylic acids or their activated derivatives, a process fraught with significant logistical and chemical challenges. Thiols are notoriously malodorous, volatile, and prone to oxidation, creating severe handling difficulties in both laboratory and industrial settings. Furthermore, the sulfur atoms in thiols possess a strong affinity for transition metals, often leading to catalyst poisoning which drastically reduces reaction efficiency and necessitates higher catalyst loadings or more expensive noble metals. Alternative methods involving the oxidative coupling of aldehydes or substitution reactions of halogenated alkanes often suffer from limited substrate scope, harsh reaction conditions, or poor atom economy. These conventional pathways frequently generate substantial amounts of waste and require complex purification steps to remove unreacted thiols and their oxidation byproducts, thereby inflating production costs and extending lead times for critical intermediate supplies.
The Novel Approach
In stark contrast to these legacy methods, the novel carbonylation strategy disclosed in the patent utilizes inexpensive and stable benzyl chlorides alongside sulfonyl chlorides to construct the thioester backbone with remarkable efficiency. This approach ingeniously employs tungsten hexacarbonyl as a dual-purpose reagent, serving simultaneously as the carbonyl source and the reducing agent, which eliminates the need for external reducing additives and simplifies the reaction stoichiometry. The use of sulfonyl chlorides as the sulfur source completely circumvents the handling of foul-smelling thiols, thereby improving the working environment and reducing the risk of catalyst deactivation. The reaction proceeds smoothly in polar aprotic solvents like acetonitrile at moderate temperatures, demonstrating excellent compatibility with a wide range of functional groups including halogens, alkyls, and alkoxy substituents. This robustness allows for the synthesis of diverse thioester derivatives from readily available commercial building blocks, significantly enhancing the designability of the final molecular architecture.
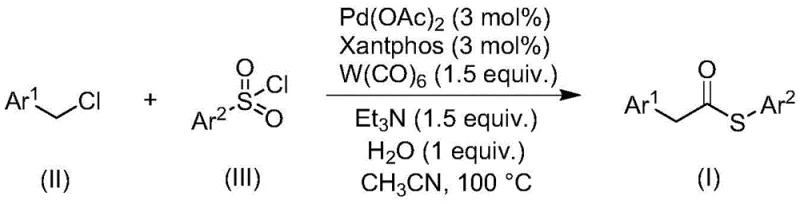
Mechanistic Insights into Pd-Catalyzed Carbonylation
The core of this transformative synthesis lies in the intricate interplay between the palladium catalyst, the Xantphos ligand, and the unique reactivity of tungsten hexacarbonyl. The catalytic cycle likely initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species generated in situ, forming a benzyl-palladium(II) intermediate. Subsequently, carbon monoxide released from the decomposition of tungsten hexacarbonyl inserts into the palladium-carbon bond, generating an acyl-palladium complex. The presence of water and triethylamine facilitates the activation of the sulfonyl chloride, potentially generating a sulfinate species or activating the sulfur center for nucleophilic attack. The acyl-palladium species then undergoes a substitution or transmetallation event with the activated sulfur species, followed by reductive elimination to release the desired thioester product and regenerate the palladium(0) catalyst. This mechanism is highly efficient because the tungsten carbonyl not only provides the necessary CO gas in a controlled manner but also acts as a sacrificial reductant to maintain the palladium in its active low-valent state throughout the cycle.
From an impurity control perspective, this mechanism offers distinct advantages over thiol-based routes by minimizing the formation of disulfide byproducts and metal-sulfur complexes that are difficult to separate. The use of a bulky bidentate phosphine ligand like Xantphos stabilizes the palladium center and promotes the reductive elimination step, which is often the rate-determining step in carbonylation reactions. The tolerance for various substituents on both the benzyl and aryl sulfonyl rings suggests that the electronic and steric properties of the substrates do not significantly hinder the catalytic turnover. This broad substrate scope is crucial for medicinal chemistry applications where rapid analog synthesis is required. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups, ensuring that the final thioester products maintain high chemical integrity and purity profiles suitable for downstream biological testing or further synthetic elaboration.
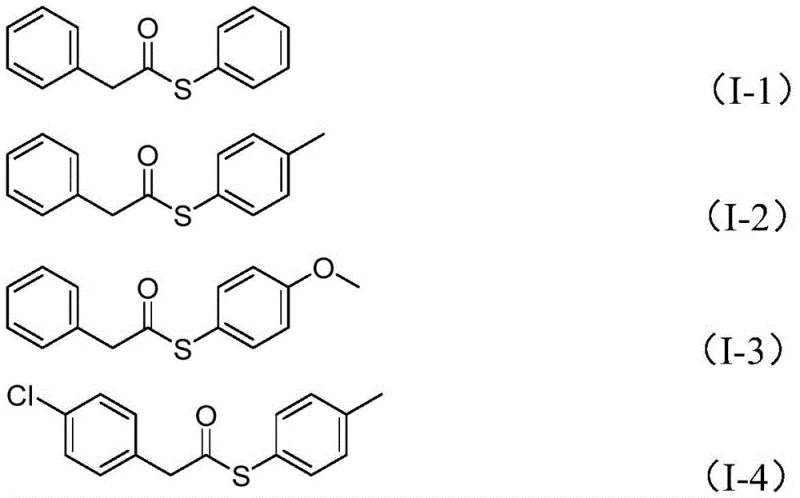
How to Synthesize Thioester Compounds Efficiently
Implementing this carbonylation protocol requires precise attention to reagent ratios and reaction parameters to maximize yield and minimize side reactions. The process begins with the careful weighing of palladium acetate and the Xantphos ligand to ensure the formation of the active catalytic species, followed by the addition of tungsten hexacarbonyl which serves as the solid CO source. The reaction mixture, containing the benzyl chloride, sulfonyl chloride, triethylamine base, and a stoichiometric amount of water, is suspended in anhydrous acetonitrile to facilitate homogeneous catalysis. Heating the sealed vessel to 100°C for 24 hours allows for the complete consumption of starting materials, after which a simple workup involving filtration and silica gel treatment removes metal residues and inorganic salts. For a comprehensive understanding of the specific molar ratios and purification techniques required for different substrates, please refer to the standardized synthesis steps provided below.
- Combine palladium acetate, Xantphos ligand, tungsten carbonyl, triethylamine, water, benzyl chloride, and sulfonyl chloride in an organic solvent like acetonitrile.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 24 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel thioester synthesis method presents a compelling value proposition centered on cost optimization and supply reliability. By replacing volatile and hazardous thiols with stable sulfonyl chlorides and benzyl chlorides, the process significantly reduces the costs associated with specialized storage, ventilation, and personal protective equipment required for handling noxious sulfur compounds. The elimination of external reducing agents further simplifies the bill of materials, lowering the overall raw material expenditure while reducing the complexity of inventory management. Additionally, the use of commercially available and inexpensive starting materials ensures a robust supply chain that is less susceptible to market fluctuations or shortages of exotic reagents. This stability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The dual functionality of tungsten hexacarbonyl as both a carbonyl source and a reducing agent creates a substantial reduction in reagent costs by consolidating two roles into a single additive. This efficiency minimizes the total mass of inputs required per kilogram of product, directly translating to lower variable manufacturing costs. Furthermore, the simplified post-treatment process, which avoids complex extraction procedures often needed to remove excess thiols, reduces solvent consumption and waste disposal fees. The high reaction efficiency and yield reported across various substrates mean that less raw material is wasted, maximizing the return on investment for every batch produced and enhancing the overall economic viability of the manufacturing process.
- Enhanced Supply Chain Reliability: Sourcing stable solids like benzyl chlorides and sulfonyl chlorides is far more reliable than procuring liquid thiols, which often have shorter shelf lives and stricter transportation regulations due to their odor and toxicity. This shift in raw material profile mitigates the risk of supply disruptions caused by regulatory changes or logistical bottlenecks associated with hazardous cargo. The broad functional group tolerance of the reaction allows manufacturers to utilize a wider range of commercially available feedstocks, providing flexibility to switch suppliers or substitute specific derivatives without revalidating the entire process. This adaptability ensures a consistent flow of high-quality intermediates, safeguarding the production pipeline against unforeseen market volatility.
- Scalability and Environmental Compliance: The operational simplicity of running the reaction in standard organic solvents like acetonitrile at atmospheric pressure equivalents facilitates easy scale-up from laboratory to pilot and commercial scales. The absence of gaseous CO cylinders, replaced by the solid tungsten carbonyl source, significantly enhances process safety and reduces the regulatory burden associated with high-pressure gas handling. Moreover, avoiding the generation of thiol-containing waste streams simplifies effluent treatment and aligns with increasingly stringent environmental regulations regarding sulfur emissions. This green chemistry advantage not only reduces compliance costs but also strengthens the company's sustainability profile, a key factor for modern supply chain partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonylation technology in industrial settings. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is essential for process engineers and quality assurance teams evaluating the feasibility of adopting this route for large-scale production. The answers highlight the practical benefits of the new methodology while acknowledging the specific reaction conditions required for optimal performance.
Q: What are the advantages of using sulfonyl chlorides over thiols in thioester synthesis?
A: Using sulfonyl chlorides eliminates the handling of unpleasant-smelling and toxic thiol compounds, which often poison catalysts. This method improves operational safety and simplifies waste management.
Q: What is the role of Tungsten Hexacarbonyl in this reaction?
A: Tungsten Hexacarbonyl serves a dual function as both the carbonyl source (providing CO) and the reducing agent, removing the need for additional reducing reagents and simplifying the reaction stoichiometry.
Q: Is this method suitable for large-scale production of pharmaceutical intermediates?
A: Yes, the method utilizes cheap and readily available starting materials like benzyl chlorides and operates under relatively mild conditions (100°C), making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain a competitive edge in the global fine chemicals market. Our team of expert process chemists has extensively evaluated the carbonylation method described in CN113004181B and confirmed its potential for delivering high-purity thioester intermediates with exceptional consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of thioester compound meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to request specific COA data and route feasibility assessments that demonstrate how this palladium-catalyzed carbonylation process can optimize your supply chain and reduce your overall manufacturing footprint. Let us help you transform your thioester synthesis strategy with a solution that combines scientific excellence with commercial pragmatism.
