Advanced Benzimidazole Synthesis via CO2 Fixation: A Scalable Green Chemistry Solution for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking sustainable and efficient pathways to construct nitrogen-containing heterocycles, which serve as critical scaffolds in drug discovery. Patent CN112898205A introduces a groundbreaking methodology for the synthesis of benzimidazole derivatives by utilizing carbon dioxide and o-phenylenediamine compounds. This innovation represents a significant shift from traditional stoichiometric reagents to catalytic CO2 fixation, addressing both environmental concerns and economic inefficiencies. The core of this technology lies in the deployment of an amino-containing functionalized ordered mesoporous polymer as a heterogeneous catalyst. This specific catalyst architecture not only facilitates the activation of inert CO2 molecules but also ensures high catalytic activity under relatively mild reaction conditions. By integrating dimethylamino borane as a hydrogen reduction reagent in an NMP solvent system, the process achieves high yields while maintaining operational simplicity. For R&D directors and procurement specialists, this patent offers a compelling value proposition: a route that minimizes waste, reduces reliance on expensive carboxylic acid precursors, and leverages a recyclable catalytic system. The ability to synthesize high-purity benzimidazole intermediates using such a green chemistry approach positions this technology as a vital asset for modern supply chains aiming for sustainability and cost-effectiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole scaffolds has relied heavily on the condensation of o-phenylenediamine with carboxylic acids or their derivatives under strong acidic conditions. These traditional protocols often necessitate harsh reaction environments, including high temperatures and the use of corrosive mineral acids, which pose significant challenges for equipment longevity and operator safety. Furthermore, the alternative oxidative cyclization of o-phenylenediamine with aldehydes frequently results in complex reaction mixtures containing numerous by-products. This complexity drastically complicates the downstream purification process, requiring extensive chromatographic separation or recrystallization steps that lower the overall material throughput. From a supply chain perspective, the reliance on stoichiometric amounts of oxidizing agents or activated carboxylic acids increases the raw material cost burden and generates substantial chemical waste. The difficulty in separating homogeneous catalysts or acidic residues from the final product also raises concerns about metal contamination and residual impurities, which are critical quality attributes for pharmaceutical intermediates. Consequently, there is an urgent industrial need for a methodology that circumvents these operational bottlenecks while maintaining high selectivity and yield.
The Novel Approach
The methodology disclosed in patent CN112898205A offers a transformative solution by employing carbon dioxide as a sustainable C1 building block in the presence of a specialized heterogeneous catalyst. This novel approach eliminates the need for pre-functionalized carboxylic acids, thereby simplifying the raw material sourcing and reducing the associated procurement costs. The use of an amino-functionalized ordered mesoporous phenolic resin polymer ensures that the catalytic active sites are uniformly distributed and accessible, leading to superior reaction kinetics compared to conventional supported catalysts. 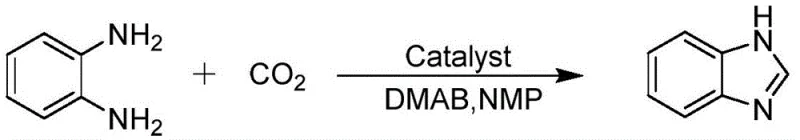 As illustrated in the reaction scheme, the process operates under mild pressure (0.1-2 MPa) and moderate temperatures (60-180°C), which significantly reduces energy consumption and safety risks associated with high-pressure reactors. The heterogeneous nature of the catalyst allows for straightforward filtration and recovery, enabling multiple reuse cycles without significant loss of activity. This not only enhances the economic viability of the process but also aligns with green chemistry principles by minimizing waste generation. For procurement managers, this translates to a more stable and predictable supply of high-purity benzimidazole intermediates, free from the complexities of traditional acidic workups.
As illustrated in the reaction scheme, the process operates under mild pressure (0.1-2 MPa) and moderate temperatures (60-180°C), which significantly reduces energy consumption and safety risks associated with high-pressure reactors. The heterogeneous nature of the catalyst allows for straightforward filtration and recovery, enabling multiple reuse cycles without significant loss of activity. This not only enhances the economic viability of the process but also aligns with green chemistry principles by minimizing waste generation. For procurement managers, this translates to a more stable and predictable supply of high-purity benzimidazole intermediates, free from the complexities of traditional acidic workups.
Mechanistic Insights into CO2 Fixation and Heterogeneous Catalysis
The success of this synthesis relies heavily on the unique structural properties of the amino-functionalized ordered mesoporous polymer catalyst. This material possesses a high specific surface area and a uniform pore channel arrangement, which facilitates the diffusion of reactants to the active sites while preventing the aggregation of catalytic species. The amino groups embedded within the polymer framework act as basic sites that activate the carbon dioxide molecule, promoting its nucleophilic attack on the o-phenylenediamine substrate.  Simultaneously, the dimethylamino borane serves as a mild yet effective reducing agent, driving the dehydration and cyclization steps forward without the need for harsh oxidants. The ordered mesoporous structure provides a confined environment that stabilizes the transition states, thereby lowering the activation energy required for the reaction. This mechanistic advantage allows the reaction to proceed with high selectivity, minimizing the formation of side products that typically plague homogeneous catalytic systems. For R&D teams, understanding this mechanism is crucial for optimizing reaction parameters such as solvent polarity and catalyst loading to achieve maximum efficiency. The robustness of the polymer support ensures that the catalyst maintains its structural integrity over multiple cycles, which is a key factor for long-term process stability.
Simultaneously, the dimethylamino borane serves as a mild yet effective reducing agent, driving the dehydration and cyclization steps forward without the need for harsh oxidants. The ordered mesoporous structure provides a confined environment that stabilizes the transition states, thereby lowering the activation energy required for the reaction. This mechanistic advantage allows the reaction to proceed with high selectivity, minimizing the formation of side products that typically plague homogeneous catalytic systems. For R&D teams, understanding this mechanism is crucial for optimizing reaction parameters such as solvent polarity and catalyst loading to achieve maximum efficiency. The robustness of the polymer support ensures that the catalyst maintains its structural integrity over multiple cycles, which is a key factor for long-term process stability.
Impurity control is another critical aspect where this novel catalytic system excels over traditional methods. In conventional acidic cyclizations, the formation of polymeric by-products or incomplete reaction intermediates is common, requiring rigorous purification to meet pharmaceutical standards. The heterogeneous nature of the mesoporous catalyst prevents the leaching of active species into the reaction medium, thereby ensuring that the final product is free from metal contamination. The mild reaction conditions also suppress thermal degradation pathways that often lead to the formation of tarry residues. Furthermore, the use of CO2 as a reagent avoids the introduction of extraneous carbon sources that could lead to isotopic impurities or structural analogs. The workup procedure, involving simple filtration and extraction, effectively removes the catalyst and soluble by-products, yielding a crude product of high purity. This inherent purity profile reduces the burden on downstream purification units, such as crystallization or chromatography, leading to significant time and cost savings in the manufacturing process. For quality assurance teams, this means a more consistent impurity profile and easier validation of the manufacturing process.
How to Synthesize Benzimidazole Efficiently
The practical implementation of this synthesis route involves a straightforward sequence of steps that can be easily adapted for both laboratory and pilot-scale operations. The process begins with the preparation of the catalyst, followed by the reaction setup in a pressure vessel, and concludes with a simple workup procedure. The detailed standardized synthesis steps are provided below to guide technical teams in replicating this efficient methodology. This structured approach ensures reproducibility and safety, making it an ideal candidate for technology transfer to commercial manufacturing sites.
- Preparation of the amino-functionalized ordered mesoporous phenolic resin polymer catalyst via condensation and calcination.
- Reaction of o-phenylenediamine with CO2 in NMP solvent using dimethylamino borane as a reductant and the polymer catalyst.
- Workup involving filtration to recover the heterogeneous catalyst, followed by extraction and purification to isolate the benzimidazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this CO2-fixation technology offers substantial strategic advantages in terms of cost structure and supply reliability. The shift from expensive carboxylic acid precursors to carbon dioxide represents a fundamental reduction in raw material costs, as CO2 is an abundant and inexpensive commodity chemical. Additionally, the recyclability of the heterogeneous catalyst eliminates the recurring cost of purchasing stoichiometric catalysts or oxidants, leading to significant long-term savings. The simplified workup procedure reduces the consumption of solvents and purification media, further driving down the variable costs associated with production. From a supply chain perspective, the robustness of the catalyst and the mild reaction conditions enhance the reliability of the manufacturing process, reducing the risk of batch failures or delays. The ability to scale this process from laboratory to commercial production without significant re-engineering ensures a continuous supply of critical intermediates. Furthermore, the green nature of the process aligns with increasingly stringent environmental regulations, mitigating the risk of compliance-related disruptions. These factors collectively contribute to a more resilient and cost-effective supply chain for benzimidazole derivatives.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants and carboxylic acid precursors drastically lowers the direct material costs associated with benzimidazole production. The heterogeneous catalyst can be recovered and reused multiple times without significant loss of activity, which amortizes the catalyst cost over a larger production volume. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility costs. The simplified purification process minimizes the need for expensive chromatographic resins or large volumes of recrystallization solvents. These cumulative efficiencies result in a substantially lower cost of goods sold, providing a competitive edge in the marketplace.
- Enhanced Supply Chain Reliability: The use of carbon dioxide as a feedstock ensures a stable and abundant supply of raw materials,不受 geographic or seasonal constraints that might affect agricultural or fermentation-derived precursors. The robustness of the mesoporous polymer catalyst ensures consistent performance across multiple batches, reducing the variability in production timelines. The straightforward reaction setup and workup procedure minimize the complexity of the manufacturing process, lowering the risk of operational errors or equipment downtime. This reliability is crucial for maintaining just-in-time inventory levels and meeting the demanding delivery schedules of downstream pharmaceutical customers. The ability to source raw materials locally and produce intermediates efficiently strengthens the overall resilience of the supply chain.
- Scalability and Environmental Compliance: The heterogeneous nature of the catalyst facilitates easy scale-up from gram to ton scale without the mass transfer limitations often encountered with homogeneous systems. The process generates minimal waste, as the catalyst is recycled and the by-products are easily separated, aligning with green chemistry principles. This reduced environmental footprint simplifies the permitting process and lowers the costs associated with waste disposal and treatment. The use of CO2 as a carbon source also contributes to carbon capture and utilization goals, enhancing the sustainability profile of the manufacturing site. These factors make the process highly attractive for large-scale commercial production in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzimidazole synthesis technology. These answers are derived directly from the patent data and practical considerations for industrial application. This section aims to provide clarity on the feasibility and benefits of adopting this novel catalytic route.
Q: What are the advantages of using a heterogeneous catalyst for benzimidazole synthesis?
A: The use of an amino-functionalized ordered mesoporous polymer allows for easy separation and recycling of the catalyst, significantly reducing metal contamination risks and operational costs compared to homogeneous systems.
Q: Can this process utilize carbon dioxide as a raw material effectively?
A: Yes, the patent describes a method where CO2 is fixed into the benzimidazole structure under mild pressure (0.1-2 MPa), offering a sustainable alternative to traditional carboxylic acid sources.
Q: Is this synthesis method scalable for industrial production?
A: The process operates at moderate temperatures (60-180°C) and uses a robust heterogeneous catalyst, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced catalytic technologies to deliver high-quality pharmaceutical intermediates to the global market. Our expertise in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet the demanding volume requirements of multinational corporations. We possess stringent purity specifications and rigorous QC labs that guarantee every batch of benzimidazole derivative meets the highest industry standards. Our technical team is well-versed in the nuances of heterogeneous catalysis and CO2 fixation, allowing us to optimize this specific patent technology for maximum efficiency and yield. By partnering with us, clients gain access to a supply chain that is not only cost-effective but also environmentally sustainable and technically robust.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. Request a Customized Cost-Saving Analysis to understand the specific economic benefits for your organization. We are prepared to provide specific COA data and route feasibility assessments to support your decision-making process. Our commitment to transparency and technical excellence ensures that you receive the best possible solution for your benzimidazole sourcing needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
