Advanced Iridium-Catalyzed Asymmetric Hydrogenation for Commercial Scale-up of Complex Pharmaceutical Intermediates
Advanced Iridium-Catalyzed Asymmetric Hydrogenation for Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical industry continuously demands efficient routes to chiral heterocyclic scaffolds, particularly tetrahydroisoquinolines, which serve as critical structural motifs in numerous bioactive compounds. Patent CN103724264A introduces a groundbreaking method for synthesizing chiral tetrahydroisoquinoline derivatives through the asymmetric hydrogenation of isoquinoline salts catalyzed by an iridium complex. This technology addresses long-standing challenges in stereoselective synthesis, offering a robust pathway to high-value intermediates used in drugs such as Solifenacin, Cryptostyline II, and Salsolidine, as illustrated in the structural diversity shown below.
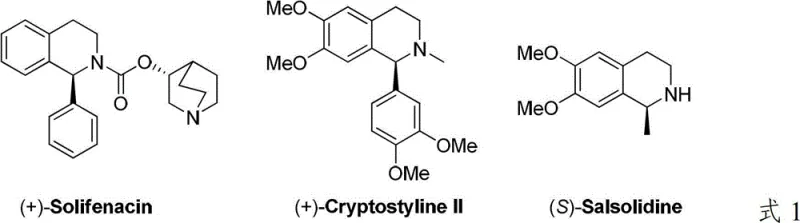
This innovation represents a significant leap forward for reliable pharmaceutical intermediate supplier networks, providing a method that combines high enantioselectivity with operational simplicity. By leveraging a chiral bisphosphine iridium catalyst system, the process achieves exceptional control over stereochemistry, ensuring the production of high-purity compounds essential for modern drug development. The technical breakthrough lies not only in the catalyst design but also in the strategic activation of the substrate, which overcomes the inherent stability and low reactivity of neutral isoquinoline rings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct asymmetric hydrogenation of isoquinolines has been plagued by significant technical hurdles that limit its utility in large-scale manufacturing. Neutral isoquinoline molecules possess stable aromatic properties and low hydrogenation activity, making them resistant to reduction under standard catalytic conditions. Furthermore, the nitrogen atom in the isoquinoline ring exhibits strong coordination ability, which frequently leads to the poisoning of transition metal catalysts, thereby deactivating the system before the reaction can proceed to completion. Previous attempts, such as those involving chloroformate activation, have yielded only moderate productivity and enantioselectivity, failing to meet the rigorous standards required for commercial API production.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by converting isoquinolines into isoquinolinium salts prior to hydrogenation. This activation strategy dramatically increases the electrophilicity of the heterocyclic ring, facilitating rapid and selective reduction. The general reaction scheme demonstrates how 1-position or 3-position substituted isoquinoline salts are transformed into their corresponding chiral tetrahydroisoquinoline derivatives with high efficiency.
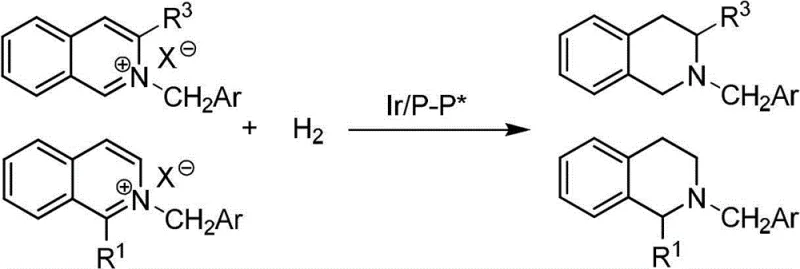
By employing this salt-based methodology, the process achieves reaction speeds that are significantly faster than traditional methods, while simultaneously minimizing side reactions. The result is a clean reaction profile where nuclear magnetic resonance spectroscopy detects virtually no impurities, simplifying downstream purification. This advancement supports cost reduction in pharmaceutical intermediate manufacturing by reducing waste and improving overall yield, making it an attractive option for procurement teams seeking sustainable supply chains.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The core of this technology relies on a sophisticated catalytic system comprising a (1,5-cyclooctadiene) iridium chloride dimer precursor and a specialized chiral bisphosphine ligand. The interaction between the iridium center and the chiral ligand creates a highly defined steric environment that dictates the facial selectivity of hydrogen addition to the substrate. Among the various ligands screened, (Rax,S,S)-C3*-TunePhos has emerged as a standout performer, enabling enantiomeric excess values to reach up to 96 percent. The mechanism involves the formation of a cationic iridium-hydride species that transfers hydrogen to the activated isoquinolinium ring in a concerted manner, ensuring precise stereocontrol at the newly formed chiral center.
Impurity control is inherently built into the reaction design due to the high specificity of the catalyst-substrate interaction. The use of isoquinolinium salts prevents the formation of over-reduced byproducts or regioisomers that often complicate the purification of neutral isoquinoline hydrogenations. Additionally, the reaction conditions are optimized to maintain catalyst stability throughout the process, with temperatures ranging from 25 to 60 degrees Celsius and hydrogen pressures between 13 and 50 atmospheres. This robustness ensures consistent batch-to-batch quality, a critical factor for regulatory compliance in the production of high-purity pharmaceutical intermediates.
How to Synthesize Chiral Tetrahydroisoquinoline Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing ease of operation and reproducibility. The process begins with the in situ generation of the active catalyst, followed by the introduction of the substrate under an inert atmosphere. Reaction monitoring is straightforward, and the workup procedure involves simple extraction and chromatography steps that are compatible with standard chemical processing equipment. For detailed operational parameters and specific examples of substrate scope, refer to the standardized synthesis steps provided in the technical guide below.
- Prepare the catalyst by stirring (1,5-cyclooctadiene) iridium chloride dimer with a chiral bisphosphine ligand in a mixed solvent of THF and dichloromethane.
- Transfer the catalyst solution to a reaction vessel containing the isoquinoline salt substrate and add additional mixed solvent to reach the desired concentration.
- Pressurize the autoclave with hydrogen gas (approx. 600 psi) and react at room temperature for 20-24 hours, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this iridium-catalyzed hydrogenation technology offers substantial benefits that align with the strategic goals of modern chemical procurement and supply chain management. The ability to produce chiral tetrahydroisoquinolines with high fidelity and minimal waste translates directly into improved cost structures and supply security. Unlike older methods that require harsh conditions or expensive stoichiometric chiral auxiliaries, this catalytic approach utilizes molecular hydrogen, the most atom-economical reducing agent available, thereby drastically simplifying the material balance of the process.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral reagents and the use of a highly active catalyst system significantly lower the raw material costs associated with chiral synthesis. Because the catalyst loading is relatively low compared to the substrate mass, and the reaction proceeds with near-quantitative conversion, the overall cost per kilogram of the final product is substantially reduced. Furthermore, the simplified purification process reduces solvent consumption and energy usage, contributing to a leaner and more cost-effective manufacturing operation without compromising on quality.
- Enhanced Supply Chain Reliability: The starting materials for this process, including isoquinolines and benzyl bromide derivatives, are readily available commodity chemicals, ensuring a stable and continuous supply of feedstock. The robustness of the catalytic system means that production schedules are less likely to be disrupted by sensitive reaction failures or inconsistent yields. This reliability is crucial for maintaining just-in-time inventory levels and meeting the demanding delivery timelines of downstream pharmaceutical clients who depend on consistent availability of key intermediates.
- Scalability and Environmental Compliance: The reaction operates under mild conditions, avoiding the need for extreme temperatures or pressures that often complicate scale-up efforts in large reactors. The use of common solvents like tetrahydrofuran and dichloromethane, which can be efficiently recovered and recycled, aligns with green chemistry principles and environmental regulations. This scalability ensures that the transition from laboratory discovery to multi-ton commercial production is seamless, supporting the long-term growth of product portfolios reliant on these chiral scaffolds.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on substrate scope, catalyst selection, and process safety. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for their specific target molecules.
Q: What are the primary advantages of using isoquinoline salts over neutral isoquinolines in this hydrogenation process?
A: Using isoquinoline salts significantly enhances reactivity and enantioselectivity compared to neutral isoquinolines, which often suffer from low hydrogenation activity and strong coordination abilities that poison catalysts. The salt activation strategy allows for faster reaction speeds and easier product separation with minimal side reactions.
Q: Which chiral ligands provide the highest enantiomeric excess in this iridium-catalyzed system?
A: The patent identifies several effective chiral bisphosphine ligands, with (Rax,S,S)-C3*-TunePhos demonstrating superior performance. This specific ligand, when combined with the iridium precursor, enables enantiomeric excess values reaching up to 96% for various 1-position or 3-position substituted derivatives.
Q: Is this synthesis method scalable for industrial manufacturing of pharmaceutical intermediates?
A: Yes, the method is designed for scalability, utilizing mild reaction conditions such as room temperature and moderate hydrogen pressure. The process features atom economy, environmentally friendly solvents, and simple operational procedures, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Tetrahydroisoquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the iridium-mediated asymmetric hydrogenation described in CN103724264A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you optimize your supply chain and accelerate your time to market.
