Scalable Synthesis of Gemcitabine Key Intermediates via Optimized Reformatsky Reaction
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of high-value anticancer agents, particularly nucleoside analogs like Gemcitabine (2'-deoxy-2',2'-difluorocytidine). Patent CN1526711A introduces a transformative synthetic strategy for the key intermediates of this potent drug, addressing critical bottlenecks in stereoselectivity and process scalability. This technology leverages a modified asymmetric Reformatsky reaction, utilizing polymerized (R)-2,3-O-acetonide-glyceraldehyde as a starting material to achieve superior diastereoselectivity without the need for complex chromatographic separations. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for oncology APIs, ensuring consistent quality while mitigating the risks associated with low-yielding, labor-intensive legacy processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Gemcitabine intermediates has been plagued by poor stereocontrol and reliance on purification techniques that are incompatible with large-scale manufacturing. As illustrated in the foundational work by L.W. Hertel et al., the traditional substrate-controlled asymmetric Reformatsky reaction typically yields a cis-to-trans product ratio of merely 1:3. 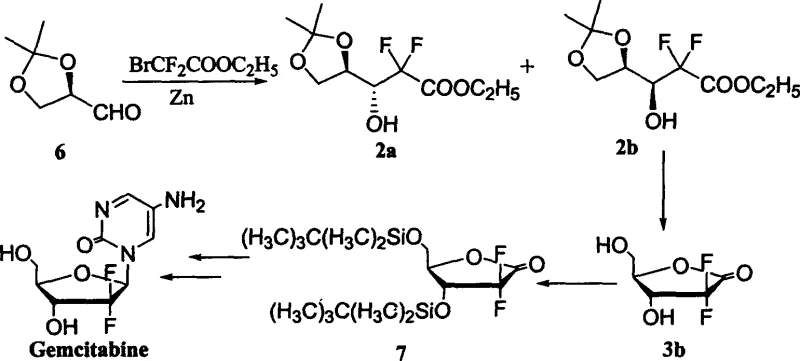 This low selectivity necessitates the use of column chromatography to isolate the desired trans-isomer, a technique that is notoriously difficult to scale, consumes vast quantities of solvents, and introduces significant batch-to-batch variability. Furthermore, alternative improvements, such as those by T.S. Chou, attempted to bypass chromatography but resulted in elongated synthetic sequences with overall yields as low as 25%, rendering them economically unviable for commercial production.
This low selectivity necessitates the use of column chromatography to isolate the desired trans-isomer, a technique that is notoriously difficult to scale, consumes vast quantities of solvents, and introduces significant batch-to-batch variability. Furthermore, alternative improvements, such as those by T.S. Chou, attempted to bypass chromatography but resulted in elongated synthetic sequences with overall yields as low as 25%, rendering them economically unviable for commercial production.
The Novel Approach
The patented process described in CN1526711A fundamentally reengineers the reaction dynamics to favor the formation of the desired trans-product. By employing polymerized (R)-2,3-O-acetonide-glyceraldehyde directly and meticulously controlling the feeding speed of ethyl difluorohaloacetate, the method achieves a dramatically improved diastereomeric ratio ranging from 1:4 to 1:14. This enhanced selectivity allows for the replacement of column chromatography with vacuum distillation, a unit operation that is far more amenable to industrial scaling. The subsequent steps utilize acidic resin (Dowex-50) for deprotection and simple recrystallization for final purification, streamlining the entire workflow and significantly reducing the environmental footprint associated with solvent waste and silica disposal.
Mechanistic Insights into Asymmetric Reformatsky Reaction Optimization
The core innovation lies in the manipulation of the Reformatsky reaction kinetics and the physical state of the aldehyde starting material. Conventional wisdom suggests that monomeric glyceraldehyde derivatives are prone to racemization and side reactions; however, this patent exploits the polymeric nature of the starting material (where n=2~20) to modulate reactivity. The reaction is conducted in organic solvents such as tetrahydrofuran or toluene at elevated temperatures (65-75°C), with zinc powder acting as the metal mediator. The critical parameter is the rate of addition: slower addition rates (e.g., over 5 hours) correlate with higher selectivity for the trans-isomer (2b), suggesting that kinetic control prevents the formation of the thermodynamically less favorable or sterically hindered cis-isomer (2a).
Following the initial coupling, the process employs a mild deprotection strategy using Dowex-50 acidic resin in a methanol-water mixture. This heterogeneous catalysis approach offers distinct advantages over homogeneous acid hydrolysis, including easier catalyst recovery and reduced corrosion risks. The resulting mixture of 3a/3b is then subjected to benzoylation using benzoyl chloride in the presence of a base like 2,6-lutidine. The final purification relies on the differential solubility of the diastereomers in a dichloromethane and n-heptane system, allowing the pure target compound 4b to crystallize out while impurities remain in the mother liquor. This sequence ensures high purity without the need for preparative HPLC or flash chromatography.
How to Synthesize Gemcitabine Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory discovery to pilot plant operations. The procedure begins with the activation of zinc dust in anhydrous THF, followed by the controlled co-addition of the polymerized aldehyde and ethyl difluorobromoacetate. Maintaining the reaction temperature between 68-70°C during the addition phase is crucial for maximizing the yield, which can reach up to 65% with optimized feeding profiles. Following the reaction, the workup involves a standard aqueous quench, extraction, and a critical vacuum distillation step at 100°C/20Pa to isolate the intermediate.
- Prepare the reaction by suspending zinc powder in anhydrous THF and activating with iodine under reflux conditions.
- Control the addition rate of ethyl difluorobromoacetate and polymerized (R)-2,3-O-acetonide-glyceraldehyde to maximize trans-selectivity.
- Purify the crude mixture via vacuum distillation and subsequent acidic resin treatment to obtain the protected ribulose derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost structure and operational reliability. The elimination of column chromatography is perhaps the most significant economic driver, as it removes the need for expensive silica gel, reduces solvent consumption by orders of magnitude, and shortens the cycle time per batch. Furthermore, the reagents utilized—zinc powder, ethyl difluorobromoacetate, and common organic solvents—are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that often plague specialty reagent-dependent processes.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing expensive transition metal catalysts, such as Titanocene used in prior art, with inexpensive zinc powder. Additionally, the shift from chromatographic purification to vacuum distillation and recrystallization drastically lowers the cost of goods sold (COGS) by minimizing solvent usage and waste disposal fees. The simplified workflow also reduces labor hours required for monitoring and purification, contributing to overall operational efficiency.
- Enhanced Supply Chain Reliability: By relying on widely available bulk chemicals rather than specialized, custom-synthesized reagents, manufacturers can secure a more resilient supply chain. The robustness of the reaction conditions, which tolerate a range of solvents and do not require cryogenic temperatures (unlike some prior art methods requiring -40°C), ensures consistent production even in facilities with varying infrastructure capabilities. This reliability is critical for maintaining continuous supply of life-saving oncology medications.
- Scalability and Environmental Compliance: The design of this process is inherently scalable, moving away from batch-limited chromatography to continuous-friendly distillation and crystallization units. This facilitates the commercial scale-up of complex pharmaceutical intermediates from kilogram to multi-ton scales with minimal process re-engineering. Moreover, the reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, simplifying compliance and reducing the carbon footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for decision-making.
Q: How does this process improve stereoselectivity compared to traditional methods?
A: By utilizing polymerized (R)-2,3-O-acetonide-glyceraldehyde and strictly controlling the feeding speed of the difluoroacetate, the process achieves a diastereomeric ratio of up to 1:14 (cis:trans), significantly higher than the 1:3 ratio observed in conventional substrate-controlled reactions.
Q: Is column chromatography required for purification in this synthetic route?
A: No, one of the primary advantages of this patented method is the elimination of column chromatography. The intermediate is purified through vacuum distillation and recrystallization, making it highly suitable for industrial scale-up.
Q: What are the key cost-saving factors in this manufacturing process?
A: The process avoids expensive catalysts like Titanocene and eliminates the need for silica gel chromatography. Additionally, the use of cheap, readily available reagents such as zinc powder and ethyl difluorobromoacetate drastically reduces raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemcitabine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the global fight against cancer. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1526711A are fully realized in practical manufacturing. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of Gemcitabine intermediate meets the highest international standards for safety and efficacy, providing our partners with absolute confidence in their supply chain.
We invite pharmaceutical companies and contract research organizations to collaborate with us on optimizing their Gemcitabine supply chains. By leveraging our expertise in process chemistry and scale-up engineering, we can help you achieve significant operational efficiencies. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your production requirements.
