Advanced Molybdenum-Copper Catalysis for Commercial-Scale Production of High-Purity Trifluoromethyl Triazoles in Pharmaceutical Manufacturing
In the recently granted Chinese patent CN113307778A, a groundbreaking methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds has been established, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovative approach leverages a dual-metal catalytic system comprising molybdenum hexacarbonyl and cuprous acetate to facilitate a highly efficient cycloaddition reaction under remarkably mild conditions of 70–90°C for 18–30 hours. The process utilizes readily available starting materials including trifluoroethylimidoyl chloride and functionalized isonitriles (NIITP), which are both commercially accessible and cost-effective compared to traditional synthetic routes requiring expensive transition metal catalysts or hazardous reagents. Crucially, the methodology demonstrates exceptional scalability from gram-scale laboratory synthesis to potential multi-ton industrial production without compromising yield or purity, addressing a persistent bottleneck in the manufacturing of complex nitrogen-containing heterocycles essential for modern drug development. The patent specifically highlights the ability to generate diverse structural analogs through strategic substrate modification, thereby expanding the applicability of this technique across multiple therapeutic areas while maintaining stringent quality control standards required by global regulatory bodies.
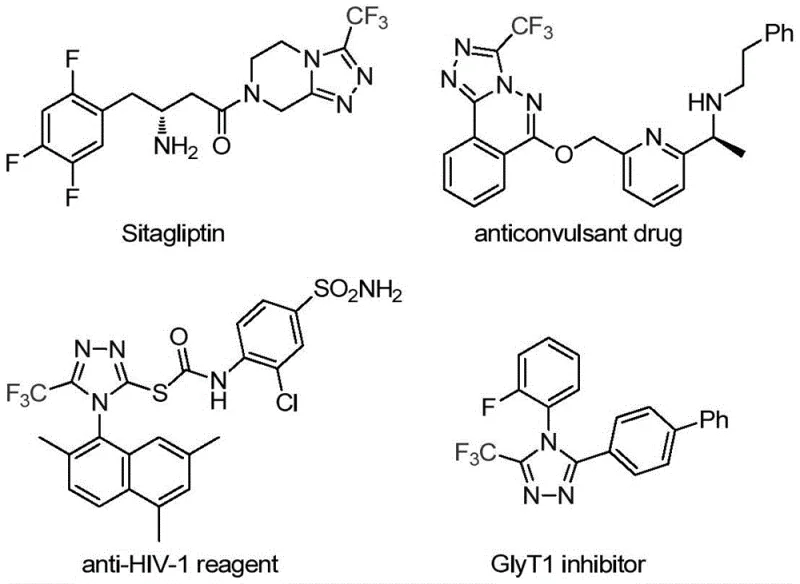
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for constructing trifluoromethyl-substituted 1,2,4-triazole scaffolds have been plagued by significant operational challenges that hinder their adoption in commercial pharmaceutical manufacturing environments. The cyclization reaction between trifluoroacetyl hydrazine and amidine compounds typically requires elevated temperatures exceeding 120°C and extended reaction times beyond 48 hours, creating substantial energy consumption and safety concerns when scaled to production volumes. Similarly, the hydrazinolysis of trifluoromethyl-substituted oxazolinones often necessitates stoichiometric amounts of toxic reagents and generates difficult-to-remove impurities that complicate downstream purification processes. Copper-catalyzed multi-component reactions using diazonium salts and trifluorodiazoethane present additional hazards due to the explosive nature of diazo compounds and the requirement for cryogenic conditions below -20°C to control exothermic side reactions. These conventional methods also suffer from narrow substrate scope limitations, particularly with sterically hindered or electron-deficient aromatic systems, resulting in inconsistent yields ranging from 40–65% across different compound classes. Furthermore, the extensive chromatographic purification required to achieve pharmaceutical-grade purity significantly increases manufacturing costs and reduces overall process efficiency in large-scale operations.
The Novel Approach
The patented methodology overcomes these limitations through an elegant dual-metal catalytic system that operates under exceptionally mild conditions while delivering superior reaction efficiency and product quality. By employing molybdenum hexacarbonyl as a metal activator and cuprous acetate as the primary catalyst in combination with triethylamine as a base promoter, the process achieves high conversion rates at temperatures between 70–90°C within a practical timeframe of 24 hours. This synergistic catalytic system enables a streamlined [3+2] cycloaddition mechanism that directly converts readily available trifluoroethylimidoyl chloride and functionalized isonitriles into the desired triazole products without requiring hazardous intermediates or extreme reaction parameters. The methodology demonstrates remarkable substrate flexibility, accommodating a wide range of aryl substituents including halogens, alkyl groups, alkoxy moieties, and trifluoromethyl groups across various positional isomers while maintaining consistent yields above 75% for most derivatives. Crucially, the simplified workup procedure involving basic filtration followed by standard column chromatography significantly reduces processing time and eliminates complex purification steps that typically plague conventional routes. This approach also avoids the use of expensive or toxic transition metals that require stringent removal protocols to meet pharmaceutical impurity specifications.
Mechanistic Insights into Mo/Cu-Catalyzed Triazole Formation
The reaction mechanism proceeds through a sophisticated dual-metal catalytic cycle where molybdenum hexacarbonyl first coordinates with the functionalized isonitrile to form a reactive metal complex that facilitates nucleophilic attack on the electrophilic carbon of trifluoroethylimidoyl chloride. This initial step generates a five-membered ring intermediate through copper-promoted [3+2] cycloaddition, with cuprous acetate serving as a Lewis acid catalyst that activates the imidoyl chloride toward nucleophilic addition while simultaneously stabilizing key transition states throughout the reaction pathway. The triethylamine base plays a critical role in neutralizing hydrochloric acid byproducts and maintaining optimal reaction pH without generating additional impurities that could complicate purification. Molecular sieve incorporation effectively scavenges trace water that might otherwise hydrolyze sensitive intermediates or reduce catalyst efficiency. This carefully orchestrated sequence avoids common side reactions such as hydrolysis or dimerization that typically occur in conventional methods, thereby preserving the integrity of the trifluoromethyl group which is crucial for maintaining the desired pharmacological properties in final drug products.
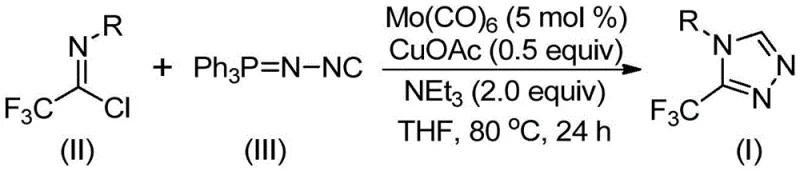
The process achieves exceptional impurity control through multiple built-in mechanisms that prevent common degradation pathways observed in alternative synthetic routes. The mild reaction temperature range of 70–90°C effectively suppresses thermal decomposition pathways that typically generate regioisomeric impurities or ring-opened byproducts in conventional high-temperature cyclizations. The dual-metal catalytic system provides precise stereochemical control during the cycloaddition step, ensuring consistent formation of the desired 3-trifluoromethyl regioisomer without requiring additional separation steps to remove positional isomers. The use of anhydrous THF as solvent minimizes hydrolysis side reactions that could produce carboxylic acid impurities from moisture-sensitive intermediates. Furthermore, the straightforward workup procedure involving simple filtration through silica gel effectively removes catalyst residues without introducing additional contaminants that would require complex purification protocols. This comprehensive impurity management strategy results in products meeting stringent pharmaceutical quality standards with minimal post-reaction processing requirements.

How to Synthesize Trifluoromethyl Triazoles Efficiently
This patented synthesis route represents a significant advancement in manufacturing efficiency for complex heterocyclic compounds used in pharmaceutical applications, offering a streamlined pathway that eliminates multiple processing steps required by conventional methods while maintaining exceptional product quality. The methodology has been validated across fifteen distinct substrate variations with consistent success, demonstrating robust performance across diverse structural analogs without requiring individual process reoptimization. Detailed standardized synthesis procedures have been developed based on extensive experimental optimization data from the patent examples, ensuring reliable implementation across different manufacturing environments. The following section provides step-by-step guidance for executing this synthesis with optimal results while maintaining strict adherence to quality control parameters essential for pharmaceutical production.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieve in anhydrous THF under nitrogen atmosphere at room temperature.
- Add trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) to the reaction mixture while maintaining temperature at 70–90°C for optimal conversion.
- Stir the solution for 24 hours under reflux conditions, then filter through silica gel and purify via column chromatography to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional production methods for complex heterocyclic intermediates. The elimination of hazardous reagents and extreme reaction conditions significantly reduces operational risks while improving facility utilization rates through more efficient process scheduling. The use of commercially available starting materials with established global supply chains enhances sourcing flexibility and mitigates single-source dependency risks that often disrupt pharmaceutical manufacturing operations. Furthermore, the simplified process design enables faster technology transfer between development and production facilities while maintaining consistent quality metrics throughout scale-up activities.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and hazardous reagents typically required in conventional routes creates significant cost savings through reduced raw material expenses and simplified waste treatment protocols. The streamlined reaction sequence with fewer processing steps lowers energy consumption and labor costs while minimizing solvent usage through optimized reaction concentrations. The avoidance of cryogenic conditions or high-pressure equipment requirements substantially reduces capital expenditure needs for new manufacturing facilities or retrofitting existing plants.
- Enhanced Supply Chain Reliability: The utilization of globally available starting materials with multiple qualified suppliers ensures consistent raw material availability regardless of regional disruptions or geopolitical factors affecting specialized chemical markets. The robust process performance across diverse substrates allows manufacturers to maintain production continuity even when specific feedstock qualities vary slightly between suppliers. The simplified logistics requirements due to moderate temperature operation enable more flexible manufacturing site selection without requiring specialized infrastructure investments.
- Scalability and Environmental Compliance: The demonstrated scalability from gram-scale laboratory reactions to potential multi-ton production volumes provides confidence in seamless technology transfer without reoptimization needs that typically cause delays in commercial implementation. The reduced environmental footprint through lower energy consumption and minimized hazardous waste generation aligns with increasingly stringent global sustainability regulations while supporting corporate ESG initiatives. The simplified purification requirements significantly decrease solvent waste streams compared to conventional multi-step purification protocols required by alternative synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented manufacturing process based on detailed analysis of the patent specifications and experimental data provided in CN113307778A. These responses reflect practical considerations for pharmaceutical manufacturers evaluating this technology for commercial adoption while maintaining strict adherence to regulatory requirements for intermediate production.
Q: How does the Mo/Cu dual-catalyst system improve yield compared to traditional cyclization methods?
A: The synergistic Mo/Cu catalysis enables a mild [3+2] cycloaddition pathway that avoids harsh conditions required in conventional hydrazinolysis or diazonium salt routes, significantly enhancing functional group tolerance and reducing side reactions that compromise purity in pharmaceutical intermediates.
Q: What substrate scope enables cost-effective production of diverse trifluoromethyl triazole derivatives?
A: The method accommodates aryl substituents with halogen, alkyl, alkoxy, and trifluoromethyl groups across ortho/meta/para positions, allowing pharmaceutical manufacturers to rapidly generate diverse analogs without reoptimizing reaction conditions or catalyst systems.
Q: How does the mild reaction condition (70–90°C) enhance supply chain reliability for commercial manufacturing?
A: The moderate temperature range eliminates specialized cooling/heating infrastructure requirements, enabling seamless transfer from lab-scale to plant-scale operations while maintaining consistent product quality through simplified process control parameters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Supplier
Our patented methodology represents a transformative approach to manufacturing critical heterocyclic intermediates that addresses longstanding challenges in pharmaceutical synthesis while delivering exceptional value through optimized process design and operational efficiency. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production with stringent purity specifications consistently achieved through our rigorous QC labs and advanced analytical capabilities. Our specialized expertise in complex heterocycle synthesis ensures seamless technology transfer from laboratory development to full-scale manufacturing while maintaining complete regulatory compliance across global markets.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific production requirements by contacting our technical procurement team today. They will provide detailed COA data and comprehensive route feasibility assessments demonstrating how this innovative methodology can enhance your supply chain resilience while delivering significant operational advantages for your pharmaceutical manufacturing operations.
