Advanced Visible-Light Mediated Arylation Strategy for High-Purity Tetrahydroquinoline Derivatives
Advanced Visible-Light Mediated Arylation Strategy for High-Purity Tetrahydroquinoline Derivatives
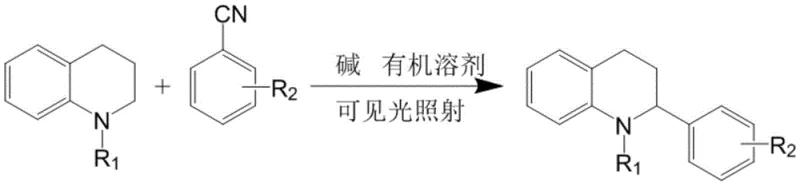
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient synthetic pathways to access complex heterocyclic scaffolds essential for drug discovery and development. A significant breakthrough in this domain is documented in Chinese Patent CN112812060B, which discloses a novel arylation method for tetrahydroquinoline derivatives. This technology leverages visible light photocatalysis to achieve C-H functionalization without the need for transition metal catalysts or harsh oxidants. For R&D directors and procurement specialists, this represents a paradigm shift towards sustainable manufacturing, offering a route that minimizes heavy metal contamination risks while maintaining high structural diversity. The method utilizes N-substituted 1,2,3,4-tetrahydroquinoline and benzonitrile derivatives as key starting materials, reacting them under mild conditions to produce valuable arylated products. This approach not only addresses the growing regulatory pressure regarding residual metals in APIs but also simplifies the downstream purification processes, thereby enhancing overall process efficiency and cost-effectiveness for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis and modification of tetrahydroquinoline derivatives have relied heavily on classical organic transformations that often involve multi-step sequences or the use of expensive transition metal catalysts such as palladium, copper, or rhodium. These conventional methods frequently require stringent reaction conditions, including high temperatures, inert atmospheres, and the use of stoichiometric oxidants, which can lead to significant safety hazards and environmental burdens. Furthermore, the reliance on metal catalysts introduces a critical bottleneck in pharmaceutical manufacturing: the removal of trace metal residues to meet strict regulatory limits for active pharmaceutical ingredients (APIs). This necessitates additional purification steps, such as specialized scavenging or recrystallization, which drastically increase production costs and extend lead times. Additionally, traditional hydrogenation methods to obtain substituted tetrahydroquinolines often suffer from selectivity issues, leading to complex impurity profiles that are difficult to characterize and control, ultimately compromising the quality and consistency of the final product.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN112812060B introduces a streamlined, metal-free protocol that utilizes visible light irradiation to drive the arylation reaction. By employing simple inorganic bases and common organic solvents, this innovative approach eliminates the need for precious metal catalysts and external oxidants, fundamentally altering the economic and environmental landscape of tetrahydroquinoline synthesis. The reaction proceeds smoothly at room temperature under blue LED light, demonstrating exceptional functional group tolerance and operational simplicity. This green chemistry strategy not only reduces the carbon footprint of the manufacturing process but also inherently improves the purity profile of the resulting intermediates by avoiding metal contamination entirely. The ability to directly couple N-substituted tetrahydroquinolines with benzonitrile derivatives opens up vast chemical space for medicinal chemists to explore new biologically active molecules with improved potency and selectivity, making it a highly attractive option for the rapid development of new drug candidates.
Mechanistic Insights into Visible-Light Mediated C-H Arylation
The core innovation of this technology lies in its unique mechanistic pathway that activates the C-H bond of the tetrahydroquinoline ring through a photo-induced electron transfer process. Under the irradiation of blue light, the reaction system generates reactive radical intermediates without the assistance of a photocatalyst metal complex, suggesting a direct excitation or base-mediated activation mechanism. This radical species then attacks the electron-deficient benzonitrile derivative, facilitating the formation of a new carbon-carbon bond at the desired position. The absence of metal catalysts means that the reaction kinetics are governed purely by organic photophysics and base strength, allowing for precise tuning of the reaction rate by adjusting the light intensity and base concentration. For process chemists, understanding this mechanism is crucial for optimizing reaction parameters to maximize yield while minimizing side reactions such as over-arylation or polymerization. The robustness of this radical pathway ensures that even sterically hindered substrates can be effectively coupled, providing a reliable method for constructing complex molecular architectures that are otherwise difficult to access through traditional nucleophilic substitution or cross-coupling reactions.
From an impurity control perspective, this metal-free mechanism offers distinct advantages for the production of high-purity pharmaceutical intermediates. Since no transition metals are introduced into the reaction matrix, the risk of forming metal-organic complexes or leaching toxic metals into the product stream is completely eradicated. This significantly simplifies the analytical workload required for release testing, as extensive ICP-MS screening for heavy metals becomes unnecessary. Furthermore, the mild reaction conditions prevent the thermal degradation of sensitive functional groups often present in advanced intermediates, thereby preserving the integrity of the molecular scaffold. The use of inorganic bases like cesium carbonate or potassium phosphate ensures that the reaction medium remains relatively neutral to slightly basic, avoiding acid-catalyzed decomposition pathways that could generate hard-to-remove byproducts. Consequently, the final product exhibits a cleaner impurity profile, which is critical for ensuring patient safety and meeting the rigorous quality standards demanded by global regulatory agencies for drug substances.
How to Synthesize Tetrahydroquinoline Arylation Derivatives Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must follow a precise sequence of mixing and irradiation steps to ensure optimal conversion and yield. The process begins with the careful selection of high-quality starting materials, specifically the N-substituted tetrahydroquinoline and the chosen benzonitrile derivative, which dictate the final structural properties of the product. The reaction is typically conducted in polar aprotic solvents such as N,N-dimethylacetamide or dimethyl sulfoxide, which facilitate the dissolution of both organic substrates and the inorganic base. Once the reagents are combined in the specified molar ratios, the mixture is subjected to continuous stirring under blue LED illumination at ambient temperature. Detailed standardized synthesis steps for scaling this process are provided in the guide below.
- Mix N-substituted 1,2,3,4-tetrahydroquinoline, benzonitrile derivative, and inorganic base in an organic solvent such as DMAc or DMF.
- Stir the reaction mixture at room temperature under blue LED light irradiation for 15 to 40 hours to facilitate the arylation process.
- Perform workup procedures including extraction with ethyl acetate, rotary evaporation, and column chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this visible-light mediated arylation technology presents a compelling value proposition centered around cost reduction and supply reliability. By eliminating the dependency on scarce and volatile transition metal catalysts, manufacturers can insulate their production costs from the fluctuations of the precious metals market. The simplified workflow, which removes the need for specialized metal scavenging resins and complex filtration units, translates directly into lower capital expenditure and reduced operational overhead. Moreover, the use of commodity chemicals like inorganic bases and common organic solvents ensures a stable and diversified supply chain, mitigating the risk of raw material shortages that often plague specialized reagent markets. This resilience is particularly vital for long-term commercial contracts where consistent delivery and predictable pricing are paramount for maintaining competitive advantage in the global marketplace.
- Cost Reduction in Manufacturing: The most significant economic benefit of this process stems from the complete removal of expensive metal catalysts and oxidants from the bill of materials. Traditional cross-coupling reactions often require palladium or copper salts that constitute a substantial portion of the raw material cost, alongside the ancillary costs of ligands and activators. By switching to a metal-free photochemical system, the direct material costs are drastically lowered, allowing for more aggressive pricing strategies without sacrificing margin. Additionally, the energy consumption is minimized since the reaction runs at room temperature, avoiding the high utility costs associated with heating large reactors or maintaining cryogenic conditions. The streamlined workup procedure further reduces labor costs and solvent usage, contributing to a leaner and more profitable manufacturing operation that maximizes return on investment for every kilogram produced.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable raw materials significantly enhances the robustness of the supply chain. N-substituted tetrahydroquinolines and benzonitrile derivatives are widely produced commodity chemicals with multiple qualified suppliers globally, reducing the risk of single-source dependency. The inorganic bases used, such as sodium carbonate or cesium acetate, are stable solids with long shelf lives, simplifying inventory management and warehousing requirements. This accessibility ensures that production schedules can be maintained consistently, even during periods of global supply chain disruption. For supply chain heads, this means shorter lead times for raw material procurement and greater flexibility to ramp up production volumes in response to sudden increases in market demand, ensuring uninterrupted service to downstream pharmaceutical customers.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge, but recent advancements in flow chemistry and LED reactor design have made this technology highly scalable for industrial applications. The absence of hazardous oxidants and the generation of minimal waste streams align perfectly with modern environmental, health, and safety (EHS) standards. This green profile simplifies the permitting process for new manufacturing facilities and reduces the costs associated with waste disposal and treatment. The ability to run the reaction at ambient pressure and temperature also lowers the safety risks associated with high-pressure hydrogenation or exothermic oxidation reactions. Consequently, this method supports sustainable growth, allowing companies to expand capacity while meeting increasingly stringent corporate sustainability goals and regulatory compliance requirements for green chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light arylation technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios. It clarifies the operational boundaries and potential advantages, ensuring that expectations are aligned with the actual capabilities of the technology.
Q: Does this arylation method require expensive transition metal catalysts?
A: No, the patented method described in CN112812060B operates under metal-free conditions, utilizing visible light and inorganic bases instead of costly palladium or copper catalysts, which significantly simplifies purification and reduces heavy metal impurities.
Q: What are the typical reaction conditions for this tetrahydroquinoline modification?
A: The reaction proceeds under mild conditions at room temperature using blue LED light irradiation. It typically requires 15 to 40 hours of stirring in polar aprotic solvents like N,N-dimethylacetamide, avoiding the need for high heat or pressure.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process demonstrates excellent industrial application value due to its simple operation, absence of oxidants, and use of readily available raw materials, making it highly scalable for commercial production of complex heterocyclic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free visible-light arylation technology for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory method into a robust, commercial-scale manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of photochemical parameters is maintained even in large reactors. We are committed to delivering products with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the highest industry standards for impurity profiles and residual solvent content. Our state-of-the-art facilities are equipped to handle the specific requirements of photochemical synthesis, guaranteeing consistent quality and supply continuity for your critical projects.
We invite you to collaborate with us to unlock the full commercial potential of these high-value tetrahydroquinoline derivatives. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. By partnering with us, you gain access to our deep process knowledge and supply chain network, enabling you to accelerate your drug development timelines. Please contact us today to request specific COA data for our available intermediates and to discuss detailed route feasibility assessments for your custom synthesis needs. Let us help you optimize your supply chain with innovative, green chemistry solutions.
