Advanced Organocatalytic Synthesis of Chiral Gamma-Butyrolactones for Commercial API Production
Introduction to Patent CN111233795A Technology
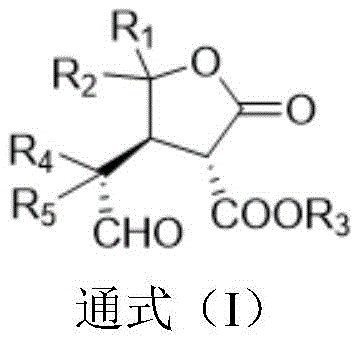
The pharmaceutical industry constantly seeks robust methodologies for constructing complex chiral scaffolds, particularly those found in bioactive natural products. Patent CN111233795A introduces a groundbreaking preparation method for chiral gamma-butyrolactone compounds and their derivatives, addressing a critical gap in the synthesis of molecules with three contiguous chiral centers. This technology leverages an organic catalytic asymmetric Michael addition reaction between aldehyde compounds and 2(5H)-furanones, achieving yields up to 89% with remarkable stereoselectivity (9.2:1 dr and 99% ee). For R&D directors and procurement specialists, this represents a significant advancement in generating high-purity pharmaceutical intermediates without the baggage of transition metal residues. The ability to construct such dense stereochemical complexity under mild conditions positions this method as a cornerstone for developing next-generation anti-tumor agents and other bioactive therapeutics.
Furthermore, the versatility of this synthetic platform allows for the generation of a broad library of derivatives, including hydrazones and various functionalized lactones, which have demonstrated inhibitory activity against multiple cancer cell lines such as U251 and Saos2. By integrating this patented approach into your supply chain, organizations can secure a reliable source of complex chiral building blocks that are otherwise difficult and expensive to procure. The following analysis details the mechanistic advantages, operational protocols, and commercial implications of adopting this organocatalytic strategy for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of gamma-butyrolactone skeletons with multiple chiral centers has relied heavily on stoichiometric chiral auxiliaries or transition metal catalysis, both of which present substantial drawbacks for modern GMP manufacturing. Metal-catalyzed processes often require rigorous purification steps to reduce residual metal levels to parts-per-million (ppm) standards, adding significant cost and time to the production cycle. Additionally, many conventional routes necessitate harsh reaction conditions, such as cryogenic temperatures or strong bases, which limit substrate scope and increase safety risks in a plant environment. The reliance on precious metals also introduces supply chain volatility, as the availability and price of catalysts like palladium or rhodium can fluctuate wildly, impacting the overall cost of goods sold (COGS) for the final API.
The Novel Approach
In contrast, the methodology disclosed in CN111233795A utilizes small organic molecule catalysis to drive the asymmetric Michael addition, effectively bypassing the need for toxic heavy metals. This organocatalytic approach operates efficiently at room temperature (25-30°C) in common solvents like toluene, drastically simplifying the engineering requirements for reactor setups. The use of chiral organic catalysts, such as the cinchona alkaloid derivatives shown in the patent, ensures high enantioselectivity while maintaining a "green" chemical profile that aligns with increasingly stringent environmental regulations. This shift not only reduces the environmental footprint but also streamlines the downstream processing, as the removal of organic catalysts is generally more straightforward than stripping trace metals from complex organic matrices.

Mechanistic Insights into Organocatalytic Asymmetric Michael Addition
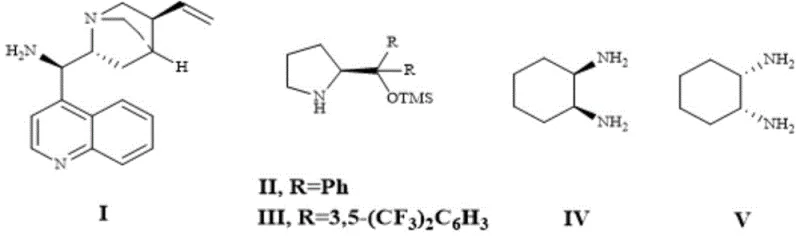
The core of this technological breakthrough lies in the precise activation of the nucleophile and electrophile through non-covalent interactions facilitated by the chiral catalyst. In this system, the chiral organic catalyst, preferably Compound II as depicted in the patent data, likely activates the aldehyde component via enamine formation or hydrogen bonding networks, rendering the alpha-carbon nucleophilic. Simultaneously, the catalyst organizes the 2(5H)-furanone electrophile within a chiral pocket, directing the attack to a specific face of the double bond. This dual activation mechanism is crucial for establishing the three consecutive stereocenters with high fidelity, preventing the formation of unwanted diastereomers that would complicate purification. The result is a highly convergent synthesis where complexity is generated in a single bond-forming event, maximizing atom economy and minimizing waste generation.
From an impurity control perspective, the high diastereoselectivity (dr > 9:1) and enantiomeric excess (ee > 99%) observed in this process significantly reduce the burden on crystallization and chromatography steps. In conventional syntheses, separating closely related stereoisomers often requires multiple recrystallizations or preparative HPLC, leading to substantial yield losses. By controlling the stereochemistry at the source through catalyst design, this method ensures that the crude product is already of high optical purity. This mechanistic elegance translates directly to process robustness, allowing for consistent batch-to-batch quality which is a paramount concern for regulatory filings and commercial supply agreements.
How to Synthesize Chiral Gamma-Butyrolactone Efficiently
To implement this synthesis in a laboratory or pilot plant setting, the process begins with the careful selection of the aldehyde and furanone substrates, which are mixed in a molar ratio ranging from 1:0.5 to 1:5, with a preferred ratio of 1:2 to drive conversion. The reaction is conducted in toluene under a nitrogen atmosphere to prevent oxidation of sensitive intermediates, stirred at ambient temperature for a period of 12 to 48 hours depending on the specific substrate reactivity. Upon completion, the reaction mixture undergoes a standard aqueous workup involving extraction with ethyl acetate and drying over anhydrous sodium sulfate, followed by purification via silica gel column chromatography. For further derivatization, the resulting aldehyde intermediate can be reacted with p-methylbenzenesulfonyl hydrazide to form stable hydrazone derivatives, expanding the utility of the core scaffold for medicinal chemistry campaigns.
- React 2(5H)-furanone derivative (1A) with an aldehyde (2A) in toluene using a chiral organic catalyst (e.g., Compound II) at room temperature for 12-48 hours.
- Perform post-reaction workup including extraction with ethyl acetate, drying over anhydrous sodium sulfate, and purification via silica gel column chromatography.
- Optionally react the resulting aldehyde intermediate (3A) with p-methylbenzenesulfonyl hydrazide in dichloromethane to form the stable hydrazone derivative (4A).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic route offers tangible strategic benefits beyond mere chemical elegance. The elimination of transition metals from the synthetic pathway removes a major bottleneck in vendor qualification and raw material sourcing, as there is no longer a need to audit suppliers for heavy metal specifications or invest in specialized scavenging resins. This simplification of the supply chain enhances reliability and reduces the lead time associated with quality control testing, allowing for faster turnover from synthesis to final API formulation. Furthermore, the use of commodity solvents like toluene and the ability to run reactions at room temperature significantly lower the energy consumption and infrastructure costs compared to cryogenic or high-pressure processes.
- Cost Reduction in Manufacturing: The organocatalytic nature of this process inherently lowers the cost of goods by removing the expense of precious metal catalysts and the associated purification technologies required to meet regulatory limits. By avoiding complex metal removal steps, manufacturers can reduce solvent usage and waste disposal costs, leading to substantial overall savings in the production budget. Additionally, the high yields and stereoselectivity minimize the loss of valuable starting materials, ensuring that the maximum amount of input is converted into saleable product.
- Enhanced Supply Chain Reliability: Relying on organic catalysts derived from abundant natural sources like cinchona alkaloids mitigates the geopolitical and economic risks associated with mining-dependent metal supply chains. This stability ensures a continuous flow of critical intermediates, preventing production stoppages due to catalyst shortages. The robustness of the reaction conditions also means that the process is less susceptible to variations in utility supply, such as cooling water temperature fluctuations, further securing the manufacturing timeline.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic metals make this process highly scalable from gram to multi-ton quantities without significant re-engineering. This scalability is complemented by a reduced environmental footprint, as the process generates less hazardous waste and avoids the discharge of heavy metals into the environment. Compliance with green chemistry principles not only satisfies regulatory bodies but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral gamma-butyrolactone synthesis technology. These insights are derived directly from the experimental data and advantageous effects reported in the patent documentation, providing a clear picture of what partners can expect regarding performance and applicability. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production pipelines or new drug development projects.
Q: What are the stereoselectivity metrics for this organocatalytic process?
A: The patented process achieves exceptional stereocontrol, with diastereomeric ratios (dr) up to 9.2:1 and enantiomeric excess (ee) values reaching 99%, ensuring high purity of the chiral gamma-butyrolactone scaffold.
Q: Why is organocatalysis preferred over metal catalysis for these intermediates?
A: Organocatalysis eliminates the risk of heavy metal contamination, which is critical for pharmaceutical applications. It also operates under mild conditions (room temperature), reducing energy costs and simplifying the supply chain for sensitive substrates.
Q: Can this synthetic route be scaled for industrial production?
A: Yes, the reaction utilizes common solvents like toluene and proceeds at ambient temperatures without requiring cryogenic conditions or high-pressure equipment, making it highly amenable to commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Gamma-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the organocatalytic synthesis described in CN111233795A for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the high enantiomeric excess and diastereomeric ratios promised by this technology, guaranteeing that every batch meets the exacting standards required for clinical and commercial use.
We invite you to collaborate with our technical team to explore how this cost-effective and environmentally friendly synthesis can optimize your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits tailored to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of innovation, reliability, and scientific excellence.
