Advanced Semisynthetic Route for Taxol Intermediates: Scalability and Commercial Viability
The pharmaceutical industry continuously seeks robust and scalable methods for producing high-value anticancer agents, and patent CN1261360A presents a significant advancement in the semisynthetic production of Taxol and its derivatives. This intellectual property outlines a sophisticated chemical pathway that converts 9-dihydro-13-acetylbaccatin III into critical taxane intermediates, addressing the historical bottlenecks associated with natural extraction from yew tree bark. By shifting the production paradigm from destructive harvesting to chemical synthesis, this technology offers a viable solution for securing the global supply of paclitaxel, a cornerstone drug in oncology. The process leverages specific protection and oxidation strategies to ensure high fidelity in molecular structure, which is paramount for maintaining therapeutic efficacy. For R&D directors and supply chain leaders, understanding the nuances of this patent is essential for evaluating long-term sourcing strategies and mitigating the risks associated with raw material volatility. The methodology described herein not only enhances the purity profile of the resulting intermediates but also aligns with modern green chemistry principles by utilizing more accessible starting materials. As we delve into the technical specifics, it becomes clear that this route represents a mature, industrial-ready approach to complex molecule manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Taxol relied heavily on the extraction of paclitaxel directly from the bark of the Pacific yew tree, a method fraught with ecological and economic inefficiencies. The slow growth rate of the yew tree and the low concentration of the active compound in the bark meant that vast quantities of plant material were required to produce clinically relevant amounts of the drug, leading to significant environmental concerns and supply instability. Furthermore, the extraction process often yielded complex mixtures requiring extensive and costly purification steps to remove structurally similar impurities that could compromise patient safety. The dependency on a specific biological source made the supply chain vulnerable to agricultural variables, seasonal fluctuations, and geopolitical factors affecting the regions where yew trees grow. These conventional methods also struggled with batch-to-batch consistency, as the natural variation in plant metabolism could alter the impurity profile of the extracted material. Consequently, manufacturers faced challenges in meeting the stringent regulatory requirements for pharmaceutical grade active ingredients, often resulting in production delays and increased costs. The inability to scale extraction processes without causing ecological damage necessitated a shift towards more sustainable and controllable synthetic alternatives.
The Novel Approach
The methodology disclosed in patent CN1261360A introduces a transformative semisynthetic route that bypasses the limitations of direct extraction by utilizing 9-dihydro-13-acetylbaccatin III as a renewable starting material. This precursor can be obtained from the needles and twigs of yew trees, which are renewable biomass sources that do not require the destruction of the tree, thereby ensuring a sustainable and continuous supply chain. The process employs a strategic sequence of protection, oxidation, and side-chain addition that allows for precise control over the stereochemistry and functionalization of the taxane core. By chemically modifying the C-7 and C-9 positions, the synthesis achieves a level of purity and structural integrity that is difficult to attain through natural extraction alone. This approach significantly reduces the complexity of downstream purification, as the chemical steps are designed to minimize the formation of hard-to-remove byproducts. The flexibility of the route allows for the production of various taxol analogs, providing manufacturers with the ability to diversify their product portfolio based on market demand. Ultimately, this novel approach represents a paradigm shift towards a more reliable, scalable, and environmentally responsible manufacturing model for critical oncology drugs.
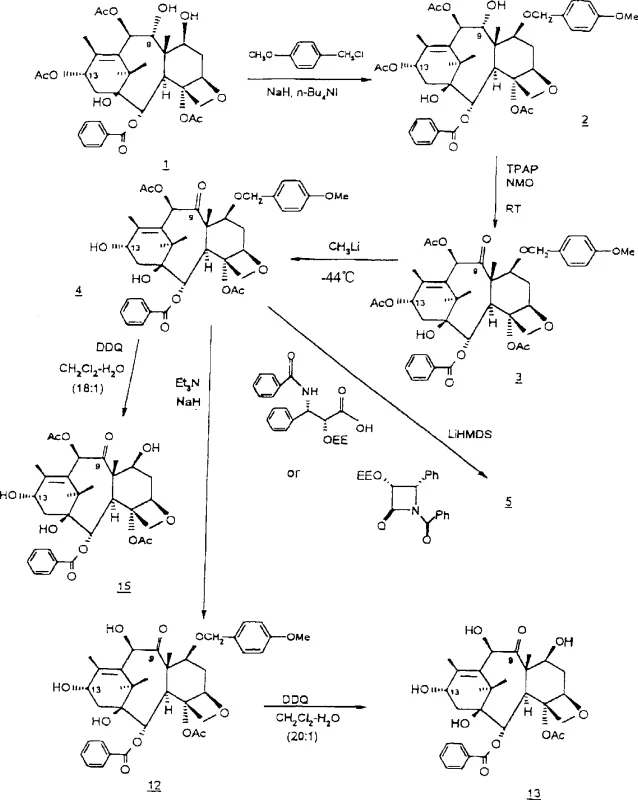
Mechanistic Insights into TPAP-Catalyzed Oxidation and Protection Strategy
The core of this synthetic innovation lies in the meticulous management of functional group reactivity, particularly through the use of specific protecting groups and mild oxidation conditions. The process begins with the protection of the C-7 hydroxy group using agents such as methoxy-benzyl chloride or tosyl chloride in the presence of a base like sodium hydride. This step is critical because it masks the C-7 hydroxyl, preventing it from participating in subsequent oxidation reactions and ensuring that the oxidant targets the C-9 position exclusively. The choice of protecting group is not arbitrary; groups like tosyl or methoxy-benzyl are selected for their stability under the oxidation conditions and their ease of removal in later stages without affecting the sensitive taxane ring system. Following protection, the C-9 hydroxy group is oxidized to a ketone using tetrapropylammonium perruthenate (TPAP) coupled with N-methylmorpholine N-oxide (NMO) as a co-oxidant. This catalytic system is preferred for its ability to operate under mild conditions, typically at room temperature or slightly elevated temperatures, which preserves the integrity of the complex polycyclic structure. The use of TPAP minimizes the risk of over-oxidation or degradation of other sensitive functional groups, such as the esters at C-10 and C-13, which are crucial for biological activity.
Following the oxidation step, the synthesis proceeds with the introduction of the C-13 side chain, a transformation that defines the biological activity of the final taxol molecule. This coupling reaction typically involves the use of a lithiated side chain precursor, such as a lithium hexamethyldisilazide (LiHMDS) activated beta-lactam or isoserine derivative, which attacks the C-13 carbonyl or displaces a leaving group depending on the specific intermediate. The stereochemistry of this addition is tightly controlled to ensure the formation of the correct (2'R, 3'S) configuration required for microtubule stabilization. After side-chain attachment, the protecting groups at the C-7 and 2' positions are removed using specific deprotection reagents like DDQ for methoxy-benzyl groups or mild acid hydrolysis for ethoxyethyl groups. This final deprotection sequence reveals the active hydroxyl groups necessary for the drug's mechanism of action while maintaining the overall yield and purity of the product. The entire mechanistic pathway is designed to maximize atom economy and minimize waste, aligning with the rigorous standards expected in modern pharmaceutical manufacturing.
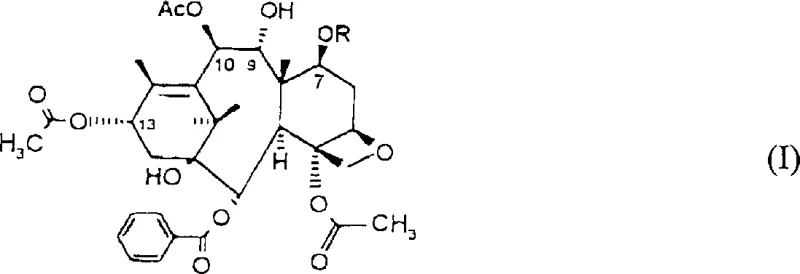
How to Synthesize Taxol Intermediates Efficiently
The implementation of this synthesis requires a deep understanding of the reaction parameters and a commitment to precise operational control to ensure consistent quality. The patent outlines a multi-step sequence that begins with the isolation of the starting material from renewable biomass, followed by a series of chemical transformations that must be monitored closely for completion and purity. Each step, from protection to oxidation and final coupling, contributes to the overall efficiency of the process, and optimizing these individual units is key to successful commercial scale-up. Operators must be trained to handle sensitive reagents like TPAP and LiHMDS under inert atmospheres to prevent degradation and ensure safety. The detailed standardized synthesis steps see the guide below.
- Protect the C-7 hydroxy group of 9-dihydro-13-acetylbaccatin III using a suitable protecting group such as methoxy-benzyl or tosyl.
- Oxidize the C-9 hydroxy group of the protected intermediate using oxidants like TPAP/NMO or Collins reagent.
- Add a suitable side chain to the C-13 position and perform deprotection steps to yield Taxol or its analogs.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the semisynthetic route described in CN1261360A offers substantial strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the decoupling of production from the volatile supply of yew bark, which has historically been a major bottleneck in the taxol market. By utilizing needles and twigs as the source of 9-dihydro-13-acetylbaccatin III, manufacturers can secure a more stable and predictable raw material supply that is less susceptible to seasonal and environmental fluctuations. This shift significantly enhances supply chain reliability, allowing for better long-term planning and inventory management without the fear of sudden raw material shortages. Furthermore, the chemical synthesis route allows for greater control over the production schedule, as reaction times and yields are more consistent compared to biological extraction processes. This predictability translates into reduced lead times for high-purity pharmaceutical intermediates, enabling companies to respond more agilely to market demands and regulatory changes.
- Cost Reduction in Manufacturing: The elimination of the need for massive quantities of yew bark significantly lowers the raw material acquisition costs and the logistical expenses associated with harvesting and transporting bulky plant material. Additionally, the use of catalytic oxidation systems like TPAP/NMO reduces the consumption of stoichiometric oxidants, which are often expensive and generate large amounts of hazardous waste. The streamlined purification process resulting from higher regioselectivity also reduces the consumption of solvents and chromatography media, further driving down the cost of goods sold. These cumulative efficiencies result in substantial cost savings that can be passed on to customers or reinvested into further R&D initiatives. The process avoids the use of expensive transition metal catalysts that require complex removal steps, thereby simplifying the downstream processing and reducing operational expenditures.
- Enhanced Supply Chain Reliability: Sourcing starting materials from renewable biomass like yew needles ensures a continuous supply that is not limited by the slow growth rate of the tree trunk. This renewable aspect mitigates the risk of supply disruptions caused by ecological restrictions or over-harvesting bans that have plagued the industry in the past. The ability to synthesize the drug from a stable intermediate allows for the establishment of multiple production sites globally, diversifying the supply base and reducing dependency on a single geographic region. This geographical diversification is crucial for maintaining business continuity in the face of geopolitical tensions or trade restrictions. Moreover, the chemical consistency of the synthetic route ensures that quality specifications are met consistently, reducing the risk of batch rejections and supply delays.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, utilizing reagents and conditions that are compatible with large-scale reactor systems commonly found in fine chemical manufacturing. The reduction in hazardous waste generation, particularly through the use of catalytic rather than stoichiometric oxidants, simplifies waste treatment and ensures compliance with increasingly stringent environmental regulations. The process avoids the generation of heavy metal waste streams that are difficult and costly to dispose of, aligning with the sustainability goals of modern pharmaceutical companies. This environmental compatibility not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturer. The robust nature of the chemistry allows for seamless scale-up from pilot plant to commercial production without significant re-optimization, accelerating time to market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis route. These answers are derived directly from the technical specifications and advantageous effects described in the patent documentation to provide clarity for potential partners. Understanding these details is crucial for making informed decisions about integrating this technology into your existing supply chain.
Q: What are the key advantages of this semisynthetic route over natural extraction?
A: This process utilizes 9-dihydro-13-acetylbaccatin III, which can be sourced from renewable plant materials like needles and twigs, avoiding the destructive harvesting of Pacific yew bark and ensuring a more sustainable and consistent supply chain.
Q: Which oxidizing agents are preferred for the C-9 oxidation step?
A: The patent specifies the use of tetrapropylammonium perruthenate (TPAP) with N-methylmorpholine N-oxide (NMO), Collins reagent, or activated DMSO, offering flexibility depending on available reagents and scale requirements.
Q: How is regioselectivity managed during the synthesis?
A: Regioselectivity is achieved by first protecting the C-7 hydroxy group with groups like tosyl or methoxy-benzyl, which prevents unwanted reactions at this position during the subsequent oxidation and side-chain coupling steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Taxol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable and high-quality intermediates in the development of life-saving oncology therapies. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex chemistry described in patent CN1261360A can be executed with precision and efficiency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our infrastructure is designed to handle the specific requirements of taxane synthesis, including the safe handling of sensitive reagents and the management of complex purification processes. By partnering with us, you gain access to a supply chain that is both resilient and responsive, capable of adapting to your specific volume and quality needs.
We invite you to discuss how our manufacturing capabilities can support your project goals and help you achieve a competitive advantage in the market. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate our commitment to transparency and technical excellence. Let us collaborate to optimize your supply chain and ensure the continuous availability of these critical pharmaceutical ingredients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
