Advanced Base-Catalyzed Synthesis of 2H-Pyran-2-One Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access biologically active scaffolds, particularly the versatile 2H-pyran-2-one core found in numerous antiviral and anticancer agents. Patent CN111646964B introduces a groundbreaking base-catalyzed methodology that addresses long-standing challenges in constructing this privileged structure. This innovation utilizes sulfur ylides as nucleophiles reacting with cyclopropenones to form carbon-carbon bonds efficiently, followed by a seamless cyclization step. Unlike conventional approaches that often suffer from low yields and cumbersome purification, this novel route offers a direct, one-pot solution that significantly streamlines the synthetic workflow. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and economically viable manufacturing protocols for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2H-pyran-2-one derivatives has been plagued by significant technical hurdles that impact both cost and timeline. Traditional methods frequently rely on multi-step sequences involving sensitive reagents, strict anhydrous conditions, or expensive transition metal catalysts that are difficult to remove to ppm levels required for API production. These legacy processes often exhibit poor atom economy, generating substantial chemical waste and requiring extensive downstream processing to isolate the pure product. Furthermore, the harsh reaction conditions typically employed can lead to the degradation of sensitive functional groups, limiting the scope of substrates that can be utilized. For procurement managers, these inefficiencies translate into higher raw material costs and unpredictable supply chains due to the complexity of sourcing specialized catalysts and managing hazardous waste disposal.
The Novel Approach
The method disclosed in the patent revolutionizes this landscape by employing a simple base-catalyzed insertion reaction that proceeds under relatively mild thermal conditions. By leveraging the unique reactivity of sulfur ylides towards strained cyclopropenone rings, the process achieves ring expansion and lactonization in a single operational step. This approach drastically reduces the number of unit operations required, eliminating the need for intermediate isolation and reducing solvent consumption. The use of inexpensive inorganic bases such as sodium acetate or cesium carbonate further enhances the economic profile, making the process accessible for large-scale production. This strategic simplification not only accelerates the timeline from bench to plant but also aligns perfectly with modern green chemistry principles, offering a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Base-Catalyzed Cyclization
The core of this technological advancement lies in the precise mechanistic pathway where the sulfur ylide acts as a potent nucleophile attacking the electrophilic carbonyl or strained bond of the cyclopropenone. Under the influence of the base, the ylide generates a reactive species that inserts into the three-membered ring, triggering a cascade of bond reorganizations. This insertion is followed by the elimination of the sulfur moiety and subsequent intramolecular cyclization to form the stable six-membered lactone ring characteristic of 2H-pyran-2-ones. Understanding this mechanism is crucial for process optimization, as it highlights the importance of base strength and solvent polarity in driving the equilibrium towards the desired product while minimizing side reactions.
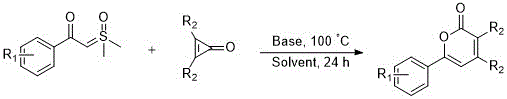
From an impurity control perspective, the mild nature of the base catalysis ensures that sensitive functional groups on the aromatic rings remain intact, preventing unwanted decomposition or polymerization. The reaction conditions, specifically the use of solvents like 1,2-dichloroethane at 100°C, provide sufficient energy to overcome the activation barrier for ring opening without promoting thermal degradation of the product. This high level of chemoselectivity results in a cleaner crude reaction profile, which simplifies the final purification via silica gel chromatography. For quality assurance teams, this means a more consistent impurity profile and higher overall purity of the final API intermediate, reducing the risk of batch failures during commercial production.
How to Synthesize 2H-Pyran-2-One Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of operation, requiring standard laboratory or plant equipment without the need for specialized high-pressure or cryogenic setups. The procedure involves a straightforward mixing of reagents followed by a controlled heating period, making it highly transferable from pilot scale to full commercial production. Detailed standardized operating procedures regarding stoichiometry, addition rates, and workup protocols are essential to maximize yield and reproducibility. The detailed standardized synthesis steps are outlined below for technical reference.
- Combine sulfur ylide, cyclopropenone, base (e.g., sodium acetate), and solvent (e.g., 1,2-dichloroethane) in a reactor.
- Heat the mixture to 100°C and stir continuously for 24 hours to facilitate the insertion reaction and cyclization.
- Upon completion, concentrate the reaction mixture and purify the residue via silica gel column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this base-catalyzed route offers transformative benefits that extend beyond mere chemical efficiency. The reliance on commodity chemicals such as simple sulfur ylides and cyclopropenones, which are increasingly available from bulk chemical suppliers, mitigates the risk of raw material shortages. Furthermore, the elimination of precious metal catalysts removes a significant cost driver and simplifies the regulatory compliance burden associated with heavy metal residues in pharmaceutical products. This shift allows for a more resilient supply chain capable of responding rapidly to market demands for complex heterocyclic intermediates.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the synthetic route and the use of low-cost reagents. By replacing expensive transition metal catalysts with inexpensive inorganic bases like sodium acetate, the direct material costs are substantially lowered. Additionally, the reduction in processing steps leads to decreased labor hours, lower energy consumption for heating and cooling cycles, and reduced solvent waste disposal fees. These cumulative effects result in a significantly improved cost of goods sold (COGS), allowing for more competitive pricing strategies in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method contributes directly to supply chain stability by reducing dependency on niche reagents with long lead times. Since the key starting materials are structurally simple and can be synthesized via established industrial processes, the risk of supply disruption is minimized. The reaction's tolerance to various substituents also means that a single production line can be adapted to manufacture a wide range of derivatives, providing flexibility to pivot production based on customer demand. This adaptability ensures continuous availability of high-purity pharmaceutical intermediates even during periods of market volatility.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage quantities is facilitated by the use of standard solvents and moderate temperatures that pose minimal safety risks in large reactors. The absence of hazardous reagents or extreme pressure conditions simplifies the engineering controls required for safe operation. Moreover, the improved atom economy and reduced waste generation align with stringent environmental regulations, lowering the ecological footprint of the manufacturing process. This sustainability advantage is increasingly critical for maintaining partnerships with major pharmaceutical companies that prioritize green chemistry initiatives in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the patent data and practical experience in scaling similar organic transformations. They serve to clarify the operational parameters and potential benefits for stakeholders evaluating this technology for integration into their supply chains.
Q: What are the primary advantages of this base-catalyzed method over traditional synthesis routes?
A: This method eliminates the need for complex multi-step sequences and harsh reaction conditions often associated with traditional pyranone synthesis. By utilizing readily available sulfur ylides and cyclopropenones with simple inorganic bases, it achieves high atom economy and simplifies the purification process, leading to significant operational efficiencies.
Q: Is this synthetic route suitable for large-scale pharmaceutical manufacturing?
A: Yes, the process is highly scalable due to the use of common solvents like 1,2-dichloroethane and stable reagents that do not require specialized handling equipment. The reaction operates at a moderate temperature of 100°C, which is easily manageable in standard industrial reactors, ensuring robust supply chain continuity.
Q: What types of substituents are tolerated in this cyclization reaction?
A: The methodology demonstrates excellent functional group tolerance, accommodating various substituents on both the sulfur ylide and the cyclopropenone rings. Examples include methyl and chloro groups on the phenyl rings, indicating broad substrate compatibility for generating diverse libraries of bioactive intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2H-Pyran-2-One Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has thoroughly evaluated the base-catalyzed synthesis of 2H-pyran-2-one derivatives and confirmed its potential for seamless technology transfer. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves efficiently from the laboratory to the marketplace. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage this innovative chemistry for your specific drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate how our manufacturing capabilities can optimize your supply chain and reduce overall project costs. Let us be your trusted partner in delivering high-quality chemical solutions that drive your business forward.
