Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Production
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Production
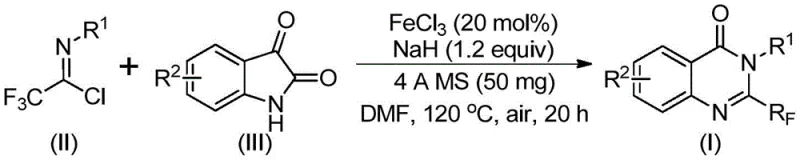
The pharmaceutical industry continuously seeks robust and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly quinazolinones, which serve as privileged scaffolds in drug discovery. Patent CN111675662B introduces a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, addressing critical bottlenecks in current synthetic routes. This technology leverages an iron-catalyzed cyclization strategy that transforms readily available trifluoroethylimidoyl chloride and isatin derivatives into high-value intermediates. For R&D directors and procurement managers alike, this innovation represents a significant shift towards sustainable and economically viable manufacturing processes. The introduction of the trifluoromethyl group is strategically vital, as it enhances the electronegativity, metabolic stability, and lipophilicity of the target molecules, thereby improving their bioavailability and therapeutic potential. By utilizing cheap metal iron catalysts instead of precious metals, this method aligns perfectly with the industry's drive for cost reduction in pharmaceutical intermediate manufacturing while maintaining high purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinones bearing trifluoromethyl functional groups has relied heavily on cyclization reactions involving synthons such as trifluoroacetic anhydride or ethyl trifluoroacetate reacting with substrates like anthranilamide or isatoic anhydride. While these methods are chemically feasible, they suffer from severe practical limitations that hinder their industrial application. The reaction conditions are often excessively harsh, requiring extreme temperatures or strong bases that can degrade sensitive functional groups on the substrate. Furthermore, the starting materials, particularly specialized trifluoroacetylating agents, are frequently expensive and difficult to source in bulk quantities, leading to inflated production costs. Another critical drawback is the narrow substrate scope; many conventional protocols fail to accommodate diverse substituents, resulting in low yields and complex impurity profiles that necessitate rigorous and costly purification steps. These factors collectively create a supply chain vulnerability, making it difficult for manufacturers to guarantee consistent delivery of high-purity intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes trifluoroethylimidoyl chloride and isatin as starting raw materials, catalyzed by inexpensive ferric chloride. This methodology fundamentally alters the economic and operational landscape of quinazolinone synthesis. The reaction proceeds through a tandem sequence involving alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization. This pathway is not only operationally simple but also demonstrates remarkable functional group tolerance, accommodating electron-donating and electron-withdrawing groups alike. The use of DMF as a solvent and 4A molecular sieves further optimizes the reaction environment, ensuring high conversion rates. By shifting away from expensive precious metal catalysts and harsh acylating agents, this process offers a streamlined route that is inherently safer and more scalable. The ability to operate under air atmosphere without stringent inert gas protection simplifies the equipment requirements, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of nucleophilic substitution and transition metal catalysis. Initially, the sodium hydride acts as a base to deprotonate the isatin nitrogen, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a key trifluoroacetamidine intermediate, establishing the crucial carbon-nitrogen bond. Subsequently, the ferric chloride catalyst facilitates a decarbonylation event, effectively removing the carbonyl oxygen from the isatin moiety. This decarbonylation is the driving force for the subsequent cyclization, where the nitrogen atom attacks the imine carbon to close the six-membered quinazolinone ring. The presence of the trifluoromethyl group at the 2-position is retained throughout this process, stabilized by the electronic effects of the adjacent nitrogen atoms. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters, as it highlights the dual role of the reagents: sodium hydride as an activator and ferric chloride as the cyclization promoter.
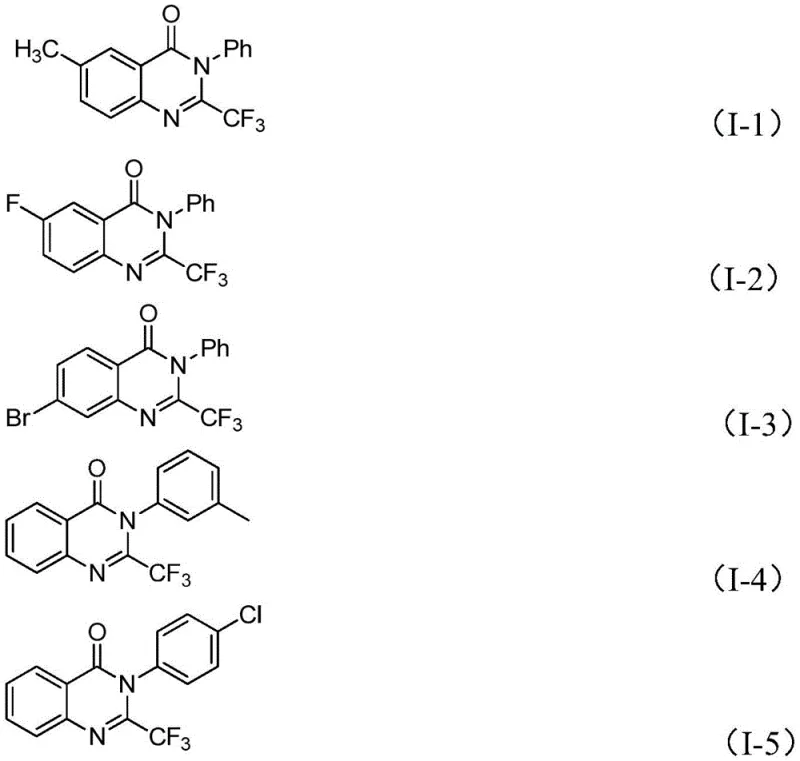
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid-catalyzed cyclizations. The mild basic conditions prevent the hydrolysis of the imine bond, a common side reaction in acidic media that leads to open-chain byproducts. Furthermore, the specificity of the iron catalyst minimizes non-selective radical pathways that could generate polymeric impurities. The patent data indicates that even with diverse substituents such as halogens (fluoro, chloro, bromo) and alkyl groups, the reaction maintains high selectivity. This robustness ensures that the crude product contains fewer structurally related impurities, significantly reducing the burden on downstream purification processes. For quality control laboratories, this translates to cleaner chromatograms and easier validation of the final active pharmaceutical ingredient (API) intermediate.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the precise weighing of ferric chloride and sodium hydride, followed by the addition of 4A molecular sieves to scavenge moisture, which is critical for the success of the reaction. The reactants, trifluoroethylimidoyl chloride and the chosen isatin derivative, are then introduced into the organic solvent, typically DMF. The reaction is initiated at a moderate temperature of 40°C for roughly 10 hours to allow for the initial coupling, followed by heating to 120°C for an additional 20 hours to drive the cyclization to completion. This two-stage temperature profile is essential for balancing reaction rate and selectivity. Detailed standardized synthesis steps see the guide below.
- Mix ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in DMF solvent.
- Stir the reaction mixture at 40°C for approximately 10 hours to initiate the coupling.
- Heat the mixture to 120°C and continue reacting for 20 hours under air to complete the cyclization.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this FeCl3-catalyzed route offers transformative benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive and hazardous trifluoroacetylating agents with stable and commercially abundant trifluoroethylimidoyl chlorides, companies can secure a more resilient supply chain. The reliance on iron, one of the most abundant and cheapest transition metals, eliminates the volatility associated with precious metal prices, ensuring long-term cost stability. Moreover, the operational simplicity—requiring no specialized high-pressure equipment or inert atmosphere gloveboxes—reduces capital expenditure for new production lines. This accessibility allows for rapid deployment of manufacturing capacity, reducing lead time for high-purity pharmaceutical intermediates and enabling faster response to market demands.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts such as palladium or rhodium results in substantial cost savings, as iron salts are orders of magnitude cheaper. Additionally, the high atom economy of the cyclization reaction minimizes waste generation, lowering disposal costs. The use of common solvents like DMF, which can be recovered and recycled, further enhances the economic viability of the process. These factors combine to significantly lower the cost of goods sold (COGS) for the final intermediate, providing a competitive edge in pricing negotiations with downstream API manufacturers.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted isatins and aromatic amines used to prepare the imidoyl chlorides, are widely available from global chemical suppliers. This diversity of sources mitigates the risk of supply disruptions caused by single-source dependencies. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental parameters, ensuring consistent batch-to-batch quality. This reliability is paramount for maintaining continuous supply to pharmaceutical clients who operate on tight just-in-time inventory schedules.
- Scalability and Environmental Compliance: The process has been demonstrated to work efficiently on a gram scale with yields reaching up to 93%, indicating strong potential for kilogram and tonne-scale production. The absence of toxic heavy metals in the catalyst system simplifies the regulatory compliance landscape, particularly regarding residual metal limits in pharmaceutical products. Furthermore, the straightforward workup procedure involving filtration and chromatography reduces the volume of aqueous waste streams, aligning with modern green chemistry principles and environmental sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the patent specifications and experimental data. These insights are intended to clarify the feasibility of implementing this technology in your existing manufacturing infrastructure. Understanding these details is crucial for making informed decisions about process adoption and supplier qualification.
Q: What are the advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: Ferric chloride is significantly cheaper and more abundant than precious metal catalysts. The patent demonstrates that FeCl3 enables high yields (up to 93%) under relatively mild conditions compared to harsh traditional methods requiring expensive trifluoroacetylating agents.
Q: Can this synthesis method tolerate various functional groups on the substrate?
A: Yes, the method exhibits excellent functional group tolerance. The patent data confirms successful synthesis with substrates containing methyl, fluoro, bromo, chloro, methoxy, and nitro groups at ortho-, meta-, and para-positions without significant loss in yield.
Q: Is the purification process complex for large-scale manufacturing?
A: The post-treatment is straightforward, involving simple filtration followed by standard silica gel column chromatography. This simplicity suggests the process is amenable to scale-up, avoiding complex extraction or crystallization steps often required in heterocyclic synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN111675662B for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our state-of-the-art facilities are equipped to handle the specific requirements of iron-catalyzed reactions, and our rigorous QC labs enforce stringent purity specifications to meet the highest international standards. We are committed to delivering high-purity 2-trifluoromethyl quinazolinones that serve as reliable building blocks for your drug discovery programs.
We invite you to collaborate with us to leverage this advanced technology for your specific pipeline needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in turning innovative chemistry into commercial success.
