Revolutionizing Alcohol and Pinacol Derivative Production via Metal-Free Electrocatalysis
Revolutionizing Alcohol and Pinacol Derivative Production via Metal-Free Electrocatalysis
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, driven by the urgent need to reduce environmental footprints and operational costs in the pharmaceutical industry. A groundbreaking development in this sector is detailed in patent CN114182272B, which discloses a novel preparation method for alcohol and pinacol derivatives utilizing electrocatalytic reduction. This technology represents a significant departure from conventional thermal and metal-catalyzed processes, offering a pathway to synthesize critical pharmaceutical intermediates under remarkably mild conditions. By leveraging electricity as a clean reagent, this method circumvents the use of stoichiometric metal hydrides or high-pressure hydrogen gas, thereby addressing long-standing safety and waste disposal challenges. For R&D directors and procurement managers seeking reliable sources of complex organic building blocks, this patent outlines a robust protocol that achieves yields of up to 95 percent while operating at room temperature and atmospheric pressure. The implications for supply chain stability and cost efficiency in the production of high-purity organic compounds are profound, positioning this electrocatalytic approach as a cornerstone for future manufacturing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the reduction of carbonyl compounds to alcohols or their dimerization to pinacols has relied heavily on methods that impose significant burdens on both the environment and the production budget. Classical approaches often necessitate the use of transition metal catalysts, such as palladium, platinum, or nickel, which not only carry high procurement costs but also introduce the risk of heavy metal contamination in the final product—a critical concern for API intermediate manufacturing. Furthermore, many of these reactions require stringent conditions, including elevated temperatures and high pressures, which escalate energy consumption and demand specialized, expensive reactor equipment. The reliance on chemical reducing agents like lithium aluminum hydride or sodium borohydride generates substantial amounts of inorganic waste, complicating downstream purification and waste treatment processes. Additionally, the functional group tolerance in these traditional methods is often limited, leading to unwanted side reactions that compromise yield and purity, thereby necessitating additional purification steps that further erode profit margins and extend lead times.
The Novel Approach
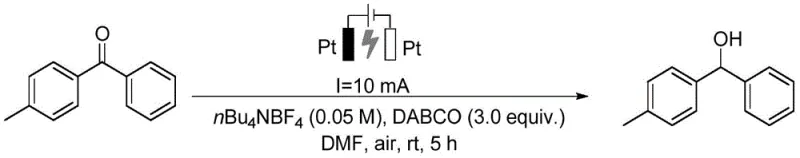
In stark contrast, the method described in patent CN114182272B introduces an elegant electrocatalytic system that fundamentally alters the reaction landscape by replacing chemical reductants with electrons. This innovative approach utilizes a simple undivided cell equipped with platinum electrodes, where the carbonyl substrate is reduced directly at the cathode surface. The process operates efficiently at room temperature and under an air atmosphere, eliminating the need for inert gas protection or energy-intensive heating systems. A key feature of this novelty is the use of DABCO (1,4-diazabicyclo[2.2.2]octane) as a sacrificial anode additive, which facilitates the reaction without the need for transition metals. This metal-free protocol not only simplifies the reaction setup but also ensures that the resulting alcohol or pinacol derivatives are free from toxic metal residues, a distinct advantage for cost reduction in pharmaceutical intermediate manufacturing. The method's versatility is demonstrated across a wide range of substrates, including aryl ketones and aldehydes, proving its robustness as a general synthetic tool for diverse chemical architectures.
Mechanistic Insights into Electrocatalytic Carbonyl Reduction
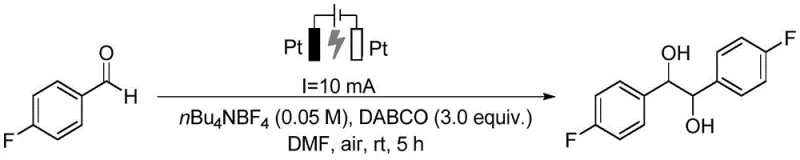
To fully appreciate the technical superiority of this method, one must delve into the mechanistic underpinnings of the electrocatalytic reduction process. The reaction initiates at the cathode, where the carbonyl compound accepts an electron to form a radical anion intermediate. In the presence of a proton source, typically derived from the solvent or trace water, this radical anion is protonated to generate a neutral radical species. For the synthesis of simple alcohol derivatives, a second electron transfer followed by protonation yields the final hydroxyl product. However, the mechanism can diverge to form pinacol derivatives through the homocoupling of two ketyl radical intermediates, a pathway that is highly dependent on the substrate structure and reaction conditions. The use of DABCO is critical here; it acts as an electron donor at the anode, preventing the oxidation of the desired product and maintaining the overall charge balance of the system. This intricate interplay of electron transfer and protonation allows for precise control over the reaction outcome, enabling the selective formation of either mono-reduced alcohols or dimerized pinacols.
Furthermore, the impurity profile of products generated via this electrocatalytic route is exceptionally clean compared to metal-catalyzed counterparts. The absence of transition metals eliminates the formation of metal-complexed byproducts, which are notoriously difficult to remove and often require specialized scavenging resins. The mild reaction conditions also suppress thermal degradation pathways and unwanted rearrangements that frequently plague high-temperature syntheses. For instance, sensitive functional groups such as halides or ethers, which might be susceptible to hydrodehalogenation or cleavage under harsh catalytic hydrogenation conditions, remain intact in this electrochemical environment. This high level of chemoselectivity ensures that the crude reaction mixture contains fewer impurities, streamlining the purification process and enhancing the overall mass balance of the synthesis. Such mechanistic elegance translates directly into higher operational efficiency and reduced material loss, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Alcohol and Pinacol Derivatives Efficiently
Implementing this electrocatalytic methodology in a laboratory or pilot plant setting requires adherence to a standardized protocol that maximizes yield and reproducibility. The process begins with the preparation of a homogeneous reaction mixture containing the carbonyl substrate, the tertiary amine additive, and the supporting electrolyte in an organic solvent. Following the electrochemical conversion, a straightforward workup procedure involving aqueous quenching and organic extraction isolates the crude product, which is then purified to meet stringent quality standards. The detailed operational parameters, including electrode spacing, current density, and reaction duration, are critical for achieving the reported high yields and must be carefully controlled. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized steps outlined below.
- Mix the carbonyl compound substrate with tertiary amine (DABCO), electrolyte (nBu4NBF4), and organic solvent (DMF) to form a homogeneous solution.
- Perform the electrocatalytic reaction at room temperature and constant current (10 mA) using platinum electrodes under an air atmosphere for 5 to 10 hours.
- Quench the reaction with saturated brine, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrocatalytic technology offers transformative benefits for procurement and supply chain management within the fine chemical sector. The elimination of expensive transition metal catalysts and the reduction in energy requirements directly contribute to a leaner cost structure, allowing for more competitive pricing of high-value intermediates. Moreover, the simplicity of the reaction setup and the use of readily available raw materials enhance supply chain resilience, mitigating risks associated with the scarcity of specialized reagents. The ability to operate under ambient conditions also reduces the capital expenditure required for specialized high-pressure reactors, facilitating faster deployment of production capacity. These factors collectively strengthen the supply continuity for critical drug substances, ensuring that downstream manufacturing schedules are met without interruption.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven primarily by the removal of costly catalytic systems and the minimization of waste treatment expenses. By utilizing electricity as the primary reductant, the process avoids the procurement of stoichiometric amounts of metal hydrides or hydrogen gas, which represent significant variable costs in traditional synthesis. Additionally, the simplified workup procedure reduces the consumption of solvents and purification media, further lowering the operational expenditure per kilogram of product. This efficiency gain allows manufacturers to offer substantial cost savings to their clients without compromising on the quality or purity of the final chemical entities.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the method's reliance on commodity chemicals such as DABCO and common organic solvents, which are widely available from multiple global suppliers. Unlike processes dependent on proprietary catalysts or rare earth metals, this electrocatalytic route is not vulnerable to geopolitical supply disruptions or price volatility in the precious metals market. The robustness of the reaction conditions also means that production can be maintained consistently across different facilities, ensuring a steady flow of materials to meet fluctuating market demands. This reliability is crucial for maintaining the integrity of long-term supply agreements with major pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling this process from benchtop to industrial production is facilitated by the modular nature of electrochemical reactors, which can be easily expanded or operated in continuous flow modes to increase throughput. The environmentally benign nature of the process, characterized by the absence of heavy metal waste and lower energy consumption, aligns perfectly with increasingly strict global environmental regulations. This compliance reduces the regulatory burden and potential liabilities associated with hazardous waste disposal, making the technology a sustainable choice for long-term manufacturing strategies. Consequently, companies adopting this method can position themselves as leaders in green chemistry, appealing to eco-conscious stakeholders and customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrocatalytic synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on the practical aspects of the technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows.
Q: What are the primary advantages of this electrocatalytic method over traditional metal catalysis?
A: This method eliminates the need for expensive transition metal catalysts and harsh conditions like high temperature and pressure. It operates at room temperature under air, significantly reducing energy consumption and environmental impact while maintaining high yields up to 95%.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes standard electrochemical setups with platinum electrodes and common solvents like DMF. The mild conditions and simple workup procedure involving extraction and chromatography make it highly suitable for commercial scale-up in fine chemical manufacturing.
Q: What types of substrates are compatible with this synthesis route?
A: The method demonstrates broad substrate scope, successfully converting various aryl ketones, aldehydes, and heteroaryl carbonyl compounds into their corresponding alcohol or pinacol derivatives with excellent functional group tolerance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alcohol and Pinacol Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced electrocatalytic technologies in reshaping the production of high-value chemical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of alcohol or pinacol derivative meets the exacting standards required by the global pharmaceutical industry. We are dedicated to leveraging cutting-edge synthesis techniques to deliver superior products that drive our clients' success.
We invite you to collaborate with us to explore how this efficient electrocatalytic method can optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can support your journey from discovery to commercialization.
