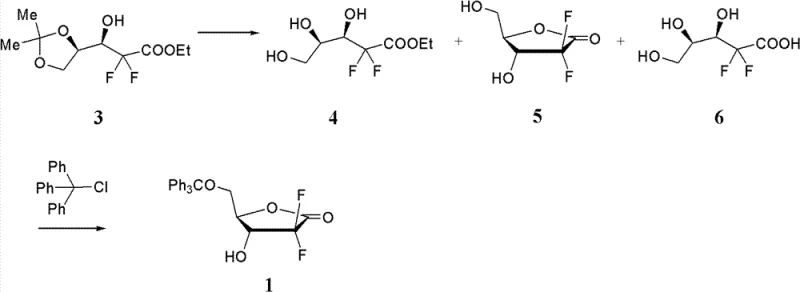
Advanced Stereoselective Synthesis of Gemcitabine Intermediates for Commercial Scale-up
The pharmaceutical industry continuously seeks robust methodologies for the production of critical anticancer agents, and the synthesis of beta-nucleosides represents a cornerstone in this endeavor. Patent CN103087132A discloses a groundbreaking stereoselective method for synthesizing 2'-deoxy-2,2'-difluorocytidine, widely known as gemcitabine, which is pivotal for treating viral infections and various forms of cancer. This technology addresses the longstanding challenge of controlling stereochemistry at the anomeric center, a factor that directly dictates the therapeutic efficacy and safety profile of the final drug substance. By employing a novel oxidative coupling strategy, the process circumvents the limitations of traditional glycosylation techniques that often yield unfavorable mixtures of stereoisomers. For R&D Directors and Procurement Managers, understanding this patent is crucial as it offers a pathway to higher purity intermediates with reduced processing complexity. The method involves reacting a specifically designed tetrahydrofuran compound with a nucleobase derivative in the presence of an oxidizing agent, ensuring that the resulting beta-nucleoside is formed as the predominant product. This innovation not only enhances the chemical efficiency of the synthesis but also aligns with the stringent quality requirements demanded by global regulatory bodies for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2'-deoxynucleosides has been plagued by the difficulty in achieving high stereoselectivity, often resulting in significant quantities of the inactive alpha-anomer alongside the desired beta-form. Conventional routes typically rely on direct coupling reactions that lack sufficient stereocontrol, necessitating extensive and costly purification steps such as repeated crystallization or chromatographic separation to isolate the therapeutically active isomer. These traditional methods frequently suffer from low overall yields due to the loss of material during separation, and the presence of stubborn impurities can compromise the safety profile of the final API. Furthermore, many older processes utilize harsh reaction conditions or expensive catalysts that are difficult to remove completely, posing risks for residual metal contamination in the final product. The inability to consistently produce high-purity beta-nucleosides at scale has been a persistent bottleneck for supply chain managers, leading to volatility in availability and increased costs for downstream drug manufacturers. The reliance on non-selective pathways means that a substantial portion of raw materials is effectively wasted, creating an unsustainable economic model for large-scale production of complex oncology intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN103087132A introduces a sophisticated oxidative coupling mechanism that inherently favors the formation of the beta-anomer. This novel approach utilizes a tetrahydrofuran intermediate bearing a specific leaving group and an ester functionality at the C-3 position, which plays a critical role in directing the stereochemical outcome of the reaction. By introducing an oxidizing agent into the reaction mixture, the process generates a reactive iodonium species in situ, which is then stabilized by the neighboring ester group to form a cyclic onium intermediate. This transient structure effectively blocks one face of the sugar ring, forcing the nucleobase to attack from the opposite side with high precision. The result is a dramatic improvement in the beta-to-alpha ratio, significantly reducing the burden on downstream purification units. This method allows for the use of readily available oxidizing agents and standard organic solvents, making it highly adaptable for industrial scale-up without requiring exotic or prohibitively expensive reagents. The robustness of this chemistry ensures consistent batch-to-batch quality, a key metric for supply chain reliability in the pharmaceutical sector.
Mechanistic Insights into Oxidative Stereoselective Glycosylation
The core of this technological breakthrough lies in the detailed mechanistic pathway that governs the stereoselective formation of the glycosidic bond. The reaction proceeds through an SN1-like mechanism where the iodide leaving group is activated by the oxidizing agent to form a triiodide ion, which subsequently departs to generate a carbocation-like onium intermediate. Crucially, the ester group located at the C-3 position of the tetrahydrofuran ring participates in neighboring group assistance, stabilizing this positive charge through the formation of a six-membered cyclic onium species. This stabilization is not merely energetic but also steric, as the cyclic structure locks the conformation of the sugar ring and shields the bottom face from nucleophilic attack. Consequently, the silyl-protected nucleobase is compelled to approach the anomeric center from the top face, which is sterically less hindered, thereby ensuring the exclusive or predominant formation of the beta-configuration. This level of mechanistic control is rare in nucleoside chemistry and represents a significant advancement over methods that rely solely on thermodynamic equilibrium or non-specific catalysts. For technical teams, understanding this mechanism is vital for optimizing reaction parameters such as temperature and oxidant concentration to maximize the yield of the desired isomer while minimizing side reactions.
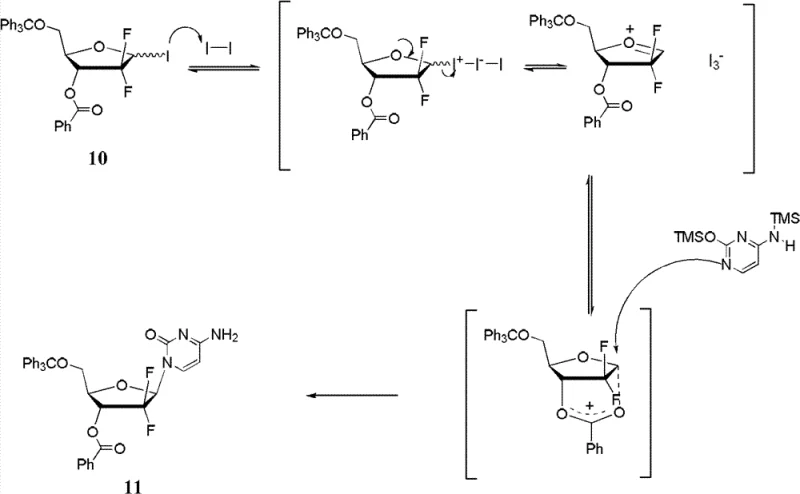
Impurity control is another critical aspect where this mechanism offers substantial advantages over conventional synthesis routes. In traditional processes, the formation of the alpha-anomer is often a competing pathway that is difficult to suppress without sacrificing overall conversion rates. However, in this oxidative system, the energetic barrier for the formation of the alpha-anomer is significantly raised due to the steric constraints imposed by the stabilized onium intermediate. This means that even if the starting iodide material exists as a mixture of anomers, the reaction pathway funnels both isomers towards the desired beta-product through the common onium intermediate. This convergent behavior simplifies the quality control of starting materials, as strict stereochemical purity of the precursor is less critical than in other methods. Furthermore, the use of specific oxidizing agents allows for fine-tuning the reaction kinetics, ensuring that the oxidative activation occurs at a rate that matches the nucleophilic attack, thereby preventing the accumulation of reactive byproducts. This inherent selectivity reduces the complexity of the impurity profile, making it easier to meet the stringent specifications required for pharmaceutical intermediates destined for clinical use.
How to Synthesize Gemcitabine Intermediates Efficiently
The practical implementation of this synthesis route involves a series of well-defined chemical transformations that begin with the preparation of a protected lactone precursor. This starting material is subjected to reduction and halogenation steps to generate the key iodide intermediate, which serves as the glycosyl donor in the subsequent coupling reaction. The detailed standardized synthesis steps see the guide below, which outlines the specific reagents, solvent systems, and temperature profiles required to achieve optimal results. Operators must pay close attention to the stoichiometry of the oxidizing agent, as this parameter directly influences the concentration of the active iodonium species and, consequently, the stereoselectivity of the coupling. The process is designed to be compatible with standard reactor equipment, utilizing common solvents like toluene or acetonitrile that are easily recovered and recycled in a commercial setting. By following this protocol, manufacturers can achieve high yields of the beta-nucleoside with minimal formation of difficult-to-remove impurities, streamlining the path from raw materials to the final active pharmaceutical ingredient.
- Prepare the 5-protected lactone intermediate from the corresponding hydroxy-valerate precursor using trityl chloride and acid treatment.
- Convert the lactone to a furanose derivative and subsequently transform it into an iodide compound using trialkylphosphine and iodine or mesylate displacement.
- React the iodide intermediate with a silyl-protected nucleobase in the presence of an oxidizing agent to stereoselectively form the beta-nucleoside.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis technology offers compelling advantages that directly address the pain points of procurement managers and supply chain heads in the pharmaceutical industry. The primary benefit lies in the significant cost reduction in manufacturing achieved through the simplification of the purification process. By inherently producing a high ratio of the desired beta-anomer, the need for extensive chromatographic separation or multiple recrystallization steps is drastically reduced, leading to lower solvent consumption and reduced waste disposal costs. This efficiency translates into a more favorable cost structure for the final intermediate, allowing for better margin management in a competitive market. Additionally, the reliance on common oxidizing agents and standard organic solvents mitigates the risk of supply chain disruptions associated with specialized or scarce reagents. The robustness of the reaction conditions ensures that the process can be scaled up from laboratory to commercial production with minimal re-optimization, providing supply chain leaders with the confidence to commit to long-term contracts. This stability is crucial for maintaining the continuity of supply for life-saving medications, where any interruption can have severe consequences for patients and healthcare providers globally.
- Cost Reduction in Manufacturing: The elimination of complex separation steps for alpha-anomer removal leads to substantial cost savings in processing time and material usage. By avoiding the need for expensive chiral catalysts or extensive purification trains, the overall cost of goods sold is significantly optimized. This efficiency allows manufacturers to offer competitive pricing without compromising on the quality or purity of the intermediate. The reduction in solvent usage also contributes to lower environmental compliance costs, further enhancing the economic viability of the process. Ultimately, this streamlined approach ensures that resources are allocated more effectively, maximizing the return on investment for production facilities.
- Enhanced Supply Chain Reliability: The use of widely available raw materials and reagents ensures that the supply chain is resilient against market fluctuations and geopolitical instabilities. Unlike processes that depend on proprietary catalysts or rare earth metals, this method utilizes commodity chemicals that can be sourced from multiple suppliers globally. This diversification of the supply base reduces the risk of single-source dependency and ensures that production schedules can be maintained even in the face of external disruptions. The robustness of the chemistry also means that batch failure rates are minimized, providing a consistent and reliable flow of materials to downstream customers. This reliability is a key differentiator for suppliers looking to establish long-term partnerships with major pharmaceutical companies.
- Scalability and Environmental Compliance: The reaction conditions are mild and compatible with large-scale reactor systems, facilitating a smooth transition from pilot plant to full commercial production. The process generates less hazardous waste compared to traditional methods, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The ability to recycle solvents and minimize waste output reduces the environmental footprint of the manufacturing operation, making it an attractive option for companies committed to green chemistry principles. This scalability ensures that supply can be rapidly ramped up to meet surges in demand without the need for significant capital investment in new infrastructure. Consequently, this technology supports a sustainable and agile supply chain capable of adapting to the dynamic needs of the global healthcare market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereoselective synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the operational and strategic benefits of adopting this method. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The information covers aspects ranging from reaction mechanisms to supply chain implications, offering a comprehensive overview of the technology's value proposition.
Q: How does the oxidizing agent improve stereoselectivity in gemcitabine synthesis?
A: The oxidizing agent facilitates the formation of an iodonium intermediate which is stabilized by the neighboring ester group at the C-3 position. This stabilization creates a specific steric environment that favors nucleophilic attack from the top face of the tetrahydrofuran ring, significantly increasing the ratio of the therapeutically active beta-anomer over the alpha-anomer.
Q: What are the advantages of this method over conventional nucleoside coupling?
A: Conventional methods often result in mixtures of alpha and beta anomers requiring difficult and yield-reducing separation steps. This novel approach leverages an SN1-like mechanism with neighboring group participation to inherently drive the reaction towards the desired beta-configuration, simplifying downstream purification and improving overall process efficiency.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the reaction conditions utilize common organic solvents like toluene or acetonitrile and operate at moderate temperatures ranging from 15 to 150 degrees Celsius. The reagents, including various oxidizing agents such as persulfates or peroxides, are commercially available and scalable, making the process robust for industrial manufacturing of high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemcitabine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and production of life-saving oncology therapies. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to delivering stringent purity specifications and maintaining rigorous QC labs to guarantee that every batch of gemcitabine intermediate meets the highest industry standards. Our state-of-the-art facilities are equipped to handle complex synthetic routes, including the oxidative stereoselective coupling described in CN103087132A, providing you with a secure and efficient source for your API requirements. By partnering with us, you gain access to a supply chain that is both robust and responsive, capable of adapting to your specific production timelines and quality expectations.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific manufacturing needs. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project goals. Whether you are in the early stages of process development or looking to optimize an existing commercial supply chain, we are here to support your success with our deep technical expertise and commitment to excellence. Let us help you secure a reliable supply of high-purity pharmaceutical intermediates that drive your business forward.
