Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Applications
Introduction to Advanced Triazole Synthesis Technology
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN113105402B discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a structural motif prevalent in high-value drugs such as Maraviroc, Sitagliptin, and Deferasirox. This innovation addresses the longstanding challenge of introducing both trifluoromethyl and acyl groups into the triazole core simultaneously, a modification known to enhance metabolic stability and lipophilicity. By leveraging a non-metal promoted strategy, this technology offers a streamlined pathway that bypasses the complexities associated with traditional transition metal catalysis.
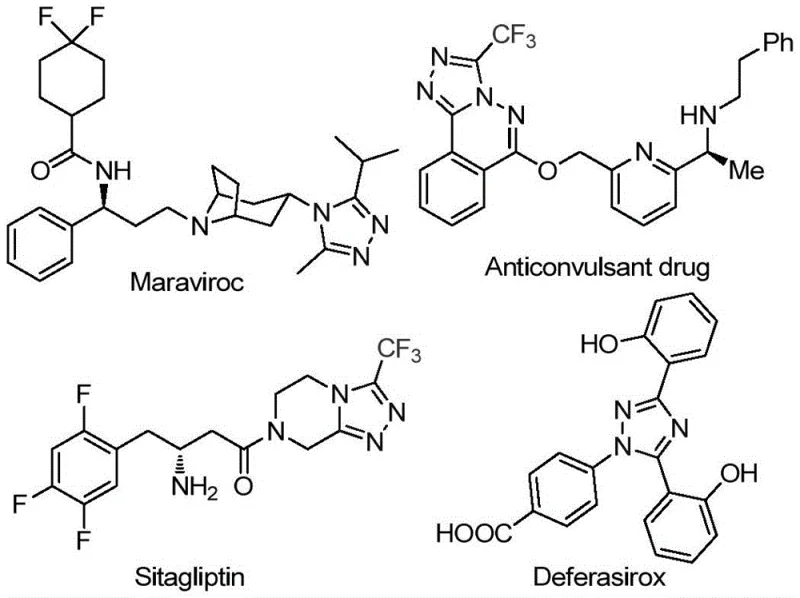
The significance of this development extends beyond academic interest, providing a practical solution for reliable pharmaceutical intermediate supplier networks aiming to optimize their supply chains. The method utilizes inexpensive aryl ethanones and trifluoroethylimide hydrazides as starting materials, ensuring that the cost of goods sold (COGS) remains competitive even at commercial volumes. Furthermore, the operational simplicity, which eliminates the need for rigorous anhydrous or oxygen-free environments, drastically reduces the infrastructure requirements for manufacturing facilities. This positions the technology as a key enabler for cost reduction in API manufacturing, allowing producers to allocate resources more efficiently while maintaining high purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles has relied heavily on transition metal catalysts or harsh reaction conditions that pose significant challenges for industrial scalability. Conventional routes often require expensive palladium or copper catalysts, which not only inflate raw material costs but also introduce complex purification steps to remove trace heavy metal residues to meet stringent regulatory limits. Additionally, many existing methods suffer from limited substrate scope, failing to accommodate diverse functional groups without compromising yield or selectivity. The necessity for strictly anhydrous and oxygen-free conditions in these traditional protocols further complicates the engineering controls required for large-scale reactors, increasing both capital expenditure and operational risk.
The Novel Approach
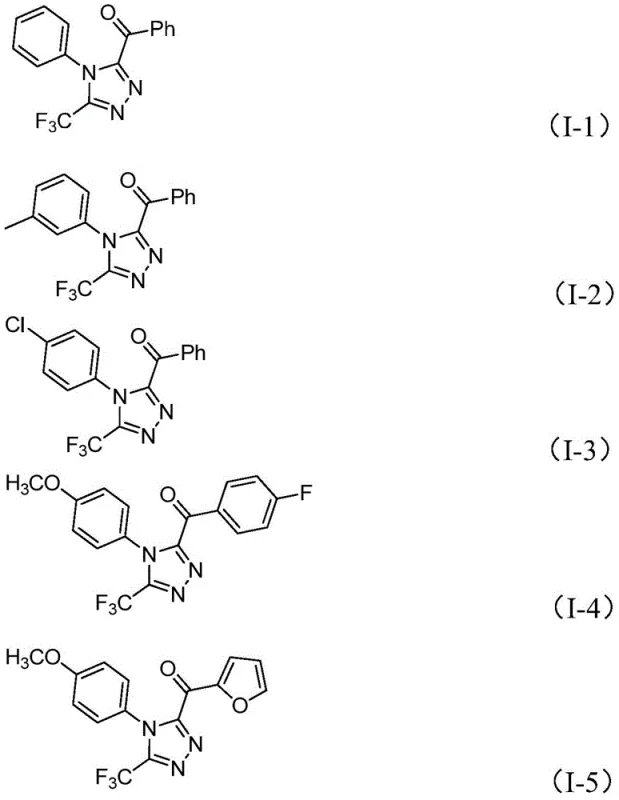
In stark contrast, the novel approach detailed in the patent employs a metal-free, iodine-promoted cyclization strategy that fundamentally simplifies the synthetic landscape. By utilizing elemental iodine and dimethyl sulfoxide (DMSO) to facilitate a tandem Kornblum oxidation and cyclization sequence, the process achieves high efficiency without the burden of heavy metal contamination. This methodology demonstrates exceptional functional group tolerance, successfully synthesizing derivatives with various substituents such as methyl, methoxy, chloro, and trifluoromethyl groups on the aromatic rings. The ability to produce compounds like I-1 through I-5 with consistent quality highlights the versatility of this route, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Iodine-Promoted Cyclization
The core of this innovative synthesis lies in a sophisticated cascade reaction mechanism initiated by the interaction between aryl ethanone and iodine in DMSO. The process begins with an iodination followed by a Kornblum oxidation, converting the aryl ethanone into an aryl diketone intermediate in situ. This reactive species then undergoes a dehydration condensation with trifluoroethylimide hydrazide to form a hydrazone intermediate. Subsequently, under the synergistic promotion of iodine and base (sodium dihydrogen phosphate/pyridine), an intramolecular cyclization occurs to construct the 1,2,4-triazole ring system. This mechanistic pathway is highly efficient, minimizing side reactions and maximizing atom economy compared to stepwise traditional syntheses.
From a quality control perspective, this mechanism offers distinct advantages in impurity profile management. The absence of transition metals eliminates the risk of metal-catalyzed decomposition or isomerization often seen in sensitive fluorinated compounds. Furthermore, the use of DMSO as both solvent and oxidant ensures a homogeneous reaction environment that promotes consistent conversion rates across different batches. The final products, characterized by rigorous NMR and HRMS analysis, exhibit high structural integrity with minimal byproduct formation. This level of control is crucial for R&D directors who require high-purity intermediates to ensure the safety and efficacy of the final drug substance, reducing the need for extensive downstream purification.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazoles Efficiently
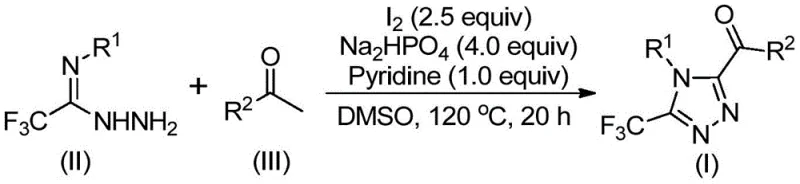
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity. The process involves a two-stage heating protocol where the initial oxidation is conducted at moderate temperatures before introducing the hydrazide component for the cyclization step. Operators must carefully monitor the stoichiometry of iodine and base to ensure complete conversion without excessive halogenation. The following guide outlines the standardized operational procedure derived from the patent data, ensuring reproducibility from laboratory to pilot plant scales.
- Combine aryl ethanone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis represents a strategic opportunity to enhance supply security and reduce overall manufacturing costs. The reliance on commodity chemicals such as aryl ethanones and elemental iodine mitigates the risk of supply chain disruptions often associated with specialized catalysts or exotic reagents. Moreover, the simplified workup procedure, which involves basic filtration and standard column chromatography, significantly shortens the production cycle time. This efficiency translates directly into improved throughput and reduced inventory holding costs, providing a competitive edge in the fast-paced pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a major cost driver from the bill of materials. Additionally, the avoidance of rigorous inert atmosphere conditions reduces energy consumption and equipment maintenance costs associated with gloveboxes or specialized nitrogen purging systems. The use of cheap and widely available starting materials further drives down the variable cost per kilogram, allowing for substantial cost savings that can be passed on to customers or reinvested in R&D.
- Enhanced Supply Chain Reliability: Since the key raw materials are bulk chemicals with established global supply networks, the risk of raw material shortages is minimized. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental parameters, ensuring consistent batch-to-batch quality. This reliability is essential for maintaining continuous supply to downstream API manufacturers, preventing costly production stoppages and ensuring timely delivery of critical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-gram levels with clear pathways to ton-scale production. The absence of heavy metals simplifies waste stream treatment, reducing the environmental footprint and compliance costs associated with hazardous waste disposal. This aligns with modern green chemistry principles, making the facility more attractive to environmentally conscious partners and regulators while ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for potential licensing or contract manufacturing opportunities.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process utilizes elemental iodine as a promoter instead of toxic or expensive heavy metal catalysts, significantly reducing raw material costs and simplifying waste treatment.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction proceeds in dimethyl sulfoxide (DMSO) at temperatures between 90°C and 130°C. It does not require strict anhydrous or oxygen-free conditions, making it operationally simple.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method can be easily expanded from gram-level laboratory synthesis to industrial scale production due to the use of cheap, readily available starting materials and simple post-processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole delivered meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage this cost-effective and scalable synthetic route for your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this technology can optimize your budget. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your path to market with confidence and precision.
