Advanced Asymmetric Catalysis for High-Purity Chiral Gamma-Butyrolactone Pharmaceutical Intermediates
Introduction to Patent CN101250171A and Chiral Synthesis
The landscape of modern pharmaceutical manufacturing is increasingly defined by the demand for single-enantiomer compounds, driven by stringent regulatory requirements and the biological necessity for precise molecular recognition. Patent CN101250171A represents a significant technological breakthrough in this domain, disclosing a novel method for constructing chiral gamma-butyrolactone ring structures through asymmetric allyl alkylation. This specific class of compounds serves as a critical scaffold in the synthesis of numerous bioactive natural products and therapeutic agents. The core innovation lies in the utilization of specifically designed chiral phosphine compounds as organocatalysts, which facilitate the reaction between alpha-acetoxy ketones and 2-substituted trimethylsilylfurans. Unlike traditional methods that rely on stoichiometric chiral auxiliaries or expensive transition metal complexes, this approach leverages the nucleophilic properties of phosphines to induce high levels of stereocontrol. For R&D directors and process chemists, this patent offers a robust pathway to access complex chiral intermediates with exceptional enantiomeric purity, addressing the persistent challenge of efficient asymmetric bond formation in fine chemical synthesis.
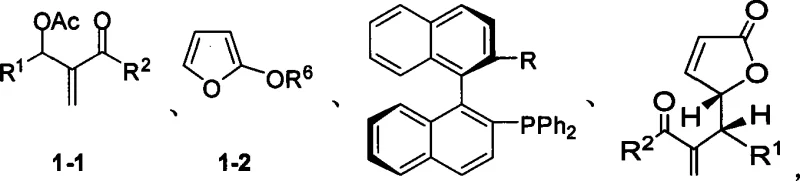
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of optically pure gamma-butyrolactones has been fraught with inefficiencies that hinder cost-effective manufacturing. The most traditional approach, racemic resolution, is inherently wasteful because it discards at least half of the synthesized material, capping the maximum theoretical yield at 50%. Furthermore, resolution processes often require multiple crystallization steps or chromatographic separations, which drastically increase solvent consumption, processing time, and overall production costs. Alternatively, the chiral pool method relies on naturally occurring chiral starting materials, which limits structural diversity and is subject to supply chain volatility regarding the source materials. While transition metal-catalyzed asymmetric synthesis has gained popularity, it frequently necessitates rigorous exclusion of air and moisture, expensive ligand systems, and extensive downstream purification to remove trace metal residues that are toxic and strictly regulated in pharmaceutical products. These factors collectively create substantial bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in CN101250171A circumvents these legacy issues by employing a chiral phosphine organocatalytic system. This novel approach enables the direct asymmetric coupling of readily available achiral precursors to form the desired chiral center with high fidelity. A distinguishing feature of this technology is its tolerance to reaction conditions that would typically deactivate sensitive metal catalysts; specifically, the reaction can be conducted in the presence of water as an additive, which surprisingly enhances stereoselectivity. By operating at room temperature and avoiding the need for cryogenic conditions or inert atmosphere gloveboxes, the process simplifies the engineering requirements for reactor setup. This shift from metal-dependent catalysis to organocatalysis not only improves the environmental profile of the synthesis by reducing heavy metal waste but also streamlines the purification workflow, as there is no need for specialized metal scavenging resins. Consequently, this method offers a more sustainable and economically viable route for the commercial scale-up of complex chiral intermediates.
Mechanistic Insights into Chiral Phosphine-Catalyzed Asymmetric Allyl Alkylation
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing process parameters and ensuring consistent product quality. The reaction proceeds via a nucleophilic catalysis cycle where the chiral phosphine catalyst attacks the electrophilic allylic carbon of the alpha-acetoxy ketone substrate. This initial attack generates a zwitterionic phosphonium enolate intermediate, which is stabilized by the chiral environment provided by the binaphthyl backbone of the catalyst. The chirality of the catalyst dictates the facial selectivity of the subsequent nucleophilic attack by the 2-trimethylsilylfuran. The specific spatial arrangement of the substituents on the catalyst framework creates a steric pocket that favors the formation of one enantiomer over the other, leading to the high enantiomeric excess (ee) values observed in the experimental data. The presence of water plays a pivotal role in this mechanism, likely by facilitating proton transfer steps or stabilizing transition states through hydrogen bonding networks, thereby accelerating the turnover frequency without compromising stereocontrol.
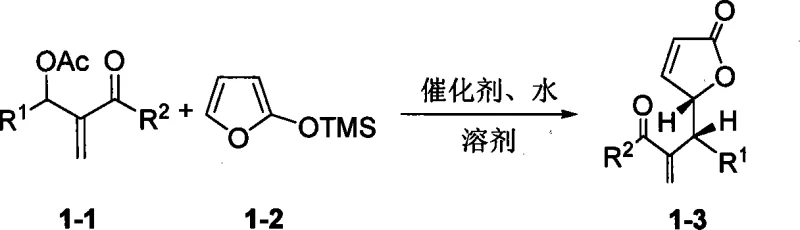
From an impurity control perspective, the mechanism offers distinct advantages. Since the catalyst is an organic small molecule rather than a transition metal complex, the risk of metal-induced side reactions, such as beta-hydride elimination or unwanted oxidation, is minimized. The primary impurities typically arise from non-enantioselective background reactions or hydrolysis of the acetoxy group, both of which can be managed by fine-tuning the catalyst loading and water equivalents. The patent data demonstrates that by selecting specific catalyst variants, such as L3 or L7, and optimizing the solvent system to include toluene with controlled water content, the formation of racemic byproducts is suppressed. This high level of chemoselectivity ensures that the crude reaction mixture is cleaner, reducing the burden on downstream purification units and enhancing the overall mass balance of the manufacturing process.
How to Synthesize Chiral Gamma-Butyrolactone Efficiently
The practical implementation of this synthesis route requires careful attention to reagent quality and mixing protocols to maximize the benefits of the catalytic system. The general procedure involves dissolving the alpha-acetoxy ketone substrate and the silylfuran nucleophile in an organic solvent, followed by the addition of the chiral phosphine catalyst. A critical operational parameter identified in the patent is the intentional addition of water, which acts as a promoter rather than a poison. The reaction is typically stirred at ambient temperature for a period ranging from 24 to 90 hours, depending on the electronic nature of the substrate substituents. Upon completion, the product is isolated via standard aqueous workup and purified using flash column chromatography. For detailed operational specifics regarding stoichiometry, solvent ratios, and workup procedures, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining the alpha-acetoxy ketone substrate and 2-trimethylsilylfuran in an organic solvent such as toluene.
- Add the chiral phosphine catalyst (e.g., L3 or L7) and a controlled amount of water as an additive to promote enantioselectivity.
- Stir the reaction at room temperature for 24 to 90 hours, then purify the resulting chiral gamma-butyrolactone via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology translates into tangible strategic benefits beyond mere chemical yield. The shift towards organocatalysis eliminates the dependency on precious metals like palladium or platinum, the prices of which are subject to significant geopolitical and market volatility. By removing the requirement for expensive metal scavengers and the associated validation testing for residual metals, the overall cost of goods sold (COGS) is substantially reduced. Furthermore, the robustness of the reaction conditions—specifically the tolerance to moisture and operation at room temperature—lowers the energy consumption associated with heating, cooling, and maintaining inert atmospheres. This energy efficiency contributes directly to a lower carbon footprint for the manufacturing process, aligning with corporate sustainability goals and reducing utility costs.
- Cost Reduction in Manufacturing: The elimination of transition metals removes the need for costly purification steps dedicated to metal removal, which often involve expensive functionalized silica or polymer resins. Additionally, the high atom economy of the allylic alkylation reaction ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal fees. The ability to use commodity solvents like toluene instead of specialized fluorinated or chlorinated solvents further drives down raw material expenses, making the process economically competitive for large-volume production.
- Enhanced Supply Chain Reliability: The catalysts described, such as the binaphthyl-based phosphines, are synthetically accessible from abundant starting materials, reducing the risk of supply bottlenecks associated with rare earth elements or proprietary ligand systems. The reaction's insensitivity to trace moisture means that raw materials do not require rigorous drying protocols, simplifying inventory management and reducing the lead time for raw material qualification. This operational flexibility allows for more agile manufacturing schedules and reduces the risk of batch failures due to environmental excursions.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to stoichiometric chiral resolution methods, as there is no discard of the unwanted enantiomer. The absence of heavy metals simplifies the regulatory filing process for new drug applications, as toxicological concerns regarding metal residues are mitigated. The mild reaction conditions also reduce the thermal load on manufacturing facilities, allowing for safer scale-up from pilot plant to commercial production without requiring specialized high-pressure or cryogenic reactor infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing pipelines.
Q: What is the primary advantage of using chiral phosphine catalysts over traditional resolution methods?
A: Traditional resolution methods are limited to a maximum theoretical yield of 50% and require complex separation steps. In contrast, the asymmetric catalytic method described in CN101250171A allows for the generation of large quantities of optically pure product from achiral starting materials with high atom economy and significantly reduced waste.
Q: How does the addition of water impact the reaction performance?
A: Contrary to traditional moisture-sensitive organometallic reactions, this protocol utilizes water as a beneficial additive. Optimization data indicates that adding approximately 6 equivalents of water can significantly enhance both the chemical yield and enantiomeric excess (ee), reaching up to 94% yield and 93% ee in specific substrates.
Q: Is this method suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process operates under mild conditions (room temperature) and uses commercially available solvents like toluene. The elimination of strict anhydrous requirements and the use of robust organocatalysts simplify the engineering controls needed for scale-up, making it highly viable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Gamma-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral building blocks for the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN101250171A can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee the enantiomeric excess and chemical purity of every batch. Our commitment to quality assurance means that clients can rely on us for consistent supply of complex pharmaceutical intermediates that meet global regulatory standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative catalytic route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this organocatalytic method. We encourage you to contact us to obtain specific COA data for related gamma-butyrolactone derivatives and to receive comprehensive route feasibility assessments that will accelerate your drug development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
