Advanced Synthesis of 2-Methylene-1-Indanone Derivatives for Commercial API Production
The pharmaceutical industry continuously seeks efficient pathways to construct privileged scaffolds that serve as the backbone for novel therapeutics. Patent CN112961086A introduces a groundbreaking methodology for the synthesis of 2-methylene-1-indanone derivatives, a structural motif prevalent in numerous bioactive natural products and approved drugs. This technology leverages a transition metal-catalyzed intermolecular oxidative coupling reaction between S,S-dithioketene compounds and diazo compounds to construct the indanone core in a single step. Unlike traditional multi-step condensations, this approach offers a direct route to highly functionalized intermediates essential for developing topoisomerase inhibitors and acetylcholinesterase inhibitors. For R&D teams focused on accelerating lead optimization, this patent represents a significant leap forward in synthetic efficiency and structural diversity.
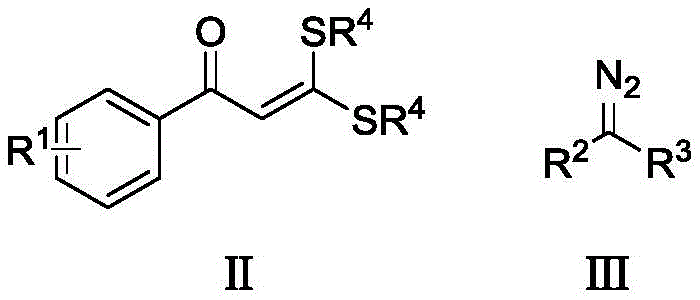
The limitations of conventional methods for synthesizing indanone derivatives have long hindered rapid scale-up and cost-effective manufacturing. Historical approaches, such as those disclosed in CN1277809C, often rely on cumbersome sequences involving bromination, amination, and selective reduction using hazardous reagents like lithium aluminum hydride. These multi-step processes not only suffer from low overall yields due to cumulative losses at each stage but also generate substantial chemical waste. Furthermore, alternative methods reported in CN108675921A require the pre-synthesis of the indanone framework under harsh oxidative conditions, which limits functional group tolerance and poses safety risks due to the release of toxic byproducts like methyl mercaptan. Such inefficiencies create bottlenecks in the supply chain, increasing both the lead time and the cost of goods for critical API intermediates.
In stark contrast, the novel approach detailed in this patent utilizes a rhodium-catalyzed oxidative coupling strategy that fundamentally simplifies the synthetic landscape. By reacting readily available S,S-dithioketene acetals with diazo compounds, the method constructs the five-membered indanone ring and installs the exocyclic methylene group simultaneously. This one-pot transformation operates under relatively mild thermal conditions, typically around 110 °C, and employs a robust catalyst system comprising [Cp*RhCl2]2 and AgSbF6. The reaction exhibits exceptional atom economy and functional group compatibility, tolerating electron-donating and electron-withdrawing substituents on the aromatic ring without the need for protecting groups. This streamlined workflow drastically reduces the number of unit operations required, directly translating to lower operational expenditures and a smaller environmental footprint for commercial production.
Mechanistic Insights into Rh(III)-Catalyzed Oxidative Coupling Cyclization
The success of this synthesis hinges on the unique reactivity of the cationic rhodium(III) species generated in situ. The catalytic cycle likely initiates with the coordination of the rhodium center to the sulfur atoms of the S,S-dithioketene substrate, facilitating the activation of the adjacent carbon center. Subsequently, the diazo compound undergoes decomposition to form a reactive rhodium-carbene intermediate. This electrophilic carbene species then engages in an insertion or coupling reaction with the activated dithioketene, followed by an intramolecular cyclization event that closes the five-membered ring. The presence of silver salts, specifically AgSbF6, serves to abstract chloride ligands from the rhodium precursor, generating the active cationic catalyst, while AgOAc acts as a terminal oxidant to regenerate the active Rh(III) species from any reduced Rh(I) forms, ensuring catalytic turnover. This sophisticated interplay between the transition metal, oxidant, and substrates allows for the precise construction of the complex 2-methylene-1-indanone skeleton with high regioselectivity.
From an impurity control perspective, the mechanism offers distinct advantages over acid or base-catalyzed condensations. The transition metal catalysis provides a specific pathway that minimizes non-selective polymerization of the diazo compound or hydrolysis of the sensitive dithioketal moiety. The use of mild basic additives like LiOAc helps to neutralize acidic byproducts without promoting the decomposition of the ester or ketone functionalities present in the R2 and R3 positions. Consequently, the crude reaction mixtures are cleaner, simplifying the downstream purification process. The ability to tolerate diverse substituents, such as halogens and alkoxy groups, without side reactions ensures that the final product meets stringent purity specifications required for pharmaceutical applications, reducing the burden on analytical quality control teams.
How to Synthesize 2-Methylene-1-Indanone Derivatives Efficiently
The experimental protocol outlined in the patent provides a robust framework for laboratory and pilot-scale synthesis. The procedure involves charging a reaction vessel with the S,S-dithioketene substrate and the diazo compound in a molar ratio of approximately 1:2 to drive the reaction to completion. The solvent system, preferably toluene or 1,2-dichloroethane, is selected to solubilize the organic substrates while withstanding the reaction temperature of 110 °C. The addition of the catalyst system ([Cp*RhCl2]2 and AgSbF6) along with AgOAc and LiOAc initiates the transformative coupling process. Following a reaction period of 24 hours, the mixture is worked up via standard silica gel column chromatography to isolate the pure yellow solid or liquid product. This standardized approach ensures reproducibility and high yields across a broad range of substrates.
- Combine S,S-dithioketene compound and diazo compound in a solvent such as toluene or DCE with a Rh(III) catalyst system.
- Add oxidant (AgOAc) and basic additive (LiOAc) to facilitate the oxidative coupling cyclization reaction.
- Heat the mixture to 110 °C for 24 hours, then purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits. The primary advantage lies in the drastic simplification of the manufacturing process. By collapsing multiple synthetic steps into a single oxidative coupling reaction, the method significantly reduces the consumption of solvents, reagents, and energy. This consolidation eliminates the need for intermediate isolation and purification stages, which are often the most time-consuming and costly parts of chemical manufacturing. Consequently, the overall cost of production is substantially lowered, providing a competitive edge in the pricing of high-value pharmaceutical intermediates. The reliance on commercially available starting materials further enhances supply security, mitigating the risks associated with sourcing specialized or custom-synthesized precursors.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as lithium aluminum hydride, which was required in previous methods for reduction steps, leads to direct material cost savings. Furthermore, the high yield of the reaction, often exceeding 80 percent, maximizes the utilization of raw materials, minimizing waste disposal costs. The simplified workflow also reduces labor hours and equipment occupancy time, allowing for higher throughput in existing manufacturing facilities without the need for significant capital investment in new infrastructure.
- Enhanced Supply Chain Reliability: The robustness of the rhodium-catalyzed system ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream API manufacturers. The tolerance for various functional groups means that a single platform technology can be used to produce a wide library of derivatives, reducing the need for multiple dedicated production lines. This flexibility allows suppliers to respond rapidly to changing market demands and customize orders for specific drug development programs without lengthy process re-validation periods.
- Scalability and Environmental Compliance: The reaction conditions are amenable to scale-up, utilizing common industrial solvents like toluene and operating at temperatures that are easily managed in standard stainless steel reactors. The use of silver salts as oxidants, while requiring recovery protocols, avoids the generation of heavy metal waste streams associated with stoichiometric chromium or manganese oxidants. This alignment with green chemistry principles facilitates regulatory approval and supports corporate sustainability goals, making the process attractive for environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners evaluating this route for their supply chain.
Q: What are the primary advantages of this Rh-catalyzed method over traditional indanone synthesis?
A: This method eliminates the need for multi-step condensation and pre-functionalization of raw materials. It achieves one-step ring construction with high yields (up to 84%) under relatively mild conditions (110 °C), significantly reducing process complexity and waste generation compared to older methods requiring harsh reagents like LiAlH4.
Q: Can this synthesis protocol accommodate diverse functional groups for drug discovery?
A: Yes, the protocol demonstrates excellent substrate adaptability. It tolerates various substituents on the aromatic ring (R1), including halogens (F, Cl, Br), alkyl groups (methyl), and alkoxy groups (methoxy). Additionally, the ester and ketone groups at the R2/R3 positions can be varied, allowing for the rapid generation of diverse libraries for SAR studies.
Q: Is the resulting 2-methylene-1-indanone suitable for further derivatization?
A: Absolutely. The exocyclic double bond and the ketone functionality provide versatile handles for downstream chemistry. For instance, the patent demonstrates the successful conversion of the derivative into polysubstituted pyrazole compounds via reaction with hydrazine hydrate, highlighting its utility as a scaffold for building more complex bioactive molecules.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylene-1-Indanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the Rh-catalyzed oxidative coupling technology described in CN112961086A and is fully prepared to translate this academic innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from gram-scale discovery to multi-ton manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-methylene-1-indanone derivative delivered meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced synthetic route for your next drug development program. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us help you optimize your supply chain and accelerate your path to market with our reliable and cost-effective manufacturing solutions.
