Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Applications
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Pharmaceutical Applications
The landscape of heterocyclic chemistry is constantly evolving to meet the rigorous demands of modern drug discovery, particularly regarding the incorporation of fluorine motifs which enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel, efficient preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift from traditional transition-metal catalysis to a safer, base-promoted metal-free protocol. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding this synthetic innovation is crucial. The patent outlines a robust pathway that utilizes readily available trifluoroethylimidoyl chlorides and diazo compounds, bypassing the need for hazardous azides or expensive copper catalysts. This report provides a deep technical analysis of this methodology, highlighting its potential for cost reduction in API manufacturing and its suitability for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on two primary strategies, both of which present substantial drawbacks for large-scale industrial application. The first conventional approach involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation. This route is plagued by the inherent toxicity of copper residues, which necessitates costly and complex purification steps to meet stringent regulatory limits for active pharmaceutical ingredients. Furthermore, the second traditional method utilizes organocatalytic 1,3-dipolar cycloaddition involving organic azides and trifluoromethyl ketones. The critical bottleneck here is safety; organic azides are thermally unstable and possess high explosive potential, creating severe risks during handling and scale-up. These safety concerns often mandate specialized equipment and rigorous safety protocols, drastically inflating production costs and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these hazardous and metal-dependent legacy methods, the technology disclosed in patent CN113121462B introduces a remarkably simple yet powerful base-promoted synthesis. This novel approach leverages the reactivity of trifluoroethylimidoyl chlorides and diazo compounds under mild basic conditions, completely eliminating the need for transition metal catalysts or explosive azide reagents. As illustrated in the reaction scheme below, this method (specifically pathway c in the comparative figure) offers a direct route to the target scaffold with high atom economy.
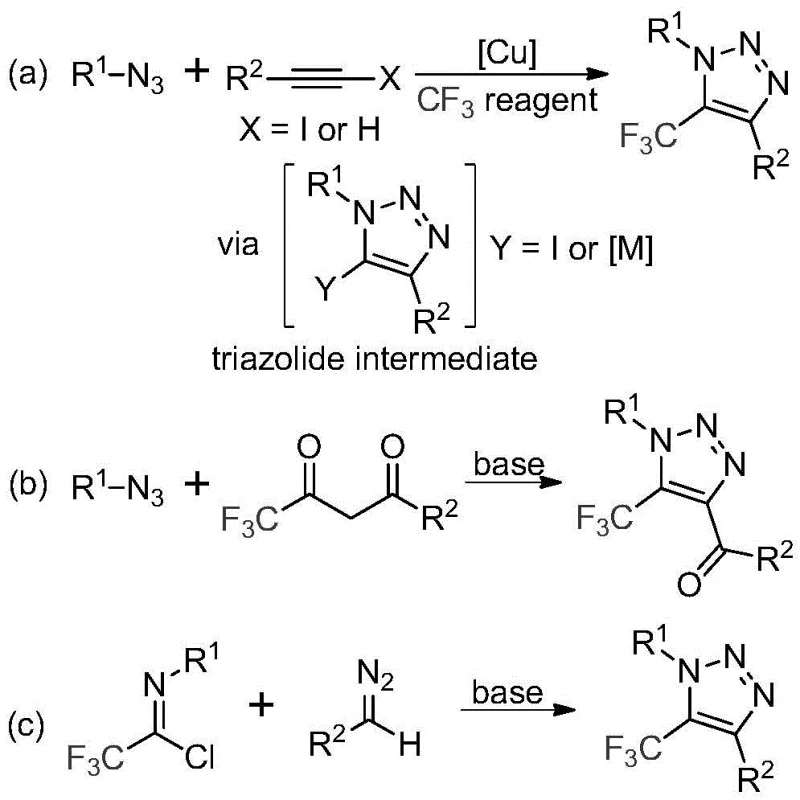
By shifting the mechanistic foundation from metal catalysis to base promotion using inexpensive salts like cesium carbonate, this process fundamentally alters the economic and safety profile of the synthesis. The avoidance of heavy metals not only simplifies the downstream purification workflow but also aligns perfectly with green chemistry principles, reducing the environmental footprint of the manufacturing process. This makes it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates while ensuring a safer working environment for production staff.
Mechanistic Insights into Base-Promoted Cyclization
To fully appreciate the technical elegance of this synthesis, one must delve into the proposed reaction mechanism which drives the formation of the 1,2,3-triazole ring. The process initiates with a base-promoted intermolecular nucleophilic addition-elimination sequence. Specifically, the diazo compound acts as a nucleophile, attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This step is facilitated by the presence of a mild base, such as cesium carbonate, which activates the diazo species without causing premature decomposition. The elimination of the chloride ion generates a key intermediate that sets the stage for ring closure. This initial C-C bond formation is critical and is highly dependent on the electronic nature of the substituents on both the imidoyl chloride and the diazo compound, allowing for significant tunability in the final product structure.
Following the initial addition-elimination, the reaction proceeds through an intramolecular 5-endo-dig cyclization. This cyclization step is the defining moment where the linear precursor folds into the characteristic five-membered triazole ring. The driving force for this cyclization is the stabilization of the resulting aromatic heterocyclic system. The general reaction equation below clearly depicts the transformation of starting materials (II) and (III) into the final product (I), highlighting the loss of HCl which is scavenged by the base.
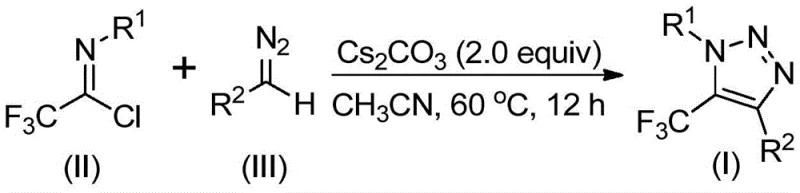
From an impurity control perspective, this mechanism offers distinct advantages. Because the reaction does not involve radical pathways or unstable metal complexes, the impurity profile is generally cleaner and more predictable. The primary side reactions typically involve the hydrolysis of the imidoyl chloride or the dimerization of the diazo compound, both of which can be effectively suppressed by controlling moisture levels (using molecular sieves as noted in the patent) and optimizing the stoichiometry of the base. The use of 4Å molecular sieves is a strategic choice to sequester trace water, thereby preventing the hydrolysis of the sensitive imidoyl chloride starting material and ensuring high conversion rates.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity. The patent provides a detailed protocol that balances reagent ratios, temperature, and reaction time to achieve optimal results. The procedure is designed to be robust, tolerating a variety of functional groups on both the nitrogen and carbon substituents of the triazole ring. Below is a summary of the standardized approach derived from the patent examples, which serves as a guideline for process chemists aiming to replicate this high-efficiency route.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, diazo compound, and 4Å molecular sieves in an organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature between 50°C and 70°C and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Filter the reaction mixture, concentrate, and purify the crude product via column chromatography to isolate the target 5-trifluoromethyl-1,2,3-triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from traditional copper-catalyzed methods to this base-promoted protocol offers tangible strategic benefits beyond mere chemical curiosity. The economic implications of removing expensive catalysts and hazardous reagents are profound, directly impacting the cost of goods sold (COGS) and the reliability of the supply chain. By adopting this methodology, manufacturers can mitigate risks associated with raw material volatility and regulatory compliance regarding heavy metal residues.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the elimination of transition metal catalysts. Copper salts, especially those used in specialized cross-coupling or cycloaddition reactions, can be costly, and their removal often requires expensive scavengers or extensive chromatography. By utilizing cesium carbonate, a commodity chemical, the catalyst cost is drastically reduced. Furthermore, the simplified workup procedure—often requiring only filtration and standard chromatography—reduces solvent consumption and labor hours. This streamlined process translates to significant cost savings in the overall manufacturing budget, making the production of high-purity pharmaceutical intermediates more economically viable without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of starting materials. Traditional methods relying on organic azides face supply bottlenecks due to safety regulations restricting their transport and storage. In contrast, the diazo compounds and imidoyl chlorides used in this novel method are commercially available and stable under standard storage conditions. This stability allows for bulk purchasing and long-term inventory planning, reducing the risk of production stoppages. Additionally, the mild reaction conditions (50-70°C) reduce energy consumption and equipment wear, further enhancing the reliability and continuity of the manufacturing schedule for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges. The metal-free nature of this synthesis inherently reduces the generation of heavy metal waste, simplifying wastewater treatment and disposal compliance. The patent explicitly notes that the method is easily extendable to the gram level and beyond, indicating strong potential for multi-kilogram or ton-scale production. The use of common organic solvents like acetonitrile, which can be readily recovered and recycled, aligns with modern sustainability goals. This environmental compatibility not only reduces disposal costs but also future-proofs the manufacturing process against tightening environmental regulations, ensuring long-term operational viability for agrochemical intermediate and pharmaceutical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in patent CN113121462B, offering clarity on the practical aspects of adopting this method for industrial applications.
Q: Why is this metal-free synthesis preferred over copper-catalyzed methods?
A: Traditional copper-catalyzed methods often require toxic transition metals that are difficult to remove to ppm levels required for APIs. This novel base-promoted method eliminates heavy metal contamination risks entirely, simplifying purification and ensuring higher purity standards for pharmaceutical applications.
Q: What are the safety advantages of using diazo compounds over organic azides?
A: Organic azides are notoriously unstable and potentially explosive, posing significant safety hazards during scale-up. The disclosed method utilizes stable diazo compounds and imidoyl chlorides, significantly reducing explosion risks and improving operational safety in commercial manufacturing environments.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly states the method is easily extendable to the gram level and beyond. The use of mild reaction conditions (50-70°C), inexpensive bases like cesium carbonate, and simple workup procedures makes it highly suitable for commercial scale-up and continuous manufacturing processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
The technological advancements detailed in patent CN113121462B underscore the dynamic nature of modern organic synthesis, where safety, efficiency, and cost-effectiveness converge. At NINGBO INNO PHARMCHEM, we recognize the critical importance of such innovations in driving the next generation of therapeutic agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of base-promoted heterocyclic synthesis, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We are committed to translating cutting-edge patent literature into reliable, commercial-grade supply chains for our global partners.
We invite R&D directors and procurement leaders to explore how this advanced synthesis route can optimize your project timelines and budgets. By leveraging our technical expertise, you can accelerate the development of your fluorine-containing drug candidates with confidence. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments tailored to your unique molecular targets, ensuring a seamless transition from bench-scale discovery to full-scale commercial manufacturing.
