Scalable Base-Catalyzed Synthesis of Bioactive 2H-Pyran-2-One Derivatives for Commercial Production
Scalable Base-Catalyzed Synthesis of Bioactive 2H-Pyran-2-One Derivatives for Commercial Production
The proprietary intellectual property detailed within Chinese Patent CN111646964B elucidates a groundbreaking synthetic methodology for the construction of 2H-pyran-2-one derivatives, a privileged scaffold ubiquitous in medicinal chemistry. This specific class of heterocyclic compounds serves as a critical backbone for numerous biologically active agents, including those exhibiting potent anti-rhinovirus and anti-HIV activities. Historically, the assembly of this core structure has been plagued by synthetic inefficiencies, yet this novel base-catalyzed approach offers a transformative solution by leveraging the unique reactivity of sulfur ylides. By utilizing 1,2-dichloroethane as a solvent and employing a straightforward thermal protocol, this method achieves efficient C-C bond formation and subsequent cyclization in a single operational step. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this technology represents a significant leap forward in process intensification, offering a pathway to high-purity materials with reduced environmental footprint and simplified downstream processing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes toward 2H-pyran-2-one scaffolds have long been characterized by cumbersome multi-step sequences that inherently suffer from low overall yields and poor atom economy. Conventional methodologies often necessitate the use of harsh reaction conditions, including strong acids or expensive transition metal catalysts, which introduce significant challenges in terms of impurity profiles and heavy metal removal. These legacy processes frequently exhibit limited substrate compatibility, failing to tolerate sensitive functional groups that are often required for downstream biological evaluation. Furthermore, the reliance on complex protecting group strategies and tedious purification protocols drastically inflates the cost of goods sold and extends the lead time for high-purity pharmaceutical intermediates. Such inefficiencies create bottlenecks in the supply chain, making it difficult for manufacturers to respond agilely to market demands for novel therapeutic candidates.
The Novel Approach
In stark contrast, the innovative strategy disclosed in the patent data utilizes a base-catalyzed insertion reaction between sulfur ylides and cyclopropenones to construct the target heterocycle with remarkable efficiency. This approach capitalizes on the inherent ring strain of cyclopropenones, which act as activated coupling reagents susceptible to nucleophilic attack, thereby driving the reaction forward under relatively mild thermal conditions. The use of simple inorganic bases such as sodium acetate or cesium carbonate eliminates the need for costly and toxic transition metals, resulting in a cleaner reaction profile that is easier to manage on a commercial scale. This one-pot transformation not only streamlines the synthetic workflow but also enhances the overall safety of the manufacturing process by avoiding hazardous reagents. Consequently, this method provides a robust platform for the rapid generation of diverse libraries of 2H-pyran-2-one derivatives, facilitating accelerated drug discovery timelines.
Mechanistic Insights into Base-Catalyzed Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between the nucleophilic sulfur ylide and the electrophilic cyclopropenone species, driven by the thermodynamic relief of ring strain. Initially, the base facilitates the generation or stabilization of the reactive ylide species, which then attacks the carbonyl or double bond of the cyclopropenone ring. This nucleophilic addition triggers a ring-opening event, followed by an intramolecular cyclization that restores aromaticity or forms the stable lactone ring characteristic of the 2H-pyran-2-one system. The precise control of reaction temperature at 100°C ensures that the activation energy barrier is overcome without promoting deleterious side reactions or decomposition of the sensitive intermediates. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters for maximum throughput and minimal waste generation.
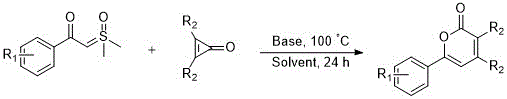
Furthermore, the impurity control mechanism inherent in this base-catalyzed system is superior to acid-catalyzed alternatives, as it minimizes the risk of polymerization or oligomerization of the reactive cyclopropenone starting material. The choice of solvent, specifically 1,2-dichloroethane, plays a pivotal role in solubilizing both the organic substrates and the inorganic base, creating a homogeneous or semi-homogeneous reaction medium that promotes consistent heat transfer. The mild basic conditions also prevent the hydrolysis of the newly formed lactone ring, ensuring high stability of the final product during the reaction course. This level of mechanistic understanding allows for fine-tuning of the molar ratios, typically maintaining a 1:1.0-2.0 ratio of ylide to cyclopropenone, to drive the reaction to completion while minimizing excess reagent costs. Such precision is vital for maintaining stringent purity specifications required by regulatory bodies for pharmaceutical applications.
How to Synthesize 2H-Pyran-2-One Derivatives Efficiently
The execution of this synthesis protocol is designed for operational simplicity, requiring standard laboratory or plant equipment without the need for specialized high-pressure or cryogenic setups. The process begins with the sequential charging of the sulfur ylide, cyclopropenone, base, and solvent into a clean reactor, followed by heating to the specified temperature for a defined period. Detailed standardized synthesis steps, including specific workup procedures and quality control checkpoints, are outlined in the technical guide below to ensure reproducibility and safety during scale-up operations. Adhering to these guidelines ensures that the full potential of this cost-reduction in pharmaceutical intermediates manufacturing is realized, delivering consistent batch-to-batch quality.
- Charge a clean reactor with sulfur ylide, cyclopropenone, a suitable base such as sodium acetate, and 1,2-dichloroethane solvent.
- Heat the reaction mixture to 100°C and maintain stirring for 24 hours to ensure complete conversion and ring closure.
- Upon completion, concentrate the reaction mixture and purify the crude residue via silica gel column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages for procurement managers and supply chain heads focused on cost optimization and reliability. The elimination of expensive transition metal catalysts and complex ligand systems translates directly into substantial cost savings on raw materials, while the simplified workup reduces solvent consumption and waste disposal fees. The use of commodity chemicals like sodium acetate and 1,2-dichloroethane ensures that the supply chain remains resilient against market volatility, as these reagents are widely available from multiple global vendors. Additionally, the robustness of the reaction conditions allows for flexible scheduling and easier integration into existing manufacturing facilities without requiring major capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom economy and the avoidance of precious metal catalysts, which removes the need for expensive scavenging steps to meet residual metal limits. By consolidating multiple synthetic transformations into a single pot, the process significantly reduces labor hours, energy consumption, and solvent usage, leading to a drastically simplified production workflow. This efficiency gain allows for a more competitive pricing structure for the final API intermediate, enhancing the overall margin potential for downstream drug products. Furthermore, the high yields reported in the patent examples indicate minimal material loss, maximizing the return on investment for every kilogram of starting material purchased.
- Enhanced Supply Chain Reliability: The reliance on readily accessible starting materials such as substituted sulfur ylides and cyclopropenones mitigates the risk of supply disruptions often associated with exotic or custom-synthesized reagents. Since the reaction does not require moisture-sensitive or air-sensitive conditions beyond standard inert atmosphere practices, logistics and storage requirements are less stringent, reducing overhead costs. This accessibility ensures that production schedules can be maintained consistently, reducing lead time for high-purity pharmaceutical intermediates and enabling faster response to urgent customer demands. The stability of the reagents also allows for bulk purchasing strategies, further securing the supply chain against short-term market fluctuations.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the straightforward thermal conditions and the use of common organic solvents that are easily recovered and recycled in a closed-loop system. The absence of heavy metals simplifies the environmental compliance landscape, reducing the burden of wastewater treatment and hazardous waste disposal, which aligns with modern green chemistry principles. This environmental friendliness not only lowers regulatory risks but also enhances the corporate sustainability profile of the manufacturing entity. The ability to scale from gram to multi-ton quantities without significant re-optimization makes this technology ideal for meeting the growing global demand for bioactive heterocycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this base-catalyzed synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this route for their specific product pipelines.
Q: What are the primary advantages of this base-catalyzed method over traditional synthesis routes?
A: This method eliminates the need for harsh conditions and multi-step sequences typical of conventional approaches. It utilizes readily available sulfur ylides and cyclopropenones to achieve high atom economy in a single pot, significantly simplifying the purification process and reducing overall production time.
Q: Is this synthetic route scalable for industrial manufacturing of pharmaceutical intermediates?
A: Yes, the process is highly amenable to scale-up. It employs common organic solvents like 1,2-dichloroethane and standard inorganic bases, avoiding exotic catalysts. The thermal requirement of 100°C is easily manageable in standard stainless steel reactors, ensuring robust supply chain continuity.
Q: What types of substituents are tolerated on the aromatic rings during this cyclization?
A: The reaction demonstrates excellent functional group tolerance. As evidenced by the patent data, both electron-donating groups like methyl and electron-withdrawing groups like chloro are compatible on the phenyl rings of the sulfur ylide, allowing for diverse structural modifications essential for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2H-Pyran-2-One Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics, and we are uniquely positioned to leverage this patented technology for your projects. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and risk-mitigated. We operate state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of 2H-pyran-2-one derivatives meets the highest industry standards for identity, strength, and quality. Our commitment to technical excellence means we can navigate the complexities of process optimization to deliver a product that supports your regulatory filings and clinical trials.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us immediately to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that accelerate your project timelines and enhance your competitive advantage in the marketplace.
