Advanced Base-Promoted Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously demand robust synthetic routes for heterocyclic scaffolds that offer both metabolic stability and lipophilicity. Patent CN113121462A introduces a transformative preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical bottlenecks in current manufacturing paradigms. This technology leverages a metal-free, base-promoted cyclization strategy that bypasses the inherent dangers of traditional azide chemistry while delivering high reaction efficiency. By utilizing readily available trifluoroacetimidoyl chlorides and diazo compounds, this innovation provides a safer, more scalable pathway for generating diverse triazole libraries essential for modern drug discovery and functional material development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted triazoles has relied heavily on copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by subsequent trifluoromethylation steps. These conventional pathways present severe operational challenges, primarily due to the use of organic azides, which are notoriously toxic and possess explosive properties that complicate handling and storage on a large scale. Furthermore, the reliance on transition metal catalysts necessitates rigorous downstream purification to remove trace metal residues, a requirement that is particularly stringent in the production of active pharmaceutical ingredients (APIs). The combination of hazardous reagents, expensive catalysts, and complex purification protocols results in elevated production costs and significant safety liabilities for chemical manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN113121462A offers a streamlined alternative that eliminates the need for both transition metals and unstable azide precursors. This novel approach utilizes a direct condensation between trifluoroacetimidoyl chlorides and diazo compounds, promoted by a simple inorganic base such as cesium carbonate. 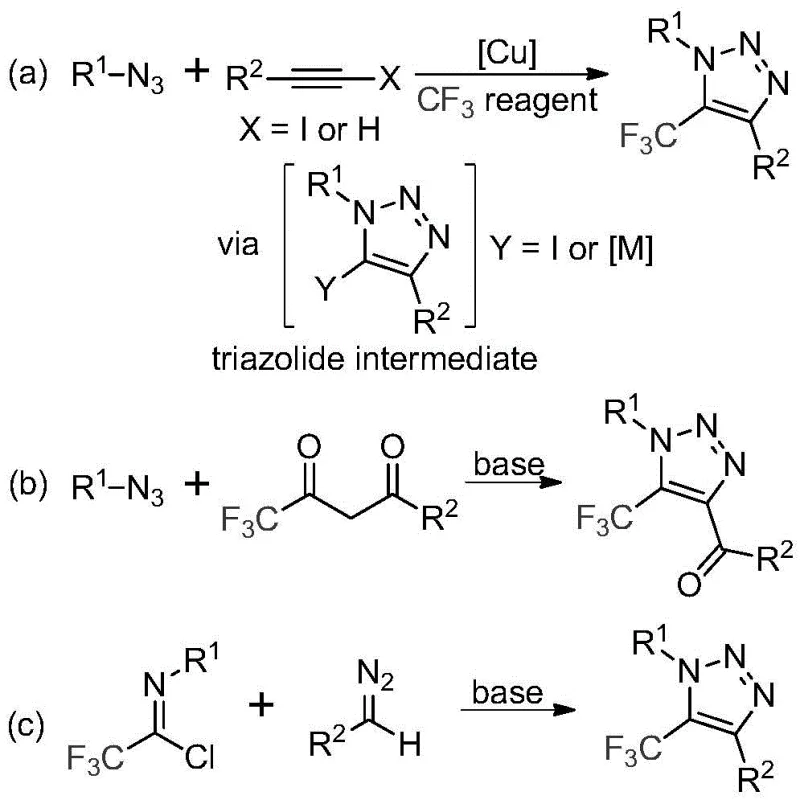 The reaction proceeds under mild thermal conditions, typically between 50 and 70 degrees Celsius, in common organic solvents like acetonitrile. By shifting the mechanistic paradigm from metal-catalyzed cycloaddition to base-promoted nucleophilic addition and cyclization, this method not only enhances operational safety but also simplifies the reaction workflow, making it highly attractive for cost reduction in pharmaceutical intermediate manufacturing.
The reaction proceeds under mild thermal conditions, typically between 50 and 70 degrees Celsius, in common organic solvents like acetonitrile. By shifting the mechanistic paradigm from metal-catalyzed cycloaddition to base-promoted nucleophilic addition and cyclization, this method not only enhances operational safety but also simplifies the reaction workflow, making it highly attractive for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Base-Promoted Cyclization
The core of this synthetic breakthrough lies in a unique intermolecular nucleophilic addition-elimination process followed by an intramolecular cyclization. The reaction initiates with the base-mediated activation of the diazo compound, which acts as a nucleophile attacking the electrophilic carbon of the trifluoroacetimidoyl chloride. This step forms a key intermediate that subsequently undergoes a 5-endo-dig cyclization to close the triazole ring. 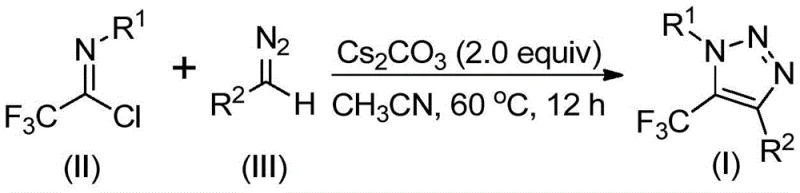 This mechanism avoids the formation of metal-carbene species or high-energy azide intermediates, thereby reducing the risk of side reactions and decomposition. The tolerance of various functional groups on both the imidoyl chloride and the diazo substrate allows for the precise tuning of the final molecular architecture, enabling the synthesis of complex derivatives without compromising yield or purity.
This mechanism avoids the formation of metal-carbene species or high-energy azide intermediates, thereby reducing the risk of side reactions and decomposition. The tolerance of various functional groups on both the imidoyl chloride and the diazo substrate allows for the precise tuning of the final molecular architecture, enabling the synthesis of complex derivatives without compromising yield or purity.
From an impurity control perspective, the absence of transition metals fundamentally alters the impurity profile of the final product. Traditional copper-catalyzed routes often generate metal-complexed byproducts that are difficult to separate and can catalyze degradation during storage. The metal-free nature of this new protocol ensures that the primary impurities are organic in origin, which are generally easier to remove via standard chromatographic techniques or crystallization. This results in a cleaner crude reaction mixture and facilitates the attainment of high-purity specifications required for regulatory compliance in the fine chemical sector.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The operational simplicity of this protocol makes it accessible for both laboratory optimization and pilot plant execution. The process involves mixing the trifluoroacetimidoyl chloride, the specific diazo compound, and cesium carbonate in an anhydrous organic solvent, typically acetonitrile, in the presence of molecular sieves to maintain dryness. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined below to guide technical teams in replicating these high-yielding results.
- Combine cesium carbonate, molecular sieves, trifluoroacetimidoyl chloride, and the specific diazo compound in an organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50 to 70 degrees Celsius and maintain stirring for 8 to 16 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical novelty. The shift away from specialized, hazardous reagents towards commodity chemicals significantly de-risks the supply chain. Since the starting materials—trifluoroacetimidoyl chlorides and diazo compounds—are either commercially available or easily synthesized from bulk precursors, manufacturers can secure raw material supplies with greater confidence and lower volatility. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of eliminating transition metal catalysts cannot be overstated. By removing the need for expensive copper sources and specialized ligands, the direct material cost of the reaction is substantially lowered. Moreover, the absence of metals obviates the need for costly metal scavenging resins and extensive analytical testing for residual metals, which are mandatory steps in API production. This streamlining of the downstream processing workflow leads to significant operational expenditure savings and improved overall process economics.
- Enhanced Supply Chain Reliability: The reliance on stable, non-explosive reagents simplifies logistics and storage requirements. Unlike organic azides, which often require special permits and controlled transportation due to their instability, the reagents used in this method can be handled with standard safety protocols. This ease of handling reduces administrative burdens and insurance costs, while also minimizing the risk of production stoppages caused by the unavailability of specialized hazardous materials. Consequently, suppliers can offer more reliable lead times and consistent availability of high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions, operating at moderate temperatures of 50 to 70 degrees Celsius, are inherently safer and more energy-efficient than high-temperature or high-pressure alternatives. This thermal mildness facilitates easier scale-up from gram to kilogram and ton scales without requiring exotic reactor configurations. Furthermore, the generation of less hazardous waste streams aligns with increasingly strict environmental regulations, supporting sustainable manufacturing practices and reducing the costs associated with waste disposal and environmental compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel triazole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: How does this method improve safety compared to traditional triazole synthesis?
A: Traditional methods often rely on organic azides, which are known to be toxic and potentially explosive. This novel protocol utilizes stable diazo compounds and trifluoroacetimidoyl chlorides under mild basic conditions, significantly reducing safety hazards in the laboratory and plant.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly demonstrates that the reaction can be expanded to gram-level scales with high efficiency. The use of inexpensive bases like cesium carbonate and common solvents like acetonitrile facilitates straightforward scale-up for industrial production.
Q: What represents the primary cost advantage of this synthetic route?
A: The primary cost advantage stems from the elimination of transition metal catalysts, such as copper, and the avoidance of complex trifluoromethylating reagents. This removes the need for expensive metal scavenging steps and simplifies the downstream purification process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain a competitive edge in the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like the base-promoted triazole synthesis can be successfully translated into industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage this efficient, metal-free technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and accelerate your time to market.
