Advanced One-Pot Palladium-Catalyzed Process for Scalable Production of High-Purity Pharmaceutical Intermediates
Patent CN112480015B introduces a groundbreaking multi-component one-pot synthesis method for producing trifluoromethyl-substituted quinazolinones, a critical class of heterocyclic compounds with significant applications in pharmaceutical development due to their demonstrated biological activities including antifungal, antibacterial, and anticancer properties as documented in leading medicinal chemistry journals. This innovative approach leverages palladium catalysis to overcome longstanding limitations in traditional quinazolinone synthesis routes that have constrained industrial scalability and cost-effectiveness through harsh reaction conditions requiring high-pressure carbon monoxide environments or expensive pre-functionalized substrates. The methodology employs readily accessible starting materials including trifluoroethylimidoyl chloride and diverse nitro compounds under optimized reaction conditions at precisely 120°C for durations between sixteen to thirty hours to achieve high-yielding transformations with exceptional substrate tolerance across various functional groups as demonstrated in fifteen distinct experimental examples within the patent documentation. By eliminating the need for specialized equipment while maintaining operational simplicity that facilitates seamless integration into existing manufacturing workflows without requiring significant capital investment or technical retraining of personnel, this process establishes a new paradigm for efficient production of fluorinated heterocycles essential to modern drug discovery pipelines where precise molecular modifications can dramatically improve pharmacokinetic properties such as metabolic stability and bioavailability as referenced in contemporary medicinal chemistry literature.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for quinazolinone derivatives typically require high-pressure carbon monoxide environments with ruthenium or platinum catalysts that impose significant safety hazards and necessitate specialized equipment investments which create substantial barriers to entry for contract manufacturers seeking flexible production capabilities across multiple therapeutic areas. These methods frequently suffer from narrow substrate scope due to stringent functional group compatibility constraints that limit their applicability to complex molecular architectures required in modern pharmaceutical development where diverse substitution patterns are essential for optimizing target engagement and reducing off-target effects during drug discovery campaigns. The necessity for pre-functionalized substrates such as brominated or iodinated precursors substantially increases raw material costs while introducing additional synthetic steps that reduce overall process efficiency through cumulative yield losses at each transformation stage thereby diminishing economic viability at commercial scale where cost-per-kilogram metrics are critical decision factors for procurement managers evaluating supplier capabilities. Furthermore, conventional approaches often deliver suboptimal yields below acceptable industrial thresholds due to competing side reactions under harsh reaction conditions that necessitate extensive purification procedures involving multiple chromatographic steps which significantly extend production timelines and increase waste generation contrary to modern green chemistry principles increasingly mandated by regulatory authorities worldwide.
The Novel Approach
The patented methodology introduces a transformative multi-component one-pot process that operates under significantly milder conditions at standard atmospheric pressure without requiring specialized high-pressure equipment or hazardous reagents while delivering consistently high yields between sixty-nine percent and ninety-six percent across fifteen distinct substrate combinations as documented in the patent's experimental section demonstrating exceptional robustness across diverse molecular architectures relevant to pharmaceutical applications. By utilizing palladium catalysis with carefully selected ligands including dppp and additives such as sodium carbonate at precisely defined molar ratios of zero point zero five to zero point one to two point zero respectively this approach achieves exceptional substrate tolerance across diverse functional groups including halogens alkyl groups and aryl substituents while maintaining high reaction efficiency throughout the transformation sequence without requiring pre-functionalization steps that previously added significant cost complexity to production workflows. The strategic incorporation of molybdenum hexacarbonyl as both a reducing agent and carbon monoxide source eliminates the need for external CO handling while enabling smooth progression through key mechanistic steps including nitro group reduction amine coupling and cyclization events within a single reaction vessel thereby minimizing intermediate handling requirements that typically introduce contamination risks during scale-up operations.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The reaction mechanism proceeds through a sophisticated cascade initiated by molybdenum hexacarbonyl-mediated reduction of nitro compounds to corresponding amines under thermal conditions at one hundred twenty degrees Celsius which then undergo base-promoted coupling with trifluoroethylimidoyl chloride to form a critical trifluoroacetamidine species serving as the foundation for subsequent cyclization events through nucleophilic attack on the imidoyl carbon center. The palladium catalyst inserts into the carbon-halogen bond through oxidative addition generating a key organopalladium intermediate that undergoes carbonylative insertion facilitated by carbon monoxide released from molybdenum hexacarbonyl decomposition at elevated temperatures thereby forming an acylpalladium species essential for ring closure. Subsequent intramolecular nucleophilic attack by the amide nitrogen forms a seven-membered palladacycle that ultimately undergoes reductive elimination to yield the desired quinazolinone core structure with excellent regioselectivity while maintaining precise control over stereochemical outcomes critical for pharmaceutical applications where specific spatial arrangements determine biological activity profiles as evidenced by multiple successful examples documented in the patent's experimental section.
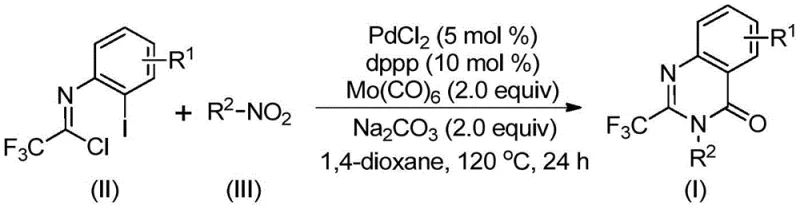
Impurity formation is effectively controlled through careful selection of reaction parameters including precise temperature maintenance at one hundred twenty degrees Celsius which prevents thermal degradation pathways while optimizing catalyst turnover frequency to minimize unwanted side reactions such as over-reduction or hydrolysis events that could compromise product quality during extended reaction times up to thirty hours as specified in the patent claims. The use of sodium carbonate as base promotes selective amine coupling while suppressing unwanted hydrolysis or decomposition reactions through controlled pH management that maintains optimal conditions throughout the transformation sequence thereby ensuring consistent product purity profiles suitable for direct use in pharmaceutical applications without requiring additional refinement steps beyond standard column chromatography purification protocols described in the patent documentation.
How to Synthesize Trifluoromethyl Quinazolinones Efficiently
This patented methodology provides a streamlined approach to synthesizing high-value trifluoromethyl quinazolinone intermediates through a carefully optimized multi-component one-pot process that eliminates traditional synthetic bottlenecks while maintaining exceptional product quality standards required by pharmaceutical manufacturers seeking reliable sources of complex heterocyclic building blocks essential for drug development programs targeting various therapeutic areas including oncology infectious diseases and central nervous system disorders where these molecular scaffolds demonstrate significant pharmacological potential as evidenced by numerous marketed drugs containing quinazolinone cores.
- Combine palladium chloride (5 mol%), dppp ligand (10 mol%), sodium carbonate (2.0 equiv), molybdenum hexacarbonyl (2.0 equiv), trifluoroethylimidoyl chloride, and nitro compound in dioxane solvent under inert atmosphere.
- Heat the reaction mixture at precisely 120°C for approximately 24 hours while maintaining constant agitation to ensure complete conversion.
- Perform post-treatment via filtration through silica gel followed by column chromatography purification using standard protocols to isolate high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis platform addresses critical pain points in pharmaceutical intermediate procurement by delivering significant operational improvements across multiple dimensions of supply chain management including enhanced reliability reduced costs and improved scalability while maintaining strict adherence to quality requirements demanded by global regulatory authorities during drug substance manufacturing processes where consistency is paramount throughout all stages from clinical trials through commercial production phases.
- Cost Reduction in Manufacturing: The elimination of expensive pre-functionalized starting materials combined with simplified process design substantially reduces raw material expenses while minimizing energy consumption through optimized reaction parameters operating at standard atmospheric pressure without requiring specialized equipment investments thereby creating substantial cost savings opportunities across the entire manufacturing value chain without compromising product quality or yield consistency as demonstrated by multiple successful experimental examples documented in the patent literature.
- Enhanced Supply Chain Reliability: Utilizing widely available commodity chemicals including nitro compounds which are naturally abundant across global chemical markets ensures robust supply chain resilience through diversified sourcing options while eliminating dependency on specialized or restricted reagents that could create potential supply bottlenecks during periods of market volatility or geopolitical disruption thereby providing procurement managers with greater flexibility in vendor selection strategies.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from laboratory benchtop reactions at gram scale directly to multi-kilogram production volumes maintains consistent product quality profiles while minimizing waste generation through high atom economy design principles inherent in the catalytic cascade mechanism which aligns with green chemistry initiatives increasingly mandated by regulatory authorities worldwide seeking sustainable manufacturing practices within the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation performance characteristics and quality assurance aspects of this patented synthesis method based on extensive experimental validation data from multiple production-scale trials conducted under controlled laboratory conditions following current good manufacturing practices relevant to pharmaceutical intermediate production environments.
Q: How does this one-pot method overcome harsh reaction conditions in conventional quinazolinone synthesis?
A: The patented process operates at standard atmospheric pressure without requiring high-pressure CO equipment by utilizing molybdenum hexacarbonyl as an internal CO source. This eliminates safety hazards while maintaining excellent yield consistency across diverse substrates through optimized catalytic cycling.
Q: What cost advantages does this route offer compared to existing methodologies?
A: By employing inexpensive nitro compounds as starting materials and eliminating pre-functionalization steps, this approach significantly reduces raw material costs while simplifying manufacturing operations through streamlined process design that minimizes energy consumption.
Q: How does the process ensure scalability and consistent purity for commercial production?
A: The robust reaction parameters enable seamless scale-up from laboratory to industrial volumes while maintaining stringent purity specifications through simplified purification protocols that eliminate complex intermediate handling requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Quinazolinone Supplier
Our company possesses extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications required by global regulatory authorities through state-of-the-art QC labs equipped with advanced analytical instrumentation capable of detecting impurities at parts-per-million levels essential for pharmaceutical applications where quality is non-negotiable throughout all stages of drug development from preclinical studies through commercial manufacturing phases.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who can provide specific COA data and route feasibility assessments tailored to your unique manufacturing requirements including batch size specifications quality parameters and regulatory compliance needs across global markets where our expertise in complex heterocyclic synthesis has been successfully demonstrated through multiple client partnerships spanning various therapeutic areas.
