Advanced Nickel-Catalyzed Thioester Synthesis for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries continuously seek robust synthetic methodologies that balance efficiency with safety, particularly for constructing privileged scaffolds like thioesters. Patent CN112239384B discloses a groundbreaking preparation method for thioester compounds that addresses long-standing challenges in carbonylation chemistry. This innovation utilizes a nickel-catalyzed system involving nickel triflate, 4,4'-di-tert-butyl-2,2'-bipyridine, and molybdenum hexacarbonyl to couple arylboronic acids with sulfonyl chlorides. Unlike traditional approaches that rely on hazardous thiols, this protocol employs sulfonyl chlorides as a stable sulfur source, operating effectively at 120°C in NMP solvent. The significance of this technology is underscored by the prevalence of thioester motifs in bioactive molecules, as illustrated by key pharmaceutical agents such as Timobesone, Pivalopril, and Spironolactone shown below.
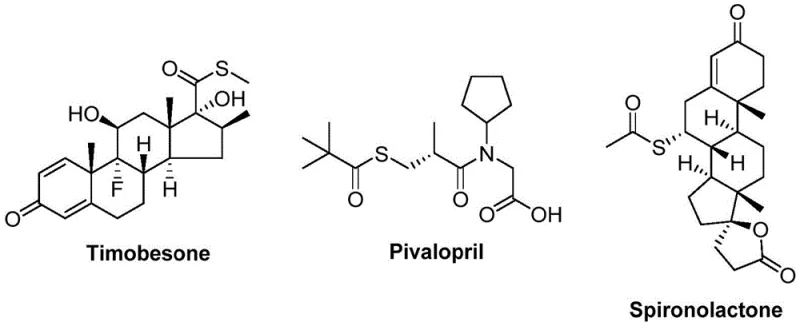
These structural examples highlight the critical role of thioesters in medicinal chemistry, serving as essential intermediates for protein synthesis and acyl donors in complex organic transformations. The ability to access these structures efficiently without the drawbacks of traditional thiol chemistry represents a significant leap forward for process chemists aiming to streamline API manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has predominantly relied on the direct acylation of thiols with carboxylic acids or their activated derivatives. While conceptually straightforward, this classical approach suffers from severe practical limitations that hinder its application in large-scale industrial settings. Thiols are notoriously malodorous, volatile, and toxic, posing significant occupational health and safety risks that require specialized containment infrastructure and rigorous waste management protocols. Furthermore, the nucleophilic nature of thiols often leads to catalyst poisoning in transition metal-mediated processes, necessitating higher catalyst loadings or forcing the use of stoichiometric activating agents that generate substantial chemical waste. These factors collectively drive up the cost of goods sold (COGS) and complicate the regulatory approval process due to residual impurity concerns.
The Novel Approach
In stark contrast, the novel methodology described in the patent leverages a transition metal-catalyzed carbonylation strategy that completely bypasses the need for free thiols. By utilizing readily available arylboronic acids and sulfonyl chlorides as starting materials, the process introduces a sulfur atom and a carbonyl group simultaneously through a streamlined catalytic cycle. The use of sulfonyl chlorides is particularly advantageous as they are inexpensive, shelf-stable solids that do not suffer from the volatility or odor issues associated with thiols. Moreover, the reaction proceeds without the need for external oxidants or reductants, relying instead on the redox interplay between the nickel catalyst and the molybdenum carbonyl source. This atom-economical design not only simplifies the reaction setup but also drastically reduces the environmental footprint by minimizing auxiliary reagent consumption and downstream purification burdens.
Mechanistic Insights into Ni-Catalyzed Carbonylative Coupling
The core of this transformation lies in the sophisticated interplay between the nickel catalyst system and the carbonyl source. The reaction initiates with the oxidative addition of the sulfonyl chloride to the low-valent nickel species generated in situ from Ni(OTf)2 and the dtbbpy ligand. Molybdenum hexacarbonyl serves as a solid, safe surrogate for carbon monoxide gas, releasing CO under the thermal conditions of 120°C to facilitate the migratory insertion step. This generates an acyl-nickel intermediate which subsequently undergoes transmetallation with the arylboronic acid, activated by the zinc iodide and base system. The final reductive elimination step releases the desired thioester product and regenerates the active nickel catalyst, closing the catalytic cycle. This mechanism ensures high turnover numbers and exceptional selectivity, avoiding the formation of homocoupling byproducts often seen in boronic acid chemistry.
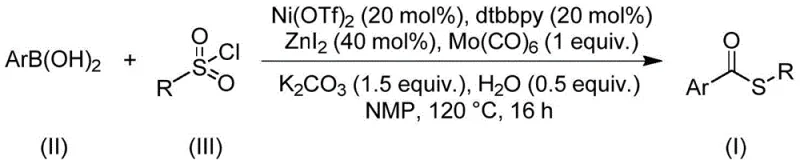
Impurity control is inherently built into this mechanistic pathway. The specific choice of ligands and additives, such as zinc iodide, helps to suppress side reactions like protodeboronation or hydrolysis of the sulfonyl chloride. The mild basicity of potassium carbonate combined with trace water facilitates the activation of the boronic acid without promoting excessive decomposition of sensitive functional groups. Consequently, the crude reaction mixtures are remarkably clean, allowing for straightforward purification via standard silica gel chromatography. This high level of chemoselectivity is crucial for pharmaceutical applications where strict limits on genotoxic impurities and heavy metals must be maintained throughout the synthesis of complex drug candidates.
How to Synthesize Thioester Compounds Efficiently
Implementing this synthesis requires precise adherence to the optimized reaction parameters to achieve the reported high yields. The process involves charging a reaction vessel with the nickel catalyst, ligand, carbonyl source, and additives in an aprotic solvent like NMP, followed by the addition of the coupling partners. The mixture is then heated to promote the carbonylation event. For detailed operational procedures and specific stoichiometric ratios validated across multiple substrate examples, please refer to the standardized protocol below.
- Combine nickel triflate, dtbbpy ligand, molybdenum hexacarbonyl, zinc iodide, potassium carbonate, water, arylboronic acid, and sulfonyl chloride in NMP solvent.
- Heat the reaction mixture to 120°C and maintain stirring for 16 hours under inert atmosphere to ensure complete carbonylation.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology offers transformative benefits that directly impact the bottom line and operational resilience. The shift from thiols to sulfonyl chlorides eliminates the need for specialized storage facilities required for malodorous and hazardous sulfur reagents, thereby reducing facility overhead costs and insurance premiums. Furthermore, the reliance on commodity chemicals like arylboronic acids and sulfonyl chlorides ensures a stable and diversified supply base, mitigating the risk of raw material shortages that can plague niche reagent markets. The robustness of the reaction conditions also implies a wider processing window, reducing the likelihood of batch failures and ensuring consistent delivery schedules for downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous thiols, coupled with the removal of external oxidants and reductants, leads to a substantial reduction in raw material costs. Additionally, the simplified workup procedure reduces solvent consumption and labor hours associated with complex purification steps, driving down the overall manufacturing expense per kilogram of product.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials that are produced on a multi-ton scale globally, manufacturers can secure long-term supply contracts with favorable pricing. The stability of sulfonyl chlorides allows for bulk purchasing and extended inventory holding without degradation, providing a buffer against market volatility and logistics disruptions.
- Scalability and Environmental Compliance: The absence of gaseous carbon monoxide cylinders and the use of solid Mo(CO)6 significantly enhance process safety, facilitating easier scale-up from laboratory to pilot and commercial plants. The reduced generation of sulfur-containing waste streams simplifies effluent treatment, ensuring compliance with increasingly stringent environmental regulations without requiring capital-intensive scrubbing systems.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this nickel-catalyzed thioester synthesis. These insights are derived directly from the experimental data and optimization studies detailed in the patent documentation, providing clarity on substrate compatibility and process parameters.
Q: What is the primary advantage of using sulfonyl chlorides over thiols in this synthesis?
A: Sulfonyl chlorides serve as a stable, odorless, and non-toxic sulfur source that avoids the catalyst poisoning and unpleasant odors typically associated with volatile thiols, significantly improving operational safety and catalyst longevity.
Q: Does this nickel-catalyzed method require external oxidants or reductants?
A: No, the process is designed to proceed without additional oxidants or reductants, utilizing the intrinsic redox properties of the nickel catalyst system and molybdenum hexacarbonyl as the carbonyl source, which simplifies the reaction setup and waste treatment.
Q: What is the functional group tolerance of this carbonylation reaction?
A: The method exhibits excellent functional group tolerance, successfully accommodating substrates with alkyl, alkoxy, halogen, and heteroaryl substituents, making it highly versatile for synthesizing complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting cutting-edge synthetic methodologies to maintain a competitive edge in the global marketplace. Our team of expert process chemists has extensively evaluated this nickel-catalyzed carbonylation route and confirmed its potential for generating high-purity thioester intermediates essential for modern drug discovery. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical clients.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can accelerate your development timelines while maximizing value.
