Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production: A Metal-Free Base-Promoted Strategy for Scalable Pharma Intermediates
Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production: A Metal-Free Base-Promoted Strategy for Scalable Pharma Intermediates
The landscape of heterocyclic chemistry is continuously evolving to meet the rigorous demands of modern pharmaceutical and agrochemical development. A significant breakthrough in this domain is documented in patent CN113121462B, which discloses a highly efficient preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. These structural motifs are ubiquitous in bioactive molecules, serving as critical pharmacophores that enhance metabolic stability, lipophilicity, and binding affinity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this metal-free, base-promoted synthesis is paramount. The technology represents a paradigm shift away from traditional transition-metal catalysis, offering a cleaner, safer, and more economically viable pathway for generating complex fluorinated heterocycles essential for next-generation therapeutics.
The strategic value of this invention lies not merely in the chemical transformation itself but in its alignment with green chemistry principles and industrial scalability. By eliminating the need for copper catalysts and hazardous azide reagents, the process addresses two major pain points in fine chemical manufacturing: heavy metal contamination and process safety. This report provides a deep technical analysis of the methodology, evaluating its mechanistic underpinnings and commercial implications for global supply chains. As the industry moves towards more sustainable manufacturing practices, adopting such innovative synthetic routes becomes a competitive advantage for companies aiming to reduce lead time for high-purity pharmaceutical intermediates while maintaining stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
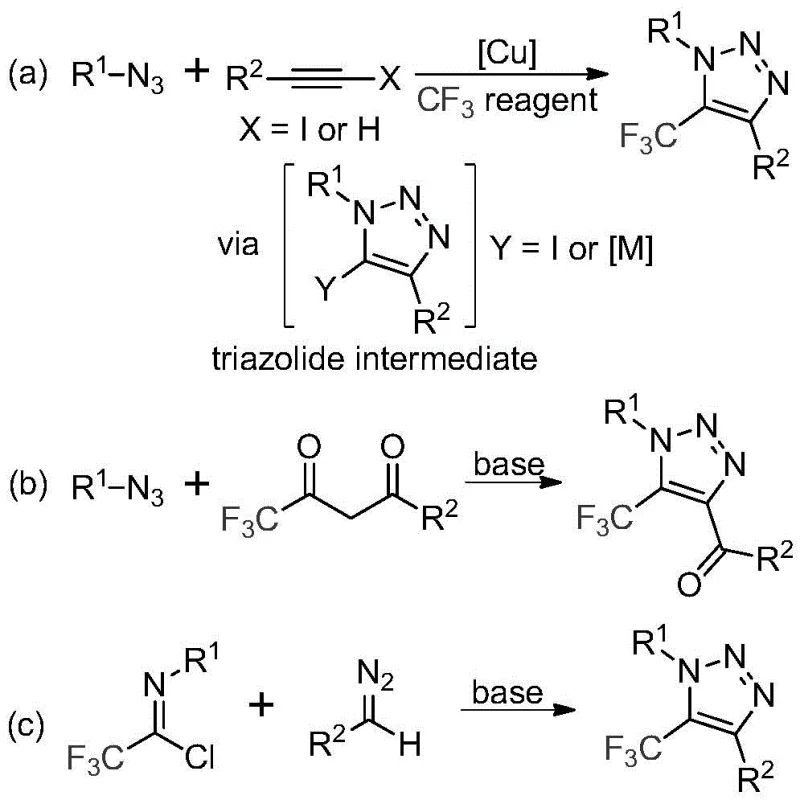
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on two primary strategies, both of which present significant logistical and safety challenges for large-scale operations. The first conventional approach involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides to form triazole metal complexes, which subsequently react with various trifluoromethylating reagents. While effective in academic settings, this method suffers from the inherent instability and explosive nature of organic azides, posing severe safety risks during storage and handling in a production environment. Furthermore, the reliance on copper catalysts introduces the complication of residual metal removal, a costly and time-consuming step required to meet the strict impurity profiles mandated by regulatory bodies for active pharmaceutical ingredients.
The second traditional method utilizes organocatalytic 1,3-dipolar cycloaddition reactions between azides and trifluoromethyl ketones. Although this avoids transition metals, it still necessitates the use of toxic and potentially explosive azide precursors. Additionally, trifluoromethyl ketones can be expensive and difficult to synthesize with high purity, driving up the overall cost of goods. Both methods often require harsh reaction conditions or specialized equipment to manage the exothermic nature of azide chemistry. For a procurement manager focused on cost reduction in API manufacturing, these factors translate into higher raw material costs, increased safety compliance expenditures, and potential supply chain disruptions due to the restricted availability of hazardous reagents. The environmental burden of disposing of azide-containing waste streams further complicates the sustainability profile of these legacy processes.
The Novel Approach
In stark contrast to these legacy techniques, the novel method described in the patent utilizes a base-promoted reaction between readily available diazo compounds and trifluoroethylimidoyl chlorides. This approach completely bypasses the need for organic azides and transition metal catalysts, fundamentally altering the safety and economic profile of the synthesis. The reaction proceeds smoothly in common aprotic solvents like acetonitrile at moderate temperatures ranging from 50°C to 70°C. By employing cesium carbonate as a promoter, the process achieves high conversion rates and excellent yields without the generation of heavy metal waste. This metal-free strategy not only simplifies the downstream purification process but also significantly reduces the environmental footprint of the manufacturing operation.
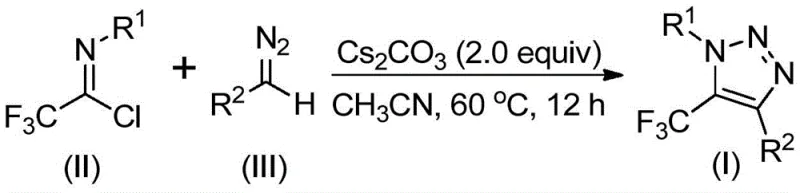
The versatility of this new methodology is evidenced by its broad substrate scope, accommodating a wide array of functional groups on both the imidoyl chloride and the diazo components. As illustrated in the reaction scheme, the process tolerates substituents such as halogens, alkoxy groups, and even phosphonate moieties, enabling the rapid generation of diverse chemical libraries for drug discovery programs. For supply chain heads concerned with the commercial scale-up of complex pharmaceutical intermediates, this robustness is a critical asset. The ability to source cheap and stable starting materials, combined with a simple workup procedure involving filtration and column chromatography, ensures a reliable and continuous supply of high-quality intermediates. This innovation effectively decouples triazole production from the volatility of the azide market, providing a more stable and predictable manufacturing baseline.
Mechanistic Insights into Base-Promoted Cyclization
Understanding the mechanistic pathway of this transformation is essential for R&D teams aiming to optimize the process for specific substrates. The reaction is hypothesized to initiate with a base-promoted intermolecular nucleophilic addition-elimination process. Specifically, the diazo compound, activated by the basic environment provided by cesium carbonate, acts as a nucleophile attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This step facilitates the formation of a crucial carbon-carbon bond while eliminating a chloride ion. The resulting intermediate then undergoes an intramolecular 5-endo-dig cyclization, a kinetically favorable process that closes the five-membered triazole ring. This mechanistic sequence is distinct from the concerted cycloaddition mechanisms seen in click chemistry, offering different regioselectivity profiles that can be exploited to access unique substitution patterns at the 1 and 4 positions of the triazole ring.
The choice of base and solvent plays a pivotal role in driving this mechanism to completion. Cesium carbonate is identified as the optimal promoter due to its solubility profile in organic solvents and its moderate basicity, which is sufficient to activate the diazo species without causing premature decomposition. Aprotic solvents like acetonitrile are preferred as they effectively dissolve the reactants and stabilize the charged intermediates formed during the nucleophilic attack. The presence of 4Å molecular sieves in the reaction mixture further enhances efficiency by sequestering trace moisture, which could otherwise hydrolyze the sensitive imidoyl chloride starting material. This attention to reaction conditions underscores the importance of precise process control in achieving the high yields reported in the patent data, where yields often exceed 80% for a variety of substrates.
Furthermore, the electronic nature of the substituents on the aromatic rings of the starting materials significantly influences the reaction kinetics. Electron-withdrawing groups on the imidoyl chloride enhance the electrophilicity of the imine carbon, accelerating the initial nucleophilic attack. Conversely, electron-donating groups on the diazo compound can increase its nucleophilicity. The patent data demonstrates that the system is robust enough to tolerate both electron-rich and electron-poor substrates, although reaction times may need adjustment to maximize conversion. This mechanistic flexibility allows chemists to fine-tune the process for specific target molecules, ensuring that impurity profiles remain within acceptable limits. By avoiding radical pathways often associated with metal catalysis, this ionic mechanism provides a cleaner reaction trajectory, minimizing the formation of side products that are difficult to separate.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-plant production. The protocol involves a straightforward one-pot procedure where all reagents are combined in a single vessel, reducing the need for complex intermediate isolations. The reaction conditions are mild, requiring only standard heating equipment and inert atmosphere techniques commonly found in pharmaceutical manufacturing facilities. Detailed standard operating procedures regarding stoichiometry, addition rates, and quenching protocols are critical for maintaining consistency across batches. For technical teams looking to implement this technology, the following guide outlines the fundamental steps derived from the patent examples to ensure successful replication and scale-up.
- Combine cesium carbonate, trifluoroethylimidoyl chloride, and diazo compounds in an aprotic organic solvent such as acetonitrile within a reaction vessel.
- Heat the reaction mixture to a temperature range of 50°C to 70°C and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Perform post-treatment by filtration and silica gel mixing, followed by column chromatography purification to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this base-promoted synthesis offers transformative advantages for procurement and supply chain management. The elimination of transition metal catalysts removes the necessity for expensive scavenging resins and extensive purification steps typically required to meet residual metal specifications in pharmaceutical products. This simplification of the downstream processing directly translates to substantial cost savings in manufacturing overhead. Moreover, the replacement of hazardous azides with stable diazo compounds and imidoyl chlorides drastically reduces the safety infrastructure costs associated with handling explosive materials. Facilities can operate with lower insurance premiums and reduced regulatory burdens, enhancing the overall economic viability of producing these valuable heterocyclic intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of commodity chemicals. Cesium carbonate, while a specialty base, is used in catalytic or stoichiometric amounts and is far less expensive than proprietary trifluoromethylating reagents or palladium/copper catalysts. The starting materials, trifluoroethylimidoyl chlorides and diazo compounds, are synthesized from readily available precursors like aromatic amines and acid chlorides, ensuring a stable and low-cost supply chain. By avoiding the multi-step sequences often required to generate azide precursors, the overall step count is reduced, leading to higher overall throughput and lower labor costs per kilogram of product. This efficiency is crucial for maintaining competitiveness in the generic pharmaceutical market where margin pressure is intense.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by decoupling production from the volatile market for organic azides, which are often subject to strict transportation regulations and limited vendor availability. The reagents used in this novel method are stable solids or liquids that can be stored for extended periods without degradation, allowing manufacturers to maintain strategic stockpiles. This stability mitigates the risk of production stoppages due to raw material shortages. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent quality, resulting in more consistent batch-to-batch performance. For supply chain heads, this predictability is invaluable for long-term capacity planning and meeting delivery commitments to downstream customers.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns perfectly with modern sustainability goals. The absence of heavy metals eliminates the generation of toxic aqueous waste streams, simplifying wastewater treatment and reducing disposal costs. The use of acetonitrile, a solvent with well-established recovery and recycling protocols, further minimizes the environmental footprint. The patent explicitly notes that the method can be easily extended to the gram level and beyond, indicating excellent scalability. The exothermic profile of the reaction is manageable, reducing the engineering controls needed for heat dissipation compared to highly energetic azide reactions. This combination of safety, scalability, and environmental friendliness makes the technology an ideal candidate for green chemistry initiatives and regulatory filings that prioritize sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled a set of frequently asked questions based on the patent specifications. These answers provide clarity on the operational parameters, safety considerations, and applicability of the method for various industrial use cases. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The following responses are grounded in the experimental data and theoretical framework provided in the intellectual property documentation.
Q: What are the primary safety advantages of this new triazole synthesis method compared to traditional routes?
A: Unlike conventional copper-catalyzed azide-alkyne cycloadditions that require toxic and potentially explosive organic azides, this novel method utilizes stable diazo compounds and imidoyl chlorides, significantly enhancing operational safety and reducing hazardous waste handling costs.
Q: Can this metal-free process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent data explicitly demonstrates that the method can be easily extended to the gram level and beyond. The use of inexpensive bases like cesium carbonate and common solvents like acetonitrile facilitates straightforward commercial scale-up without the need for expensive transition metal removal steps.
Q: What is the substrate scope for the R1 and R2 groups in this triazole formation?
A: The reaction exhibits broad functional group tolerance. R1 can be alkyl, substituted or unsubstituted aryl groups, while R2 accommodates aroyl, phospholipid, alkoxycarbonyl, or trifluoromethyl groups, allowing for the design of diverse molecular scaffolds for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and optimizing production costs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the base-promoted triazole synthesis can be seamlessly translated into industrial reality. We are committed to delivering high-purity pharmaceutical intermediates that meet the most stringent purity specifications, utilizing our state-of-the-art rigorous QC labs to verify every batch. Our capability to handle complex fluorinated chemistry positions us as a strategic partner for global pharmaceutical companies seeking to secure their supply chains for next-generation therapeutics.
We invite you to collaborate with us to explore the full potential of this metal-free triazole synthesis for your specific projects. Whether you require custom synthesis services or bulk supply of key intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your manufacturing operations. Together, we can leverage this cutting-edge chemistry to bring life-saving medicines to market faster and more sustainably.
