Advanced Catalytic Cyclization for High-Purity Benzimidazole and Derivatives Manufacturing
Advanced Catalytic Cyclization for High-Purity Benzimidazole and Derivatives Manufacturing
The pharmaceutical and agrochemical industries continuously demand more efficient, cost-effective, and environmentally benign synthetic routes for heterocyclic building blocks. Patent CN109265403B introduces a transformative methodology for the synthesis of benzimidazole and its derivatives, addressing critical bottlenecks in traditional manufacturing processes. This innovation leverages imidazole hydrochloride as a highly efficient organocatalyst to facilitate the cyclization of o-phenylenediamine with DMF or its derivatives. By eliminating the need for harsh mineral acids or expensive transition metal complexes, this technology provides a robust pathway for producing high-purity pharmaceutical intermediates. The process is characterized by exceptional operational simplicity, utilizing readily available solvents and reagents while delivering superior yields that often exceed 90 percent under optimized conditions. For R&D directors and procurement specialists, this represents a significant opportunity to streamline supply chains and reduce the cost of goods sold for key active pharmaceutical ingredient precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of benzimidazole scaffolds has relied heavily on classical condensation reactions that pose significant engineering and safety challenges. Traditional protocols frequently employ strong mineral acids such as concentrated hydrochloric acid or polyphosphoric acid to drive the cyclization of o-phenylenediamine with carboxylic acids. These acidic environments are notoriously corrosive, necessitating the use of specialized, expensive reactor linings and creating substantial maintenance overheads for manufacturing facilities. Furthermore, alternative oxidative cyclization methods using aldehydes often suffer from poor selectivity, generating complex impurity profiles that complicate downstream purification and reduce overall material throughput. Other reported methods involving Lewis acids or transition metal catalysts introduce the risk of heavy metal contamination, requiring rigorous and costly removal steps to meet stringent regulatory limits for drug substances. Additionally, some existing protocols require extreme conditions, such as high pressures of carbon dioxide or temperatures exceeding 150 degrees Celsius, which escalate energy consumption and safety risks during commercial scale-up of complex polymer additives or pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN109265403B utilizes a mild organocatalytic system centered on imidazole hydrochloride. This approach fundamentally shifts the reaction paradigm by activating the DMF solvent or amide reagent through hydrogen bonding or proton transfer mechanisms rather than brute-force acid catalysis. The reaction proceeds smoothly in polar aprotic solvents like DMF at moderate temperatures ranging from 120 to 140 degrees Celsius, significantly lowering the thermal load on the process. Crucially, the catalyst loading is remarkably low, typically between 0.1 and 0.3 equivalents relative to the diamine substrate, which minimizes reagent costs and waste generation. The method demonstrates exceptional versatility, capable of synthesizing both unsubstituted benzimidazoles and 2-substituted derivatives with high efficiency. By avoiding corrosive reagents and toxic metals, this novel route simplifies the workup procedure, often requiring only aqueous quenching and standard extraction, thereby enhancing the overall process mass intensity and sustainability profile for reliable agrochemical intermediate supplier operations.
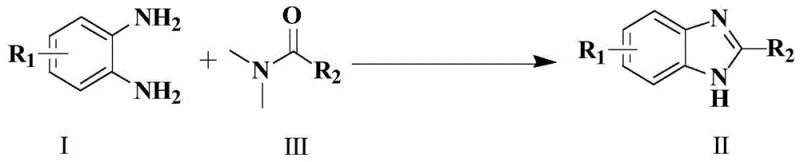
Mechanistic Insights into Imidazole Hydrochloride-Catalyzed Cyclization
The efficacy of this synthetic route lies in the unique ability of imidazole hydrochloride to activate the carbonyl species without degrading the sensitive amine functionality of the substrate. Mechanistically, the imidazolium cation likely acts as a Brønsted acid catalyst, protonating the oxygen of the DMF or amide carbonyl group to increase its electrophilicity. This activation facilitates the nucleophilic attack by one of the amino groups of the o-phenylenediamine, forming a key amidine intermediate. Subsequent intramolecular cyclization and dehydration steps lead to the formation of the benzimidazole ring system. The mild acidity of the imidazole hydrochloride is sufficient to drive this equilibrium forward but gentle enough to prevent the decomposition of acid-sensitive functional groups that might be present on the aromatic ring. This balance is critical for maintaining high purity specifications, as it suppresses the formation of polymeric byproducts or tar that are common in strongly acidic media. The reaction kinetics are favorable, with conversion typically reaching completion within 6 hours, allowing for high throughput in batch reactors.
Furthermore, the method exhibits remarkable functional group tolerance, which is a paramount concern for medicinal chemists designing diverse compound libraries. The catalytic system is compatible with a wide array of substituents on the phenylenediamine ring, including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as nitro and halogens. Experimental data indicates that substrates bearing bromine or chlorine atoms undergo cyclization efficiently, yielding the corresponding halogenated benzimidazoles in yields ranging from 85 to 88 percent. This tolerance extends to polycyclic systems as well, where naphthalene-2,3-diamine was successfully converted to the corresponding naphthimidazole derivative with an 87 percent yield. The ability to preserve these functional handles is essential for downstream diversification, allowing manufacturers to produce high-purity OLED material precursors or specialized kinase inhibitor intermediates without needing orthogonal protection strategies.
How to Synthesize Benzimidazole Derivatives Efficiently
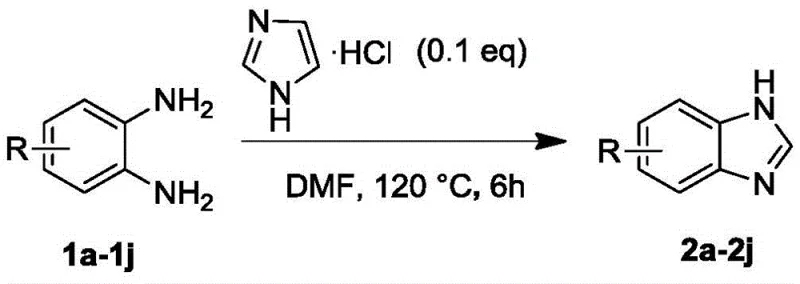
Implementing this catalytic protocol in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The general procedure involves charging a reaction vessel with the o-phenylenediamine substrate, the appropriate amide source (such as DMF or N,N-dimethylbenzamide), and the imidazole hydrochloride catalyst in a suitable solvent. The mixture is then heated to the optimal temperature range, typically between 120 and 140 degrees Celsius, and maintained under stirring until thin-layer chromatography confirms the disappearance of the starting material. Following the reaction, the mixture is cooled and quenched with water, allowing the product to be extracted into an organic phase such as ethyl acetate. The crude material can then be purified via silica gel column chromatography or recrystallization to afford the target benzimidazole derivative. Detailed standardized synthesis steps for specific derivatives are outlined below to ensure reproducibility and quality control.
- Charge a reaction vessel with o-phenylenediamine derivative, imidazole hydrochloride catalyst (0.1-0.3 equiv), and DMF solvent.
- Heat the mixture to 120-140°C and stir for approximately 6 hours until TLC indicates complete conversion.
- Quench with water, extract with ethyl acetate, wash organic layers, dry over sodium sulfate, and purify via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this imidazole hydrochloride-catalyzed synthesis offers compelling economic and logistical benefits for supply chain managers and procurement officers. The primary advantage stems from the drastic simplification of the reaction infrastructure required. By eliminating the need for corrosion-resistant reactors lined with glass or tantalum, which are mandatory for processes using concentrated hydrochloric acid or polyphosphoric acid, capital expenditure for new production lines can be significantly reduced. Furthermore, the absence of transition metal catalysts removes the necessity for expensive scavenging resins or complex filtration steps to meet residual metal specifications, directly lowering the cost of goods sold. The use of commodity chemicals like DMF and imidazole hydrochloride ensures a stable and reliable supply of raw materials, mitigating the risk of shortages associated with specialized reagents. This stability is crucial for maintaining continuous production schedules and meeting tight delivery windows for global clients.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of hazardous and expensive reagents. Traditional methods often require stoichiometric amounts of strong acids or oxidants, generating large volumes of acidic waste that incur high disposal costs. In contrast, the catalytic nature of the new method means only sub-stoichiometric amounts of imidazole hydrochloride are needed, reducing raw material consumption. Additionally, the simplified workup procedure reduces solvent usage and labor hours associated with purification. The high yields reported, often exceeding 90 percent for optimized substrates, mean less starting material is wasted, further enhancing the overall process economy. These factors combine to create a leaner manufacturing process that delivers substantial cost savings without compromising on product quality or purity standards required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, bulk chemical feedstocks. DMF and o-phenylenediamine are produced on a massive scale globally, ensuring that price volatility is minimized compared to niche catalysts or ligands. The robustness of the reaction conditions also implies a lower risk of batch failures due to minor fluctuations in temperature or reagent quality. This reliability translates to more predictable lead times for customers, a critical factor in the just-in-time manufacturing models prevalent in the pharmaceutical industry. By adopting this method, suppliers can offer more consistent availability of key intermediates, reducing the need for customers to hold excessive safety stock and freeing up working capital across the value chain.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the ton-scale often reveals hidden complexities, but this method has demonstrated promising scalability. The patent data explicitly mentions a successful scale-up to 20 grams with maintained high yields, suggesting a smooth path to kilogram and ton-scale production. The milder reaction conditions reduce the thermal hazard profile, making the process safer to operate in large reactors. From an environmental standpoint, the reduction in acidic waste and the avoidance of heavy metals align with green chemistry principles and increasingly stringent environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and enhances the sustainability credentials of the final product, which is becoming a key differentiator in B2B negotiations with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzimidazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction scope, scalability, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The information covers catalyst loading, solvent choices, and substrate compatibility to ensure a comprehensive understanding of the process capabilities.
Q: What are the advantages of using imidazole hydrochloride over traditional acid catalysts?
A: Imidazole hydrochloride offers milder reaction conditions compared to corrosive concentrated hydrochloric acid or polyphosphoric acid, reducing equipment corrosion risks and simplifying downstream purification while maintaining high yields up to 96%.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent demonstrates successful scalability, achieving a 90% isolated yield when the reaction was scaled up to 20 grams (180 mmol), indicating strong potential for commercial scale-up of complex pharmaceutical intermediates.
Q: Does the method tolerate sensitive functional groups like halogens?
A: The method exhibits excellent functional group tolerance, successfully accommodating electron-withdrawing groups such as bromine and chlorine, as well as electron-donating groups like methyl and methoxy, without significant loss in yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the imidazole hydrochloride-catalyzed cyclization in driving innovation for our partners. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including precise temperature control and efficient solvent recovery systems. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of benzimidazole intermediate meets the highest international standards. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, facilitating smoother regulatory filings for your downstream drug products.
We invite you to collaborate with us to leverage this cost-effective and robust synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with us, you gain access to deep process knowledge and the flexibility to adapt reaction conditions for optimal performance. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and competitive supply of high-quality benzimidazole derivatives for your global operations.
