Advanced BPDA Manufacturing: Technical Upgrades and Commercial Scalability for Global Supply Chains
The global demand for high-performance polyimides in flexible electronics and aerospace applications has necessitated a rigorous re-evaluation of monomer synthesis pathways. Patent CN101659647B introduces a transformative method for preparing biphenyltetracarboxylic dianhydride (BPDA), a critical precursor for heat-resistant polymers. This technical disclosure outlines a four-step sequence starting from o-xylene, bypassing the traditional reliance on costly chlorinated phthalic anhydride. By integrating halogenation, coupling, oxidation, and dehydration, the process achieves a total yield of 50% to 60% with exceptional purity profiles. For R&D Directors and Procurement Managers, this represents a significant opportunity to optimize supply chain resilience while maintaining stringent quality standards required for advanced material applications. The strategic shift to o-xylene as a feedstock not only simplifies the operational workflow but also drastically reduces the economic barriers associated with high-purity electronic chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biphenyltetracarboxylic dianhydride has been heavily dependent on 4-chlorophthalic anhydride as the primary starting material, a constraint that has long plagued the economic viability of large-scale production. The market price for 4-chlorophthalic anhydride is notoriously volatile and expensive, creating substantial cost pressure for downstream polymer manufacturers who operate on thin margins. Furthermore, conventional routes often involve multi-step syntheses to generate the chlorinated precursor from phthalic anhydride or o-xylene, introducing unnecessary complexity and increasing the cumulative loss of material at each stage. Alternative methods utilizing helical organic nickel catalysts with Grignard reagents have been explored, yet these demand extremely harsh reaction conditions and specialized equipment that are difficult to maintain in a standard industrial setting. The cumulative effect of these limitations is a supply chain that is fragile, costly, and incapable of meeting the surging demand for high-performance polyimide films in the electronics sector without significant price premiums.
The Novel Approach
The patented methodology fundamentally restructures the synthesis logic by initiating the process directly with o-xylene, a commodity chemical with abundant global availability and stable pricing structures. This novel approach eliminates the need for pre-synthesized chlorinated anhydrides, thereby cutting out an entire tier of expensive raw material procurement and logistics. The process flow is streamlined into four distinct, manageable stages that utilize cost-effective oxidants like potassium permanganate instead of precious metal catalysts or high-pressure hydrogenation systems. By coupling first and then oxidizing, the route avoids the formation of difficult-to-remove impurities that typically arise from direct oxidation of mixed xylenes. This strategic redesign not only simplifies the operational requirements for plant engineers but also ensures that the final product meets the rigorous purity specifications demanded by the semiconductor and display industries. The result is a robust, economically superior pathway that aligns perfectly with the goals of cost reduction in display material manufacturing.
Mechanistic Insights into Ni-Catalyzed Coupling and Permanganate Oxidation
The core of this synthesis lies in the nickel-catalyzed coupling reaction, which transforms halogenated xylene into 3,3',4,4'-tetramethylbiphenyl with high selectivity and efficiency. In this critical step, anhydrous nickel chloride and triphenylphosphine act in concert with zinc powder as a reducing agent within an aprotic polar solvent such as DMF or NMP. The presence of 2,2'-bipyridine as a promoter is essential, as it stabilizes the nickel complex and significantly enhances the yield of the coupling reaction, pushing conversion rates to optimal levels between 75% and 80%. The reaction is conducted under a nitrogen atmosphere at temperatures ranging from 60°C to 90°C, conditions that are mild enough to prevent thermal degradation of the intermediates yet sufficient to drive the reductive coupling to completion. Following the reaction, the crude product undergoes recrystallization using methanol, a purification step that elevates the purity of the tetramethylbiphenyl intermediate to greater than 99%, ensuring that downstream oxidation proceeds without interference from isomeric impurities.
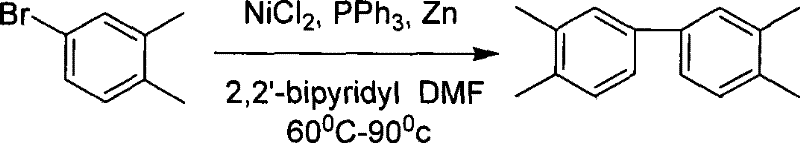
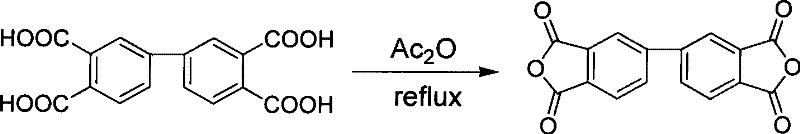
Subsequent oxidation of the tetramethylbiphenyl intermediate utilizes a two-stage potassium permanganate system that balances reactivity with selectivity to produce the tetracarboxylic acid. The first stage involves reaction with potassium permanganate in a pyridine and water mixture, which provides an alkaline environment conducive to the initial oxidation of the methyl groups. The second stage introduces sodium hydroxide and additional oxidant to ensure complete conversion of all methyl functionalities to carboxylic acids, a process that requires precise temperature control between 100°C and 130°C over 24 to 36 hours. Following oxidation, the mixture is acidified with 20% hydrochloric acid to precipitate the biphenyltetracarboxylic acid, a step designed to minimize the release of hazardous HCl gas while maximizing recovery. The final dehydration using acetic anhydride under reflux conditions cyclizes the acid into the dianhydride, achieving a yield of 90% to 95% with a melting range of 298°C to 301°C, indicative of a highly crystalline and pure final product suitable for polymerization.
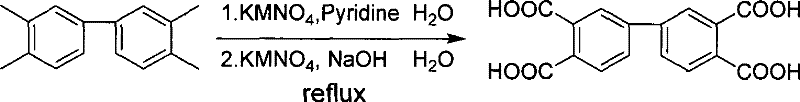
How to Synthesize Biphenyltetracarboxylic Dianhydride Efficiently
Implementing this synthesis route requires careful attention to the halogenation and dehydration steps to ensure maximum yield and safety during scale-up. The initial halogenation of o-xylene must be conducted at low temperatures between -20°C and 10°C using bromine with iron and iodine catalysts to control the isomer distribution effectively. High-efficiency rectification is then employed to isolate the 4-bromo isomer, which is critical for the subsequent coupling reaction to proceed with high regioselectivity. The final dehydration step utilizes acetic anhydride under nitrogen protection to convert the tetracarboxylic acid into the dianhydride, a process that must be monitored to prevent thermal decomposition while ensuring complete cyclization. Detailed standardized synthesis steps see the guide below for specific operational parameters and safety protocols.
- Perform halogenation of o-xylene using bromine with iron and iodine catalysts at controlled low temperatures to obtain halogenated xylene.
- Execute nickel-catalyzed coupling of halogenated xylene with triphenylphosphine and zinc powder to form 3,3',4,4'-tetramethylbiphenyl.
- Oxidize the tetramethylbiphenyl intermediate using potassium permanganate in pyridine and sodium hydroxide solutions, followed by acidification and dehydration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis pathway offers profound strategic advantages that extend beyond simple unit cost savings. By shifting the raw material base to o-xylene, manufacturers can decouple their production costs from the volatile pricing of specialized chlorinated anhydrides, leading to significantly reduced exposure to market fluctuations. The simplification of the process flow, characterized by the elimination of high-pressure equipment and expensive transition metal catalysts, translates directly into lower capital expenditure and reduced maintenance overheads for production facilities. Furthermore, the use of common reagents like potassium permanganate and acetic anhydride ensures that supply chain continuity is maintained even during periods of global chemical shortages. This robustness is essential for maintaining reliable electronic chemical supplier status in a market where downtime can result in substantial contractual penalties and loss of market share.
- Cost Reduction in Manufacturing: The elimination of 4-chlorophthalic anhydride as a starting material removes a major cost driver from the bill of materials, allowing for substantial cost savings in the final product pricing. Additionally, the avoidance of expensive transition metal catalysts and the use of simple oxidants like potassium permanganate reduce the cost of goods sold by minimizing the need for complex catalyst recovery and regeneration systems. The simplified process flow also reduces energy consumption and labor hours per kilogram of product, further enhancing the overall economic efficiency of the manufacturing operation. These cumulative effects create a competitive pricing structure that allows suppliers to offer high-purity polyimide precursors at more attractive price points without compromising margin integrity.
- Enhanced Supply Chain Reliability: Sourcing o-xylene and potassium permanganate is significantly more straightforward than procuring specialized chlorinated intermediates, which are often produced by a limited number of vendors. This broadens the supplier base and reduces the risk of supply disruptions caused by single-source dependencies or geopolitical instability in specific chemical sectors. The robustness of the synthesis route also means that production can be easily shifted between different manufacturing sites without the need for highly specialized equipment, enhancing the flexibility of the supply network. For supply chain heads, this translates to reducing lead time for high-purity polymer monomers and ensuring consistent delivery schedules to downstream polymerization customers.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex electronic chemicals, as it avoids the use of hazardous high-pressure hydrogenation or toxic heavy metal catalysts that complicate regulatory compliance. The waste streams generated are primarily inorganic salts and organic solvents that can be managed using standard treatment protocols, reducing the environmental footprint of the manufacturing site. The mild reaction conditions also lower the risk of thermal runaway incidents, enhancing plant safety and reducing insurance costs associated with chemical manufacturing. This alignment with environmental and safety standards facilitates faster regulatory approvals and smoother operations in jurisdictions with strict chemical management laws.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this BPDA synthesis route in an industrial setting. These answers are derived directly from the patent data to ensure accuracy and relevance for technical decision-makers evaluating the feasibility of this technology. Understanding these details is crucial for assessing the potential impact on your current production capabilities and supply chain strategy.
Q: What are the primary cost advantages of this BPDA synthesis route compared to conventional methods?
A: This method utilizes o-xylene as a starting material instead of expensive 4-chlorophthalic anhydride, significantly reducing raw material costs. Additionally, the use of potassium permanganate as an oxidant avoids the need for costly catalysts and high-pressure equipment, simplifying the overall production process and lowering capital expenditure.
Q: How does the process ensure high purity suitable for electronic grade polyimides?
A: The process incorporates a high-efficiency rectification step during halogenation to increase the 4-position isomer concentration to over 87%. Furthermore, recrystallization of the tetramethylbiphenyl intermediate with methanol ensures purity greater than 99%, which carries through to the final dianhydride product with purity levels reaching 99.5% to 99.8%.
Q: Is this synthesis method scalable for industrial production volumes?
A: Yes, the method is designed for industrial scalability by avoiding harsh reaction conditions and expensive equipment. The total yield of 50% to 60% and the use of common reagents like potassium permanganate and acetic anhydride make it economically viable for large-scale manufacturing of high-performance polyimide monomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyltetracarboxylic Dianhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity monomers play in the performance of next-generation polyimide materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are translated into reliable industrial reality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of BPDA meets the exacting standards required for electronic and aerospace applications. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply chain for high-performance polymer precursors.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific manufacturing needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic improvements for your operation. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will help you make informed decisions about your raw material strategy. Let us help you engineer a more efficient and cost-effective supply chain for your critical chemical needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
