Advanced Microbial Hydroxylation for High-Purity HMG-CoA Reductase Inhibitor Intermediates
The pharmaceutical industry's relentless pursuit of efficient cholesterol-lowering therapies has placed significant emphasis on the optimization of HMG-CoA reductase inhibitor synthesis. Patent CN1240844C introduces a transformative biocatalytic approach that addresses long-standing bottlenecks in the production of these critical pharmaceutical intermediates. By leveraging specific non-sporulating and non-hyphal microorganisms, this technology enables the direct hydroxylation of statin precursors in aqueous media, bypassing the complexities associated with traditional fungal fermentation. This innovation represents a pivotal shift towards more robust and scalable bioprocesses, ensuring a reliable supply chain for high-value API ingredients while maintaining stringent purity standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the biocatalytic production of hydroxylated statin derivatives has relied heavily on filamentous fungi and actinomycetes, which present substantial engineering challenges during industrial scale-up. The mycelial growth characteristic of these organisms leads to a dramatic increase in broth viscosity, severely impeding oxygen transfer and creating heterogeneous mixing conditions within large-scale fermenters. To compensate for oxygen deficiency, operators must increase agitation speeds, which unfortunately risks shearing the delicate mycelial structures and reducing overall microbial activity. Furthermore, the inherent ability of these conventional strains to form spores poses a persistent contamination risk, as spores can easily disseminate throughout the facility and survive harsh cleaning conditions, threatening the sterility of subsequent batches and complicating facility maintenance protocols.
The Novel Approach
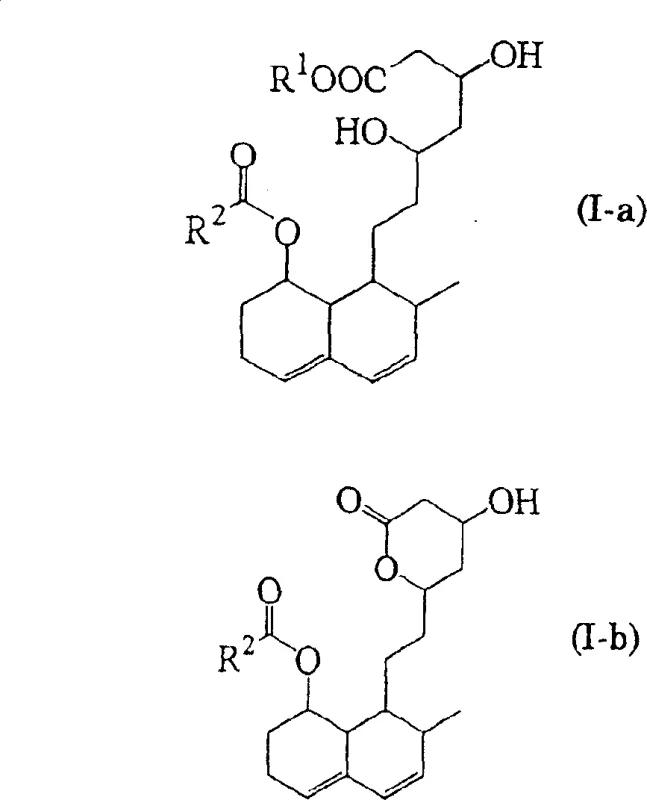
In stark contrast, the methodology disclosed in the patent utilizes a curated selection of microorganisms that lack both spore-forming capabilities and hyphal reproduction, fundamentally altering the rheological profile of the fermentation broth. By employing bacteria from genera such as Mycobacterium, Rhodococcus, and Gordona, the process maintains a low-viscosity environment that facilitates superior oxygen dissolution and uniform nutrient distribution without the need for excessive mechanical shear. This biological refinement not only enhances reaction efficiency but also drastically simplifies downstream processing, as the absence of dense mycelial mats allows for straightforward separation techniques. The core transformation involves the enzymatic hydroxylation of general formula (I-a) or its lactone (I-b) to yield the active hydroxylated forms (II-a) or (II-b), as illustrated in the reaction scheme below.
Mechanistic Insights into Microbial Side-Chain Hydroxylation
The enzymatic machinery within these non-sporulating bacteria exhibits remarkable regioselectivity and stereoselectivity, targeting specific carbon positions on the statin side chain to introduce the crucial hydroxyl group. This biotransformation occurs under mild aerobic conditions, typically between 20°C and 50°C, where the microbial enzymes function as highly specific oxidases or monooxygenases. The mechanism avoids the use of harsh chemical oxidants that often generate toxic byproducts or require complex protection-deprotection sequences, thereby streamlining the synthetic route. The patent highlights that the enzyme source can be the whole cultured cells, dried biomass, or even treated cell fractions, providing flexibility in how the biocatalyst is deployed in the reactor system to maximize turnover numbers.
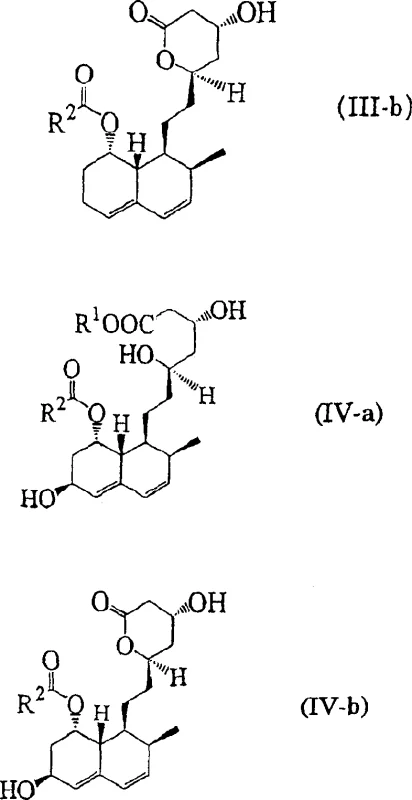
Furthermore, the structural integrity of the decalin ring system is preserved throughout the biocatalytic cycle, ensuring that the stereochemical configuration essential for HMG-CoA reductase inhibition remains intact. The process accommodates various substituents on the side chain, denoted as R1 and R2 in the general formulas, allowing for the synthesis of a diverse range of analogues including pravastatin and mevastatin derivatives. As shown in the specific examples below, the conversion of precursors like compound (V-a) to (VI-a) demonstrates the high fidelity of these bacterial strains in generating the desired pharmacophore without racemization or degradation of the sensitive lactone or acid moieties.
How to Synthesize Hydroxylated Statin Intermediates Efficiently
The implementation of this biocatalytic route requires precise control over fermentation parameters to ensure optimal enzyme expression and substrate conversion. The process begins with the cultivation of the selected bacterial strain in a nutrient-rich medium, followed by the introduction of the substrate either during the growth phase or as a resting cell bioconversion. Detailed operational guidelines regarding pH control, temperature gradients, and substrate feeding strategies are critical for achieving maximum yield. For a comprehensive understanding of the standardized operating procedures and specific strain deposition details, please refer to the technical guide below.
- Select a non-sporulating, non-hyphal microorganism such as Mycobacterium phlei or Rhodococcus erythropolis capable of hydroxylating the statin side chain.
- Cultivate the selected microorganism in an aerobic medium containing carbon and nitrogen sources at 20-50°C until sufficient biomass is achieved.
- Introduce the statin precursor (compound I-a or I-b) into the aqueous medium and maintain reaction conditions at pH 5.0-10.0 for 0.5 to 150 hours to accumulate the hydroxylated product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this non-sporulating bacterial platform offers profound advantages in terms of operational expenditure and supply chain resilience. The elimination of mycelial viscosity issues translates directly into reduced energy consumption for agitation and aeration, lowering the overall utility costs per kilogram of product. Additionally, the robust nature of the bacterial strains reduces the frequency of batch failures due to contamination or poor oxygen transfer, ensuring a more predictable production schedule that aligns with just-in-time manufacturing requirements.
- Cost Reduction in Manufacturing: The shift away from filamentous fungi eliminates the need for specialized high-shear mixing equipment and extensive filtration steps required to separate fungal biomass, resulting in significant capital and operational savings. By simplifying the downstream purification workflow through easier extraction and crystallization, manufacturers can reduce solvent usage and waste disposal costs, driving down the total cost of goods sold for these high-value intermediates.
- Enhanced Supply Chain Reliability: The use of non-sporulating organisms mitigates the risk of cross-contamination in multi-product facilities, allowing for faster turnaround times between campaigns and greater flexibility in production planning. The stability of the bacterial cultures ensures consistent performance over multiple generations, reducing the variability often seen with fungal strains and guaranteeing a steady supply of critical raw materials for API synthesis.
- Scalability and Environmental Compliance: The improved rheological properties of the bacterial fermentation broth facilitate seamless scale-up from laboratory to commercial volumes without the mass transfer limitations that plague fungal processes. Furthermore, the aqueous nature of the reaction and the avoidance of heavy metal catalysts align with green chemistry principles, simplifying environmental permitting and reducing the ecological footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this microbial hydroxylation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on strain selection and process parameters.
Q: Why are non-sporulating bacteria preferred over filamentous fungi for statin hydroxylation?
A: Non-sporulating bacteria eliminate the risk of spore contamination in production facilities and avoid the high viscosity issues associated with mycelial growth, leading to better oxygen transfer and reaction efficiency.
Q: What specific microorganisms are suitable for this hydroxylation process?
A: The patent identifies several effective genera including Mycobacterium, Rhodococcus, Gordona, Corynebacterium, and Arthrobacter, with specific strains like Mycobacterium phlei JCM5865 showing high conversion rates.
Q: How does this process impact the purification of the final intermediate?
A: The use of bacterial cultures simplifies downstream processing by reducing broth viscosity, allowing for easier extraction and crystallization compared to the complex filtration required for fungal mats.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable HMG-CoA Reductase Inhibitor Intermediate Supplier
As the global demand for cardiovascular therapeutics continues to rise, securing a dependable source of high-quality intermediates is paramount for pharmaceutical developers. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver these complex biocatalytic solutions with unmatched consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch meets the exacting standards required for regulatory submission and commercial launch.
We invite potential partners to engage with our technical procurement team to discuss how this advanced microbial hydroxylation platform can optimize your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into the economic benefits of switching to this bacterial process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines.
