Advanced Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial API Manufacturing
Introduction to Next-Generation Triazole Synthesis
The 1,2,4-triazole scaffold represents a cornerstone structure in modern medicinal chemistry, serving as a critical pharmacophore in a diverse array of bioactive molecules including antiviral agents like Maraviroc, antidiabetic drugs such as Sitagliptin, and iron chelators like Deferasirox. The strategic incorporation of a trifluoromethyl group onto this heterocyclic core further enhances metabolic stability and lipophilicity, properties that are highly sought after by R&D teams developing next-generation therapeutics. However, traditional synthetic routes often suffer from complex multi-step sequences, harsh reaction conditions, or reliance on scarce reagents. A significant breakthrough in this domain is detailed in patent CN113105402B, which discloses a robust preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. This innovation offers a streamlined pathway that bypasses the limitations of prior art, utilizing a non-metallic iodine promotion system that is both economically viable and operationally simple.
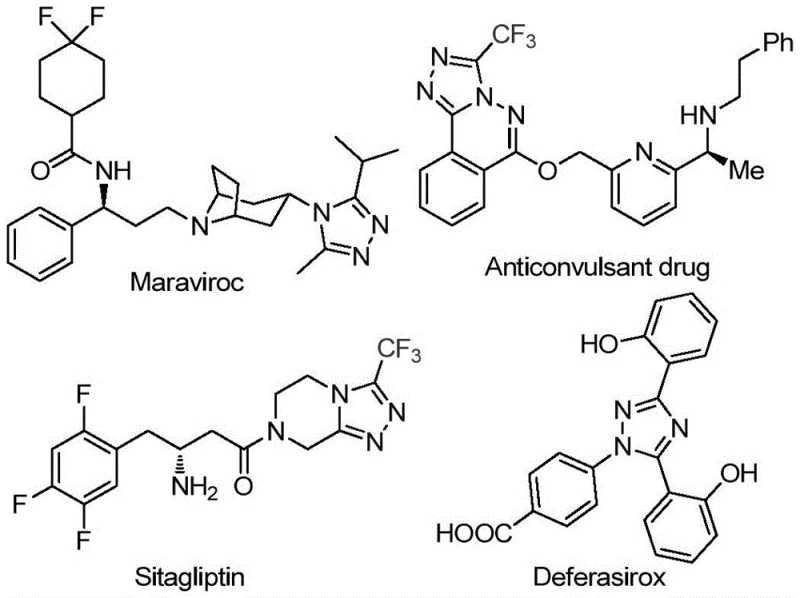
This novel methodology addresses the pressing need for efficient access to fluorinated heterocycles, which are increasingly vital in the design of high-performance active pharmaceutical ingredients (APIs). By leveraging a tandem oxidation-cyclization strategy, the process achieves high conversion rates while maintaining a clean impurity profile, a factor of paramount importance for regulatory compliance in pharmaceutical manufacturing. The ability to synthesize these complex architectures from readily available aryl ethanones and trifluoroethylimide hydrazides positions this technology as a game-changer for reliable pharmaceutical intermediate suppliers seeking to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazoles has been fraught with synthetic challenges that hinder cost reduction in API manufacturing. Conventional protocols frequently necessitate the use of expensive transition metal catalysts, such as copper or palladium complexes, which not only inflate raw material costs but also introduce significant downstream purification burdens due to stringent limits on residual heavy metals in final drug substances. Furthermore, many existing methods require rigorous anhydrous and oxygen-free environments, demanding specialized equipment and inert gas handling that complicate process safety and scalability. The reliance on pre-functionalized starting materials or unstable intermediates often leads to lower overall yields and generates substantial chemical waste, creating environmental and economic inefficiencies that supply chain heads strive to eliminate.
The Novel Approach
In stark contrast, the method described in patent CN113105402B introduces a metal-free paradigm shift by employing elemental iodine as a dual-function promoter for both oxidation and cyclization. This approach eliminates the need for toxic heavy metal catalysts entirely, thereby simplifying the workup procedure and ensuring a cleaner final product suitable for sensitive biological applications. The reaction proceeds in dimethyl sulfoxide (DMSO), a polar aprotic solvent that facilitates the in situ generation of aryl diketones via Kornblum oxidation, which then immediately engage in condensation with trifluoroethylimide hydrazide. This telescoped sequence reduces the number of isolation steps, minimizes solvent consumption, and operates under relatively mild thermal conditions without the need for exotic reagents, offering a distinct advantage for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this synthesis lies in its sequential activation of simple precursors through iodine-mediated transformations. Initially, the aryl ethanone undergoes an iodination followed by a Kornblum oxidation in the presence of DMSO and iodine at temperatures between 90°C and 110°C. This step generates a reactive aryl diketone species, which serves as the electrophilic partner for the subsequent ring-forming event. Upon the addition of trifluoroethylimide hydrazide, along with sodium dihydrogen phosphate and pyridine as bases, a dehydration condensation occurs to form a hydrazone intermediate. This intermediate is then poised for the critical intramolecular cyclization step.
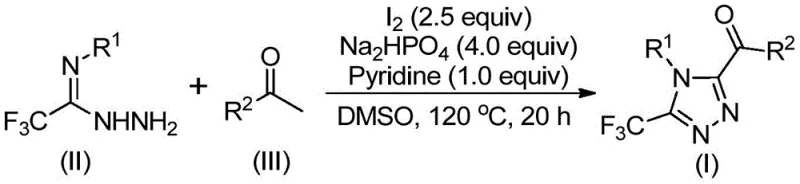
Under the continued influence of iodine and elevated temperatures ranging from 110°C to 130°C, the hydrazone undergoes oxidative cyclization to close the 1,2,4-triazole ring. The presence of the trifluoromethyl group on the hydrazide moiety is preserved throughout this rigorous process, demonstrating the robustness of the C-F bonds under these reaction conditions. The use of sodium dihydrogen phosphate acts as a buffer to maintain optimal pH levels, preventing the decomposition of sensitive intermediates while facilitating the elimination of water. This precise control over the reaction environment ensures high regioselectivity and minimizes the formation of side products, resulting in a superior impurity profile that meets the exacting standards of high-purity OLED material and pharmaceutical intermediate production.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazoles Efficiently
Implementing this synthesis requires careful attention to the stoichiometry and thermal profile to maximize yield and purity. The process is designed to be operationally straightforward, avoiding the need for glovebox techniques or ultra-dry solvents, which makes it accessible for standard pilot plant facilities. The following guide outlines the critical operational parameters derived from the patent data to ensure successful replication and optimization of the route.
- Oxidation Phase: React aryl ethanone with elemental iodine in DMSO at 90-110°C for 4-6 hours to generate the aryl diketone intermediate in situ.
- Cyclization Phase: Add trifluoroethylimide hydrazide, additional iodine, sodium dihydrogen phosphate, and pyridine. Heat to 110-130°C for 12-20 hours to complete the ring closure.
- Purification: Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodine-promoted synthesis offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials; aryl ethanones and elemental iodine are commodity chemicals available in bulk quantities from multiple global vendors, reducing the risk of supply disruption associated with specialized catalysts. This abundance ensures consistent availability and stabilizes pricing, allowing for more accurate long-term budget forecasting and cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts represents a significant direct cost saving, as there is no need to purchase expensive ligands or pay for specialized metal scavenging services during purification. Furthermore, the use of DMSO as a single solvent system throughout the reaction reduces solvent switching costs and waste disposal fees. The high atom economy of the tandem reaction means less raw material is wasted in side reactions, leading to a more efficient use of resources and a lower cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: By relying on stable, shelf-stable reagents that do not require cold chain logistics or inert atmosphere storage, the logistical burden on the supply chain is significantly reduced. The robustness of the reaction conditions means that production can be maintained even if minor fluctuations in environmental controls occur, ensuring consistent output and reducing lead time for high-purity pharmaceutical intermediates. This reliability is crucial for maintaining continuous manufacturing schedules and meeting tight delivery windows for downstream API producers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to proceed efficiently from milligram to gram scales without loss of performance, indicating a smooth path to multi-kilogram production. The absence of heavy metals simplifies wastewater treatment and aligns with increasingly stringent environmental regulations regarding metal discharge. This green chemistry aspect not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity, appealing to eco-conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis method. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide clarity for technical teams evaluating this route for potential adoption.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process utilizes elemental iodine as a non-metallic promoter, eliminating the need for costly palladium or copper catalysts and simplifying heavy metal removal.
Q: What are the critical reaction conditions for optimal yield?
A: The reaction requires dimethyl sulfoxide (DMSO) as the solvent to facilitate the Kornblum oxidation, with a temperature profile ranging from 90°C to 130°C across two stages.
Q: Is the method suitable for large-scale production?
A: Yes, the protocol avoids strict anhydrous or oxygen-free conditions and uses commercially available raw materials, making it highly amenable to industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of life-saving medications and high-performance materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this innovative iodine-promoted synthesis for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with reliable, high-quality chemical solutions.
