Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Pharmaceutical Intermediates
Introduction to Novel Biheterocyclic Synthesis Technology
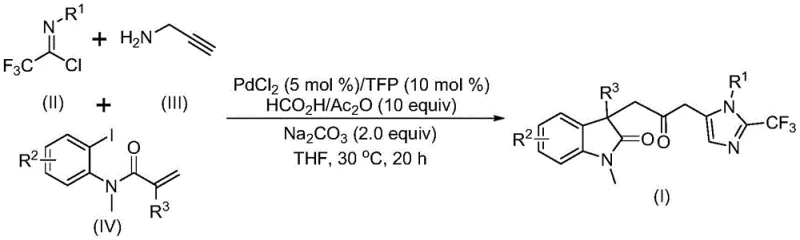
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, which serve as the core backbone for numerous bioactive molecules. Patent CN115353511A introduces a groundbreaking multi-component synthesis strategy for producing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technological advancement addresses critical challenges in modern organic synthesis by replacing hazardous reagents with safer alternatives while maintaining high reaction efficiency. The disclosed method utilizes a palladium-catalyzed cascade reaction that operates under remarkably mild conditions, typically at 30°C, thereby minimizing energy consumption and thermal degradation risks. By integrating trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives in a one-pot procedure, this innovation streamlines the production of high-value pharmaceutical intermediates. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering complex structures with consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-bridged biheterocyclic systems has relied heavily on traditional carbonylation techniques that present significant safety and logistical hurdles. Conventional protocols often necessitate the use of pressurized carbon monoxide gas, which is highly toxic and requires specialized infrastructure for safe handling and storage. Furthermore, many existing synthetic routes involve harsh reaction conditions, including elevated temperatures and strong bases, which can lead to poor functional group tolerance and the formation of complex impurity profiles. These limitations not only increase the operational risk for manufacturing facilities but also complicate the purification process, resulting in lower overall yields and higher production costs. Additionally, the direct coupling of two distinct heterocyclic substrates often suffers from low atom economy and requires pre-functionalized starting materials that are expensive and difficult to source commercially. These factors collectively hinder the scalable production of diverse biheterocyclic libraries needed for drug discovery campaigns.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN115353511A employs a transition metal palladium-catalyzed carbonylation cascade that fundamentally reshapes the synthetic landscape. This novel approach eliminates the need for external carbon monoxide gas by utilizing an in situ generation system comprising formic acid and acetic anhydride. This substitution drastically improves the safety profile of the reaction, making it accessible to standard laboratory and pilot plant environments without specialized gas handling equipment. The reaction proceeds efficiently at a mild temperature of 30°C in tetrahydrofuran (THF), demonstrating exceptional compatibility with a wide range of functional groups including halogens, nitro groups, and trifluoromethyl substituents. By merging three readily available components—trifluoroethylimidoyl chloride, propargylamine, and acrylamide—the method achieves high atom economy and structural diversity in a single operational step. This streamlined process not only reduces the number of synthetic steps but also simplifies downstream processing, offering a compelling solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps that ensure high regioselectivity and yield. The cycle initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This is immediately followed by an intramolecular Heck-type reaction, which constructs the indolinone core and forms a divalent alkyl-palladium species. Subsequently, the in situ generated carbon monoxide inserts into the palladium-carbon bond, creating an acyl-palladium intermediate that serves as the electrophilic center for the subsequent cyclization. Parallel to this metal cycle, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine species, which undergoes isomerization to become nucleophilic. The final ring-closing step is catalyzed by the activated acyl-palladium intermediate, facilitating the intramolecular cyclization that yields the final carbonyl-bridged biheterocyclic product. This intricate interplay between the palladium cycle and the amine condensation ensures that the reaction proceeds with high fidelity, minimizing side reactions and byproduct formation.
From an impurity control perspective, the mild reaction temperature of 30°C plays a pivotal role in maintaining product purity. High-temperature processes often promote decomposition pathways or non-selective polymerization of the acrylamide and alkyne components; however, the low thermal energy input in this protocol suppresses these degradation mechanisms. Furthermore, the use of trifurylphosphine (TFP) as a ligand enhances the stability of the palladium catalyst, preventing the formation of palladium black and ensuring consistent catalytic turnover throughout the 12 to 20-hour reaction window. The compatibility with various substituents on the aromatic rings (R1, R2, R3) indicates that electronic effects are well-managed by the catalyst system, allowing for the synthesis of electron-deficient and electron-rich derivatives without significant loss in efficiency. This robustness is critical for commercial scale-up of complex pharmaceutical intermediates where batch-to-batch consistency is paramount.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
The practical execution of this synthesis relies on precise stoichiometric control and the selection of high-purity reagents to maximize yield. The patent outlines a standardized protocol where palladium chloride and trifurylphosphine are combined with sodium carbonate and the CO-source mixture in THF prior to the addition of substrates. Maintaining an inert atmosphere is recommended to protect the active palladium species from oxidation, although the system shows reasonable tolerance. The reaction mixture is stirred at 30°C for a duration of 12 to 20 hours, after which the crude product is isolated via filtration and purified using standard silica gel column chromatography. This straightforward workup procedure avoids complex extraction sequences, further contributing to process efficiency. For detailed operational parameters and specific molar ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Combine palladium chloride, trifurylphosphine ligand, sodium carbonate, and an in situ carbon monoxide source (formic acid/acetic anhydride mixture) in an organic solvent such as THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the acrylamide derivative to the reaction mixture under inert atmosphere.
- Stir the reaction at a mild temperature of 30°C for 12 to 20 hours, followed by filtration, silica gel treatment, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical novelty. The shift away from toxic carbon monoxide gas cylinders removes a significant regulatory and safety burden from the manufacturing facility, reducing insurance costs and compliance overheads associated with hazardous gas storage. Moreover, the starting materials identified in the patent, such as propargylamine and various acrylamides, are commodity chemicals available from multiple global suppliers, ensuring supply chain resilience and reducing the risk of single-source bottlenecks. The ability to operate at near-ambient temperature (30°C) translates directly to lower energy consumption compared to refluxing conditions, aligning with sustainability goals and reducing utility costs. These factors collectively contribute to a more stable and cost-effective supply chain for high-purity pharmaceutical intermediates.
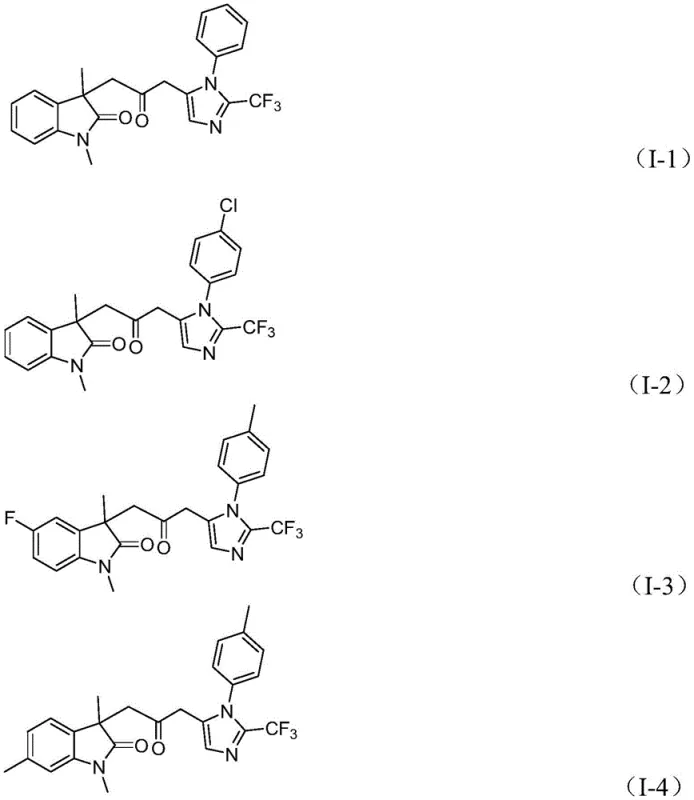
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous pressurized CO gas infrastructure represents a significant capital expenditure saving for manufacturing sites. Additionally, the use of palladium chloride, a relatively inexpensive palladium source compared to specialized complexes, combined with the efficient ligand system, optimizes catalyst costs. The high yields reported in the patent examples, reaching up to 92% for certain substrates, minimize raw material waste and reduce the load on purification units, leading to substantial cost savings in the overall production budget. The simplified one-pot nature of the reaction reduces labor hours and solvent usage compared to multi-step linear syntheses.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures that production schedules are not disrupted by the scarcity of exotic reagents. The broad substrate scope demonstrated in the patent allows for the rapid adaptation of the process to synthesize various analogues required for structure-activity relationship (SAR) studies without needing to develop entirely new synthetic routes. This flexibility is crucial for maintaining continuity in drug development pipelines. Furthermore, the robustness of the reaction conditions means that minor fluctuations in raw material quality are less likely to cause batch failures, enhancing overall supply reliability.
- Scalability and Environmental Compliance: The patent explicitly mentions the successful expansion of the reaction to the gram scale, providing a clear pathway for kilogram and ton-scale production. The mild conditions and the absence of toxic gas emissions make this process environmentally friendlier, facilitating easier permitting and compliance with increasingly stringent environmental regulations. The use of THF as a solvent, while requiring recovery systems, is a standard practice in the industry, and the simple filtration workup minimizes aqueous waste generation. This scalability ensures that the technology can meet the demands of clinical trial material production as well as commercial API manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this technology for adoption. Understanding these specifics helps in assessing the feasibility of integrating this route into existing manufacturing workflows.
Q: What is the primary safety advantage of this carbonylation method?
A: Unlike traditional carbonylation reactions that require handling toxic carbon monoxide gas cylinders, this patent utilizes a formic acid and acetic anhydride mixture to generate CO in situ, significantly enhancing operational safety.
Q: What is the substrate compatibility for the R1 and R3 groups?
A: The method demonstrates excellent functional group tolerance, accommodating alkyl, substituted aryl (with methyl, methoxy, halogens, trifluoromethyl, or nitro groups), and benzyl groups without compromising reaction efficiency.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly confirms that the reaction conditions are mild (30°C) and the procedure has been successfully expanded to gram-scale reactions, indicating strong potential for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
As the pharmaceutical landscape evolves, the demand for complex heterocyclic building blocks continues to grow, necessitating partners who can bridge the gap between academic innovation and industrial reality. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging deep expertise in palladium-catalyzed transformations to deliver high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably translated into bulk supply. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of carbonyl-bridged biheterocyclic compounds meets the exacting standards required for drug substance manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to this safer, more efficient route. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to initiate route feasibility assessments for your proprietary targets, ensuring a seamless supply chain for your next-generation therapeutics.
