Advanced Rhodium-Catalyzed Synthesis of Chiral Gamma-Butyrolactone Derivatives for Oncology Drug Development
Advanced Rhodium-Catalyzed Synthesis of Chiral Gamma-Butyrolactone Derivatives for Oncology Drug Development
The pharmaceutical industry is constantly seeking robust and scalable methodologies for constructing complex chiral scaffolds, particularly those found in bioactive natural products and potent therapeutic agents. Patent CN111196791B introduces a groundbreaking approach to synthesizing chiral gamma-butyrolactone derivatives, a structural motif prevalent in over ten percent of natural compound molecules. This innovation addresses the critical need for efficient access to optically active gamma-butyrolactones, which serve as pivotal intermediates in the development of anti-cancer, anti-inflammatory, and antiviral medications. The disclosed technology leverages a sophisticated dual-catalyst system to achieve high stereoselectivity under remarkably mild conditions, representing a significant leap forward in synthetic methodology for fine chemical manufacturing.
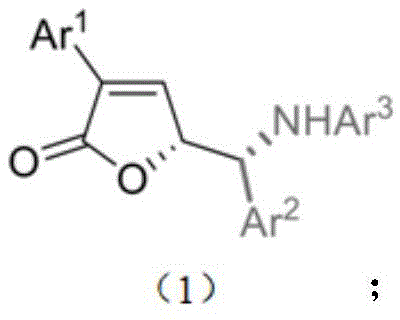
These novel derivatives, characterized by the general structure shown in Formula 1, possess unique substitution patterns that enhance their biological efficacy. Specifically, the patent highlights their potent inhibitory activity against human breast cancer cells (MCF-7) and human lung adenocarcinoma cells (A549). For R&D directors and procurement specialists in the oncology sector, this represents a valuable new entry point for drug discovery pipelines. The ability to access these complex architectures through a streamlined, one-pot transformation not only accelerates lead optimization but also lays the groundwork for cost-effective commercial production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active gamma-butyrolactone derivatives has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Traditional routes often rely on harsh reaction conditions, such as extreme temperatures or highly acidic environments, which can degrade sensitive functional groups and compromise the integrity of the final product. Furthermore, many existing methodologies require unstable or difficult-to-obtain raw materials, leading to supply chain vulnerabilities and inflated costs for the resulting API intermediates. The multi-step nature of conventional syntheses frequently results in low overall yields and poor atom economy, generating substantial chemical waste that complicates environmental compliance and disposal protocols.
From a process chemistry perspective, the lack of stereocontrol in older methods necessitates cumbersome resolution steps to isolate the desired enantiomer, drastically reducing throughput and increasing manufacturing lead times. These inefficiencies create bottlenecks for pharmaceutical companies aiming to bring new therapies to market rapidly. The operational complexity associated with these legacy processes also poses safety risks in industrial settings, requiring specialized equipment and rigorous monitoring to prevent runaway reactions or hazardous exposures. Consequently, there has been a persistent demand for a more reliable pharmaceutical intermediate supplier capable of delivering these motifs through a safer, greener, and more economically viable pathway.
The Novel Approach
The methodology described in CN111196791B fundamentally disrupts the status quo by employing a direct coupling strategy between 3-aryl cyclopropene carboxylic acids and imines. This novel approach eliminates the need for pre-functionalized precursors and reduces the synthetic sequence to a single, highly efficient transformation. By utilizing cheap and easily available compounds as starting materials, the process significantly lowers the barrier to entry for cost reduction in API manufacturing. The reaction proceeds under mild conditions, typically at ambient temperature (25°C), which minimizes energy consumption and reduces the thermal stress on reactor infrastructure, thereby enhancing operational safety and equipment longevity.
Moreover, this innovative route boasts exceptional atom economy and selectivity, directly addressing the waste generation issues inherent in traditional synthesis. The high yields reported, often exceeding 90%, coupled with the simplicity of the workup procedure involving standard rotary evaporation and column chromatography, streamline the purification process. This efficiency translates directly into improved supply chain reliability, as fewer processing steps mean fewer opportunities for material loss or quality deviations. For supply chain heads, this translates to a more predictable and scalable production model that can adapt quickly to fluctuating market demands for complex oncology intermediates without compromising on purity or delivery schedules.
Mechanistic Insights into Rhodium and Chiral Phosphoric Acid Cooperative Catalysis
The core of this technological breakthrough lies in the synergistic interaction between a dirhodium catalyst and a chiral phosphoric acid organocatalyst. The reaction mechanism initiates with the activation of the 3-aryl cyclopropene carboxylic acid by the dirhodium species, likely forming a reactive metal-carbenoid intermediate. This highly electrophilic species is then poised for nucleophilic attack by the imine substrate. The presence of the chiral phosphoric acid is critical, as it acts as a Brønsted acid activator for the imine while simultaneously providing a chiral environment through hydrogen bonding interactions. This dual activation mode ensures that the bond formation occurs with precise spatial orientation, dictating the stereochemical outcome of the cyclization.
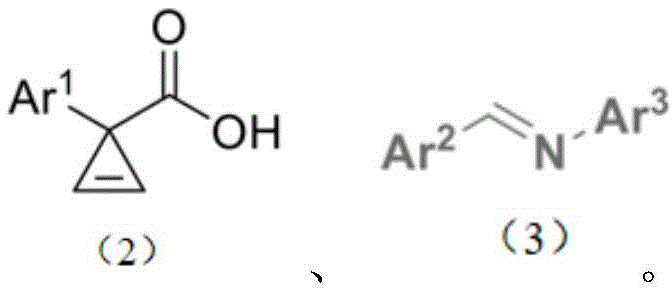
This cooperative catalysis model allows for exquisite control over both diastereoselectivity and enantioselectivity. The chiral pocket created by the bulky substituents on the phosphoric acid catalyst shields one face of the reacting species, forcing the approach of the nucleophile from the less hindered trajectory. As a result, the process consistently delivers products with diastereomeric ratios (dr) greater than 95:5 and enantiomeric ratios (er) reaching up to 98:2. Such high levels of optical purity are paramount for pharmaceutical applications, where the wrong enantiomer can be inactive or even toxic. The mechanistic elegance of this system ensures that impurities are minimized at the source, reducing the burden on downstream purification units and ensuring a cleaner impurity profile for the final drug substance.
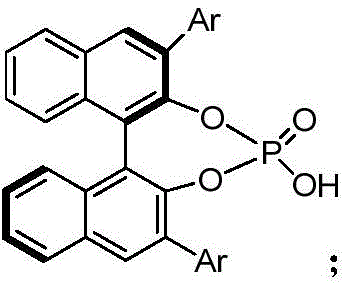
Furthermore, the choice of catalyst loading and reaction parameters has been optimized to balance reaction rate with selectivity. The use of tetrahydrofuran as the preferred solvent facilitates the solubility of both organic substrates and the catalyst complex, promoting homogeneous reaction kinetics. The tolerance of various substituents on the aryl rings of both the cyclopropene and the imine demonstrates the robustness of the catalytic cycle. Whether electron-withdrawing groups like nitro or halogens, or electron-donating groups are present, the system maintains its efficacy. This broad substrate scope is a testament to the versatility of the catalytic mechanism, making it a powerful tool for generating diverse libraries of chiral gamma-butyrolactones for structure-activity relationship (SAR) studies in drug discovery.
How to Synthesize Chiral Gamma-Butyrolactone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific protocol parameters to ensure reproducibility and optimal yield. The process is designed to be operationally simple, avoiding the need for exotic equipment or inert atmosphere techniques beyond standard laboratory practices. The key to success lies in the precise stoichiometric balance of the reactants and catalysts, as well as maintaining the recommended reaction time to allow the slow but highly selective cyclization to reach completion. Detailed standard operating procedures regarding reagent quality and mixing rates are essential for scaling this chemistry from gram to kilogram quantities.
- Prepare the reaction mixture by dissolving 3-aryl cyclopropene carboxylic acid and imine substrates in tetrahydrofuran solvent.
- Add the dual catalyst system comprising bis[(alpha,alpha,alpha',alpha'-tetramethyl-1,3-benzenedipropionic acid) rhodium] and chiral phosphoric acid.
- Stir the reaction at 25°C for 96 hours, then purify the crude product via column chromatography to obtain the high-purity derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented synthesis route offers compelling economic and logistical benefits that extend beyond mere technical feasibility. The shift towards this methodology aligns with modern green chemistry principles, which are increasingly becoming a prerequisite for vendor qualification in the global pharmaceutical market. By simplifying the synthetic route and utilizing benign reaction conditions, manufacturers can achieve substantial cost savings through reduced utility consumption and lower waste disposal fees. The reliance on commercially available, inexpensive starting materials further insulates the supply chain from volatility associated with specialty reagents, ensuring a stable and continuous flow of critical intermediates.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and the avoidance of expensive protecting group strategies inherently lower the cost of goods sold (COGS). Since the reaction proceeds with high atom economy, the amount of raw material required per unit of product is minimized, directly impacting the bottom line. Additionally, the mild temperature requirements eliminate the need for energy-intensive heating or cryogenic cooling systems, resulting in significant operational expenditure reductions. The high selectivity of the process also means that less material is lost to side products, maximizing the value extracted from every kilogram of input material and driving down the effective cost per gram of the high-purity chiral gamma-butyrolactone.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term drug development projects. This method utilizes robust, shelf-stable reagents that are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The simplicity of the reaction setup reduces the likelihood of batch failures due to operational errors, leading to more consistent production schedules. For supply chain heads, this predictability is invaluable, as it allows for more accurate inventory planning and reduces the need for excessive safety stock. The ability to scale this reaction from small discovery batches to commercial production without changing the fundamental chemistry ensures a seamless transition through the drug development lifecycle.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental footprint of a synthesis route is a key differentiator. This process generates minimal waste and avoids the use of hazardous heavy metals or toxic solvents often found in alternative methods. The straightforward workup procedure facilitates easier solvent recovery and recycling, further enhancing the sustainability profile of the manufacturing process. From a scalability perspective, the exothermic profile of the reaction is manageable, allowing for safe scale-up in standard stainless steel reactors. This ease of commercial scale-up of complex oncology intermediates ensures that production capacity can be expanded rapidly to meet clinical trial demands or commercial launch volumes without requiring massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this chiral gamma-butyrolactone synthesis technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this chemistry into their existing manufacturing portfolios or drug discovery programs.
Q: What are the primary advantages of this Rhodium-catalyzed method over traditional lactone synthesis?
A: This method utilizes mild reaction conditions (25°C) and readily available raw materials, avoiding the harsh conditions and unstable reagents often required in conventional multi-step syntheses, thereby improving safety and atom economy.
Q: What level of stereoselectivity can be achieved with this process?
A: The process demonstrates exceptional stereocontrol, achieving diastereomeric ratios (dr) greater than 95:5 and enantiomeric ratios (er) up to 98:2, which is critical for producing single-enantiomer pharmaceutical ingredients.
Q: Does this compound show biological activity relevant to drug development?
A: Yes, the synthesized derivatives exhibit significant inhibitory effects against human breast cancer cells (MCF-7) and human lung adenocarcinoma cells (A549), validating their potential as oncology drug candidates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Gamma-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route disclosed in CN111196791B for advancing oncology therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of rhodium-catalyzed reactions, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We are committed to delivering high-purity chiral gamma-butyrolactone derivatives that empower your research and development teams to push the boundaries of cancer treatment.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug candidate. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Together, we can accelerate the delivery of life-saving medicines to patients worldwide while optimizing your supply chain efficiency and cost structure.
