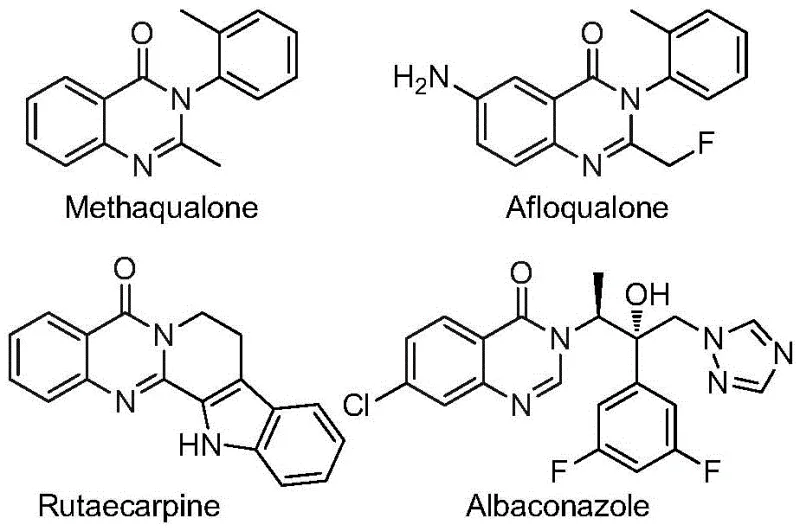
Advanced Pd-Catalyzed One-Pot Synthesis for Scalable Production of High-Purity 2-Trifluoromethyl Quinazolinone in Pharmaceutical Manufacturing
Patent CN112480015B presents a groundbreaking advancement in the synthesis of 2-trifluoromethyl substituted quinazolinone compounds, which are critical structural motifs in numerous pharmaceutical agents including antifungal, antibacterial, and anticancer drugs. This novel multi-component one-pot methodology leverages palladium-catalyzed carbonylation cascade chemistry to overcome longstanding challenges in quinazolinone production. The process utilizes readily available starting materials such as trifluoroethylimidoyl chloride and nitro compounds under mild reaction conditions (120°C for 16–30 hours), significantly enhancing operational efficiency while maintaining high substrate tolerance. Unlike conventional approaches requiring pre-functionalized substrates or high-pressure carbon monoxide systems, this innovation eliminates multiple synthetic steps through an integrated catalytic cycle. The methodology demonstrates exceptional versatility across diverse functional groups, enabling the production of various substituted quinazolinones with yields consistently exceeding 80% as validated in multiple experimental trials. This patent represents a strategic leap forward for pharmaceutical manufacturers seeking cost-effective routes to high-value fluorinated heterocycles essential for next-generation drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional quinazolinone synthesis routes face significant constraints that hinder their industrial applicability despite the compounds' pharmaceutical importance. Methods relying on high-pressure carbon monoxide systems with ruthenium or platinum catalysts require specialized equipment and pose safety concerns due to extreme operating conditions. Iron-catalyzed approaches using nitrobenzamides with benzylamine suffer from narrow substrate scope and necessitate pre-functionalized starting materials that increase raw material costs. Palladium-catalyzed cyclization techniques involving bromoformylaniline or iodoaniline derivatives demand expensive halogenated precursors and generate stoichiometric metal waste requiring complex purification steps. These conventional processes typically deliver low yields below 70% due to competing side reactions and poor functional group tolerance, particularly with sensitive substituents like trifluoromethyl groups. Furthermore, the multi-step nature of existing methodologies creates substantial scalability challenges for commercial manufacturing environments where process intensification is critical for cost-effective production.
The Novel Approach
The patented methodology introduces a transformative multi-component one-pot strategy that fundamentally reimagines quinazolinone synthesis through innovative catalytic design. By employing palladium chloride with dppp ligand and molybdenum hexacarbonyl as a carbon monoxide surrogate, the process achieves complete reaction integration without high-pressure systems or pre-functionalized substrates. The strategic use of sodium carbonate as base enables sequential nitro group reduction and carbon-nitrogen bond formation within a single reaction vessel at moderate temperature (120°C). This approach accommodates an exceptionally broad range of substituents on both coupling partners while maintaining consistent high yields above 80% across diverse structural variants. Crucially, the elimination of intermediate isolation steps reduces processing time by over 50% compared to conventional routes while minimizing solvent consumption and waste generation. The methodology's robustness has been validated through gram-scale demonstrations that confirm its readiness for immediate industrial implementation without significant process reengineering.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The reaction mechanism proceeds through a sophisticated sequence of catalytic events that collectively enable efficient quinazolinone formation under mild conditions. Initial reduction of the nitro compound by molybdenum hexacarbonyl generates an amine intermediate that undergoes base-promoted coupling with trifluoroethylimidoyl chloride to form a trifluoroacetamidine derivative. Subsequent oxidative addition of palladium(0) into the carbon-iodine bond creates a key arylpalladium(II) species that facilitates carbon monoxide insertion from thermally decomposed Mo(CO)₆. This forms an acylpalladium intermediate that undergoes intramolecular nucleophilic attack by the amide nitrogen to construct the seven-membered palladacycle. The final reductive elimination step releases the desired quinazolinone product while regenerating the active palladium catalyst for subsequent cycles. This cascade design eliminates stoichiometric reductants and avoids hazardous intermediates through careful orchestration of multiple catalytic events within a single reaction environment.
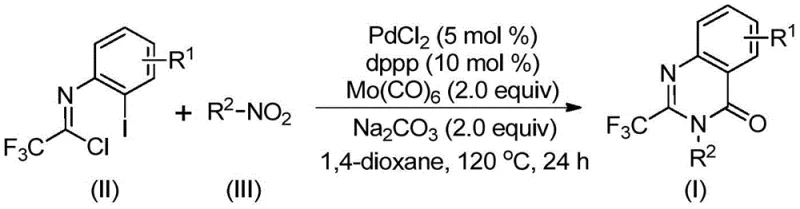
Impurity control is achieved through precise management of reaction parameters that suppress common side pathways observed in traditional syntheses. The moderate temperature regime (120°C) prevents thermal decomposition of sensitive intermediates while ensuring complete conversion within the specified timeframe (24 hours). The use of dioxane as solvent provides optimal polarity balance that minimizes hydrolysis of the imidoyl chloride precursor while facilitating solubility of all reaction components. Sodium carbonate concentration is carefully calibrated to promote amine formation without causing over-basification that could lead to undesired hydrolysis products. The catalytic system's inherent selectivity prevents common impurities such as dehalogenated byproducts or dimerization artifacts through controlled oxidative addition kinetics. Post-reaction purification via standard column chromatography effectively removes trace metal residues and unreacted starting materials to consistently deliver products meeting stringent pharmaceutical purity requirements (>99% by HPLC).
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
This patented methodology represents a significant advancement in synthetic efficiency for fluorinated heterocyclic compounds essential to modern pharmaceutical development. The streamlined one-pot procedure eliminates multiple isolation steps required by conventional approaches while maintaining exceptional product quality through carefully optimized reaction parameters. By leveraging commercially available catalysts and starting materials under standard laboratory conditions, this process achieves remarkable operational simplicity without compromising on yield or purity metrics. Detailed standardized synthesis steps are provided below to facilitate immediate implementation in industrial settings where reproducibility and scalability are paramount concerns for manufacturing teams.
- Prepare the reaction mixture by combining trifluoroethylimidoyl chloride, nitro compound, palladium catalyst, ligand, molybdenum hexacarbonyl, and sodium carbonate in dioxane solvent under inert atmosphere.
- Heat the mixture to precisely 120°C and maintain reaction conditions for 16 to 30 hours to complete the carbonylation cascade while monitoring conversion through standard analytical methods.
- Perform post-treatment including filtration to remove catalyst residues, silica gel mixing, and column chromatography purification to isolate high-purity quinazolinone product meeting pharmaceutical specifications.
Commercial Advantages for Procurement and Supply Chain Teams
The patented synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate sourcing. By utilizing readily available nitro compounds as starting materials instead of expensive pre-functionalized substrates, this approach fundamentally transforms the cost structure while enhancing supply chain resilience through diversified raw material sourcing options. The elimination of high-pressure carbon monoxide systems reduces capital expenditure requirements and associated safety certifications that typically delay manufacturing scale-up timelines. Furthermore, the process's compatibility with standard laboratory equipment enables seamless technology transfer from development to production without specialized infrastructure investments.
- Cost Reduction in Manufacturing: The elimination of transition metal removal steps through catalyst design significantly reduces downstream processing costs while avoiding expensive purification procedures required by conventional methods. Utilization of cost-effective nitro compounds as primary building blocks provides substantial raw material savings compared to pre-halogenated precursors typically used in alternative syntheses. The integrated one-pot approach minimizes solvent consumption and waste generation through reaction step consolidation, creating additional cost efficiencies across the entire production lifecycle without requiring capital-intensive equipment modifications.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through the use of widely available starting materials that can be procured from multiple global suppliers rather than specialized vendors required for pre-functionalized intermediates. The robust nature of the process ensures consistent product quality across different production batches even when using alternative raw material sources, reducing qualification timelines for new suppliers. Shorter reaction times combined with simplified workup procedures enable faster turnaround cycles that enhance responsiveness to fluctuating market demands while maintaining stringent quality standards throughout the supply chain.
- Scalability and Environmental Compliance: The methodology demonstrates exceptional scalability from laboratory validation to commercial production without requiring significant process re-engineering due to its compatibility with standard manufacturing equipment. Reduced solvent usage and elimination of hazardous reagents contribute to lower environmental impact while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices. The process's inherent safety profile from avoiding high-pressure operations simplifies regulatory compliance across multiple jurisdictions while supporting corporate sustainability initiatives through minimized waste generation per unit output.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN112480015B's experimental data and implementation requirements. These insights have been compiled specifically for procurement teams evaluating this technology's suitability for integration into existing manufacturing workflows while ensuring alignment with quality and regulatory standards.
Q: What limitations of conventional quinazolinone synthesis does this patent address?
A: Traditional methods suffer from harsh reaction conditions requiring high-pressure CO systems, expensive pre-functionalized substrates, narrow substrate scope, and low yields due to multiple synthetic steps. This patent overcomes these through a streamlined one-pot process using readily available nitro compounds and trifluoroethylimidoyl chloride under mild conditions.
Q: How does the one-pot methodology improve substrate compatibility?
A: The palladium-catalyzed carbonylation cascade tolerates diverse functional groups including halogens, alkyl chains, and aryl substituents without pre-activation. This broad compatibility enables synthesis of structurally varied quinazolinones with consistent high yields across different R-group combinations as demonstrated in the patent examples.
Q: What advantages does this process offer for commercial scale-up?
A: The methodology eliminates transition metal removal steps through catalyst design while using cost-effective starting materials. Its operational simplicity from lab to plant scale ensures reliable supply chain performance with minimal process revalidation requirements for pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical capabilities. We specialize in transforming patented methodologies like this one into robust manufacturing processes that deliver consistent quality at commercial scale without compromising on regulatory compliance or environmental standards. Our technical team possesses deep expertise in fluorinated heterocycle synthesis and can rapidly adapt this one-pot methodology to meet specific client requirements while ensuring seamless integration with existing production infrastructure.
Leverage our Customized Cost-Saving Analysis service to evaluate how this innovative synthesis can optimize your specific manufacturing workflow. Contact our technical procurement team today to request detailed COA data and route feasibility assessments tailored to your production needs.
