Advanced Metal-Free Synthesis of Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Introduction: Revolutionizing Triazole Scaffold Construction
The 1,2,4-triazole ring system represents a cornerstone structural motif in modern medicinal chemistry, serving as a critical pharmacophore in a vast array of bioactive molecules. As illustrated in the provided structural analysis, prominent pharmaceutical agents such as Maraviroc, Sitagliptin, and Deferasirox rely heavily on this nitrogen-containing five-membered heterocycle for their therapeutic efficacy. The strategic incorporation of a trifluoromethyl group into this scaffold further amplifies its utility by enhancing electronegativity, metabolic stability, and membrane permeability, properties that are essential for optimizing drug candidates. However, traditional synthetic routes to access these complex 3,4,5-trisubstituted derivatives often suffer from significant limitations, including the reliance on scarce reagents or harsh reaction conditions. Patent CN113105402B introduces a transformative approach to this challenge, detailing a robust preparation method that utilizes inexpensive aryl ethanones and trifluoroethylimine hydrazides as starting materials.
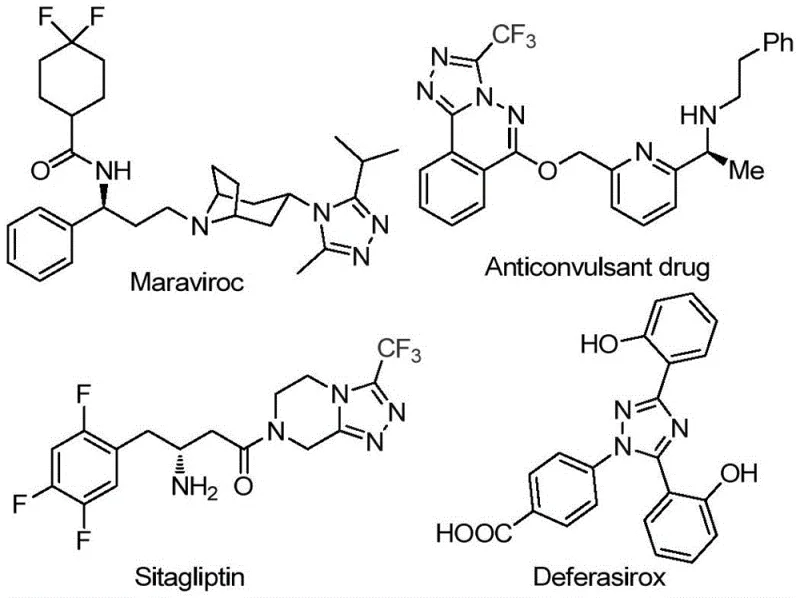
This novel methodology eliminates the necessity for stringent anhydrous or oxygen-free environments, thereby lowering the barrier for entry for process chemists and manufacturing teams alike. By leveraging a non-metallic iodine promotion strategy within a dimethyl sulfoxide solvent system, this invention provides a streamlined pathway to high-value intermediates. For R&D directors and procurement specialists seeking reliable sources for complex heterocyclic building blocks, understanding the nuances of this metal-free catalytic system is paramount. It represents a shift towards greener, more cost-effective chemistry that does not compromise on the structural complexity required for high-performance active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, particularly those bearing both acyl and trifluoromethyl functionalities, has been a formidable challenge in organic synthesis. Conventional literature often describes methods that rely heavily on transition metal catalysis, which introduces a myriad of downstream complications for process development. The presence of heavy metals such as palladium, copper, or rhodium necessitates rigorous purification protocols to meet the strict residual metal limits imposed by regulatory bodies like the FDA and EMA. Furthermore, many existing protocols require specialized ligands that are prohibitively expensive and difficult to source in bulk quantities, creating bottlenecks in the supply chain. Additionally, traditional cyclization strategies frequently demand strictly anhydrous conditions and inert atmospheres, requiring specialized equipment and increasing the operational expenditure (OPEX) of the manufacturing process. These factors collectively contribute to higher production costs and longer lead times, making the commercial scale-up of complex polymer additives or pharmaceutical intermediates less attractive from a business perspective.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN113105402B offers a paradigm shift by employing a simple, non-metallic iodine-promoted system. This approach utilizes elemental iodine, a commodity chemical that is not only inexpensive but also easy to handle and dispose of compared to heavy metal salts. The reaction proceeds through a tandem sequence involving an initial iodination and Kornblum oxidation of the aryl ketone, followed by a condensation and cyclization with the hydrazide derivative. Crucially, this process tolerates a wide range of functional groups on both the aryl ketone and the hydrazide components, allowing for the rapid generation of diverse chemical libraries without the need for protecting group strategies. The ability to conduct the reaction in dimethyl sulfoxide (DMSO) without the need for rigorous drying or degassing significantly simplifies the operational workflow. This robustness translates directly into enhanced process safety and reduced downtime, addressing key pain points for supply chain heads who prioritize continuity and reliability in their vendor partnerships.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this synthesis lies in its dual-function utilization of iodine and DMSO to drive the formation of the triazole ring. The reaction initiates with the activation of the aryl ethanone by molecular iodine in the presence of DMSO. This step facilitates an alpha-iodination followed by a Kornblum oxidation, effectively converting the methyl ketone moiety into a reactive 1,2-dicarbonyl or alpha-keto species in situ. This oxidative transformation is critical as it generates the electrophilic center necessary for the subsequent nucleophilic attack. Once the oxidized intermediate is formed, it undergoes a dehydration condensation with the trifluoroethylimine hydrazide. This step forms a hydrazone intermediate, which serves as the precursor for the final ring closure. The presence of sodium dihydrogen phosphate and pyridine acts as a buffering system, maintaining the optimal pH balance to facilitate proton transfer events without degrading the sensitive trifluoromethyl group.
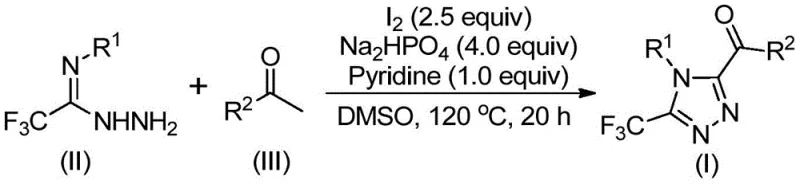
Following the formation of the hydrazone, the system undergoes an intramolecular cyclization promoted by the continued presence of iodine and the basic environment provided by the additives. This cyclization step constructs the 1,2,4-triazole core while simultaneously installing the acyl group at the 3-position and the trifluoromethyl group at the 5-position. The mechanism avoids the formation of stable metal-complex intermediates, which are often the source of difficult-to-remove impurities in transition metal-catalyzed reactions. Instead, the byproducts are primarily inorganic salts and reduced iodine species, which can be easily removed during the aqueous workup or silica gel filtration. This clean reaction profile ensures a superior impurity spectrum, a metric that is closely monitored by quality control laboratories to ensure batch-to-batch consistency. The tolerance for various substituents, including halogens, alkoxy groups, and alkyl chains on the aromatic rings, further demonstrates the versatility of this mechanistic pathway, making it applicable to a broad scope of substrate variations required for structure-activity relationship (SAR) studies.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the iodine promoter and the thermal profile of the two-stage reaction. The process is designed to be operationally simple, yet precise control over the addition sequence and temperature ramps is essential to maximize yield and minimize side reactions. The initial oxidation phase must be allowed to proceed to completion before the introduction of the hydrazide component to prevent competitive reactions. Detailed standard operating procedures (SOPs) regarding the specific molar ratios of sodium dihydrogen phosphate to pyridine are critical for buffering the reaction medium effectively. For a comprehensive guide on executing this protocol in a pilot plant or laboratory setting, please refer to the standardized synthesis steps outlined below.
- Oxidation Phase: React aryl ethyl ketone with elemental iodine in DMSO at 90-110°C for 4-6 hours to generate the aryl diketone intermediate via Kornblum oxidation.
- Cyclization Phase: Add trifluoroethylimide hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine to the mixture, then heat to 110-130°C for 12-20 hours.
- Purification: Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity 3,4,5-trisubstituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial standpoint, the adoption of this iodine-promoted synthesis offers substantial strategic advantages for organizations focused on cost reduction in pharmaceutical intermediate manufacturing. The primary driver of value is the complete elimination of precious metal catalysts. By removing the dependency on palladium or other rare earth metals, manufacturers can insulate themselves from the volatile pricing fluctuations characteristic of the precious metals market. This substitution not only lowers the direct material costs but also drastically simplifies the downstream processing requirements. The absence of heavy metals means that expensive scavenging resins or complex extraction protocols are no longer necessary, leading to a significant reduction in waste generation and disposal costs. Furthermore, the use of commodity chemicals like elemental iodine and DMSO ensures a stable and secure supply chain, as these reagents are produced globally in massive quantities and are not subject to the same geopolitical supply risks as specialized catalytic ligands.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, extending beyond simple reagent costs. By utilizing a metal-free system, the process avoids the capital expenditure associated with dedicated metal-removal units and the ongoing operational costs of purchasing metal scavengers. The reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors without the need for exotic materials of construction. Additionally, the high atom economy of the tandem reaction minimizes the generation of organic waste, aligning with green chemistry principles that are increasingly mandated by environmental regulations. This efficiency translates into a lower cost of goods sold (COGS), providing procurement managers with greater flexibility in negotiating contracts and improving overall profit margins for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is a top priority for global pharmaceutical companies, and this synthesis method directly contributes to that goal. The starting materials, specifically aryl ethanones and trifluoroethylimine hydrazides, are widely available from multiple chemical suppliers, reducing the risk of single-source dependency. The robustness of the reaction, which does not require stringent exclusion of moisture or oxygen, reduces the likelihood of batch failures due to environmental excursions. This reliability ensures consistent delivery schedules and reduces the need for safety stock inventory. Moreover, the scalability of the process from gram to kilogram levels allows for seamless technology transfer from R&D to commercial production, ensuring that supply can be ramped up quickly to meet market demand without extensive process re-optimization.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of synthetic processes has become a critical evaluation criterion. This iodine-promoted method generates significantly less hazardous waste compared to traditional heavy metal-catalyzed routes. The solvents and reagents used are easier to recycle or treat in standard wastewater facilities, facilitating compliance with strict environmental discharge standards. The simplicity of the workup procedure, involving filtration and standard chromatography, reduces the energy consumption associated with distillation and solvent recovery. This alignment with sustainable manufacturing practices not only mitigates regulatory risk but also enhances the corporate social responsibility (CSR) profile of the supply chain, a factor that is increasingly important for stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a non-metal promoter, completely avoiding the need for costly and toxic heavy metal catalysts like palladium or copper, which simplifies purification and reduces environmental impact.
Q: What are the key advantages of introducing a trifluoromethyl group in this scaffold?
A: Introducing a trifluoromethyl group significantly enhances the physicochemical properties of the parent compound, including improved metabolic stability, lipophilicity, and bioavailability, making these intermediates highly valuable for developing next-generation pharmaceuticals.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It uses cheap, commercially available raw materials, operates without strict anhydrous or oxygen-free conditions, and has been demonstrated to easily expand from gram-level laboratory synthesis to larger commercial batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of life-saving medications. Our team of expert process chemists has extensively analyzed advanced synthetic methodologies, including the metal-free iodine-promoted cyclization described in recent intellectual property, to optimize our own production capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of even the largest multinational pharmaceutical partners. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole we deliver meets the highest industry standards for impurity profiles and physical characteristics.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Whether you are in the early stages of drug discovery or preparing for commercial launch, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your molecule. We encourage you to contact us directly to request specific COA data for our catalog compounds or to discuss route feasibility assessments for custom synthesis projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner committed to innovation, quality, and long-term value creation.
